Depression and other psychiatric conditions with a mood component are common and debilitating chronic illnesses that are often difficult to treat. A high prevalence of vitamin D insufficiency has been reported among most Western societies Reference Mithal, Wahl, Bonjour, Burckhardt, Dawson-Hughes and Eisman1 and the level of insufficiency is most marked among elderly people. Reference Pasco, Henry, Nicholson, Sanders and Kotowicz2–Reference Park and Johnson4 In Australia, the prevalence of insufficiency (25-hydroxy-vitamin D (25D) below 50 nmol/l) is approximately 50% among older community-dwelling women. Reference Pasco, Henry, Nicholson, Sanders and Kotowicz2,Reference Pasco, Henry, Kotowicz, Sanders, Seeman and Pasco5 Clinical psychiatric cohorts may have even higher rates of vitamin D insufficiency. Reference Berk, Jacka, Williams, Ng, Dodd and Pasco6 In particular, it is postulated that low levels of vitamin D, known to be common in winter, Reference Mithal, Wahl, Bonjour, Burckhardt, Dawson-Hughes and Eisman1,Reference Pasco, Henry, Kotowicz, Sanders, Seeman and Pasco5 may contribute to seasonal affective disorder Reference Young7 and that older adults with vitamin D deficiency are more likely to have depressed mood. Reference Milaneschi, Shardell, Corsi, Vazzana, Bandelli and Guralnik8–Reference Berk, Dodd, Williams, Jacka and Pasco10
Recent advances in the understanding of neural physiology strengthen the argument for a link between vitamin D status and mental health. Using animal models, cross-talk between vitamin D and glucocorticoids has been demonstrated in the hippocampus, and these signalling pathways are germane to disorders with dysregulated glucocorticoid signalling such as depression. Reference Obradovic, Gronemeyer, Lutz and Rein11 The enzymes vitamin D 25-hydroxylase and 25D-1α-hydroxylase, necessary for the hydroxylation of 25D to active 1,25-dihydroxyvitamin D (1,25(OH)2D) are present in various areas of the human brain including the hypothalamus, cerebellum, substantia nigra as well as retinal neurons. Reference Obradovic, Gronemeyer, Lutz and Rein11 Locally produced 1,25(OH)2D has a documented role in brain function. Reference Garcion, Wion-Bardot, Montero-Menei, Berger and Didier12
Since vitamin D insufficiency is a treatable condition, vitamin D repletion in older adults may prevent or improve depressive symptoms. Despite epidemiological and in vitro evidence supporting a positive association between vitamin D levels and mental health there remains a clear paucity of evidence from large clinical trials. Reference Young7 Among the small intervention trials there is heterogeneity in dose and inconsistent, albeit, suggestive findings. Reference Wilkins, Sheline, Roe, Birge and Morris13–Reference Dumville, Miles, Porthouse, Cockayne, Saxon and King18 The Vital D study (ISRCTN83409867 and ACTR12605000658617) is the first large randomised placebo-controlled trial to investigate the impact of vitamin D supplementation on mood and depressive symptoms in a community-based cohort of older women. Furthermore, the utility of a single large annual dose of vitamin D is also investigated since previous trials reporting no benefit in the prevention of fractures have suggested that the likelihood of positive findings were hampered by low to moderate adherence to treatment regimes. Reference Grant, Anderson, Avenell, Campbell, Cooper and Donaldson19–Reference Porthouse, Cockayne, King, Saxon, Steele and Aspray21
The aims of the mental well-being study were to determine in community-dwelling older women whether a single annual dose of vitamin D supplementation has a beneficial effect on mood and is a practical health intervention for the prevention of depressive symptoms; and whether there is an association between serum vitamin D levels and mood and well-being. The study's hypothesis was that older women treated with a large annual dose of vitamin D (500 000 IU cholecalciferol) would perform better than women treated with placebo on mood-related psychiatric assessments. In a biochemistry substudy we hypothesised higher serum 25D levels would be predictive of better mental well-being compared with women with a lower vitamin D status.
Method
Study design
The Vital D study was a single-centre, large-scale double-blind, randomised, placebo-controlled trial involving women 70 years or older. The participants were randomly assigned to receive either an oral dose of vitamin D3 500 000 IU or matched placebo once each year for 3–5 years (in the autumn/winter). The primary end-point was falls and fractures, and participants were selected on the basis of hip fracture risk. Reference Sanders, Stuart, Merriman, Read, Kotowicz and Young22 These results have been reported elsewhere. Reference Sanders, Stuart, Williamson, Simpson, Kotowicz and Young23 The Vital D study concurrently investigated the effect of the vitamin D3 supplementation on mental well-being. This analysis relates to the three sets of mental well-being data that were prospectively collected.
The study was approved by the institutional ethical research boards of Barwon Health and the University of Melbourne and carried out in compliance with the Helsinki Declaration. An early protocol amendment enabled baseline data on mental well-being to be collected from participants recruited to the study during 2005. All women provided written informed consent and those enrolled in the substudy consented to the extra assessments.
Participants
The Vital D Study recruited 2317 community-dwelling women between 2003 and 2005. Reference Sanders, Stuart, Merriman, Read, Kotowicz and Young22 Invitation letters were sent to all women aged at least 70 years on the electoral roll of the Barwon and Mornington Peninsula regions (latitude approximately 38° South). Voting and voter registration is compulsory in Australia. Inclusion criteria has been previously described, but briefly included females ≥70 years who had an identified risk factor for hip fracture such as maternal hip fracture, self-reported ‘faller’, fracture since aged 50, and/or at high risk of low vitamin D and osteoporosis. Reference Cummings, Nevitt, Browner, Stone, Fox and Ensrud24 The exclusion criteria were: inability to provide informed consent or falls/fracture data; permanent resident of high-level care facility; albumin-corrected calcium >2.65 mmol/l; vitamin D supplementation >400 IU/day; and other parameters relating to bone health.
Randomisation and intervention
Eligible participants were randomised to receive either 500 000 IU vitamin D3 or placebo. The allocation of treatment arm was performed by an independent statistician using computer randomisation of participants’ study identification number (Minitab). The participants and study staff were masked to the treatment allocation until completion of the study.
Identical tablets of placebo and 50 000 IU vitamin D3 were supplied by PSM Healthcare, Auckland, New Zealand. Study medication (10 tablets) was posted to participants annually during autumn/winter (March to August, determined by recruitment month), with instructions to take all 10 tablets on 1 day. Study staff confirmed ingestion by written or verbal confirmation within 2 weeks of postage.
Outcome measures
Age, calcium intake and fracture risk profile were documented at baseline in all participants. Three sets of mental well-being outcomes were included in the study. The General Health Questionnaire (GHQ) Reference Goldberg25 was completed at three time points: baseline, 12 months post-dose (immediately prior to the second dose of study medication) and at 15 months post-dose (3 months after the second dose). The data do not include the first 693 participants recruited into the study as the GHQ was added after a protocol amendment in April 2005. As baseline GHQ data were not available for these participants the GHQ data include all participants except those recruited prior to this amendment. The 12-item Short Form Health Survey (SF-12) Reference Ware, M, Turner-Bowker and Gandeck26 questionnaire provided physical and mental component summary scores and was completed once by all participants immediately prior to their study completion in 2008, corresponding to an intervention period of 3–5 years.
The third set of mental health outcomes was collected only in a subgroup of participants who underwent serial biochemistry including serum 25D. These substudy participants were randomly selected from the entire group of participants and completed all the study data collection requirements at the same time points as all other participants. Additional mental well-being questionnaires were completed to coincide with the serial assessments of 25D status done in this subgroup of participants (as described below).
Substudy participants: biochemistry and mental well-being testing
Substudy participants (n = 150) were randomly selected (masked to treatment status) using their study identification numbers in a statistical program (Minitab, version 13 for Windows). These participants completed the two mood-related psychiatric questionnaires at six time points concurrently, with blood sampling during 2006 and 2007. The World Health Organization (WHO) Well-Being Index Reference Bonsignore, Barkow, Jessen and Heun27 and the Patient Global Impression Improvement (PGI-I) scale Reference Yalcin and Bump28 questionnaires were completed and serum levels of 25D were assessed at: pre-dose (immediately prior to the current dose of study medication and corresponding to 12 months following last year's annual dose), 1 and 3 months post-dose of study medication. Samples were frozen at –80°C until being batch analysed. Serum 25D was measured using radioimmunoassay (DiaSorin; Stillwater, Minnesota, USA).
Scoring of mental well-being assessments
The GHQ-12 questionnaire Reference Goldberg25 is a well-validated instrument for the identification and measurement of non-psychotic psychiatric disorders in community and medical settings. Reference Goldberg, Gater, Sartorius, Ustun, Piccinelli and Gureje29 The self-reported GHQ-12 consists of 12 questions, each with a choice of four responses: ‘better than usual’ or ‘same as usual’ are both scored as ‘0’, whereas ‘less than usual’ or ‘much less than usual’ are both scored as ‘1’. Using the GHQ scoring method there is a maximum total score of 12 and a cut-off threshold of 3 analysed as a binary ‘yes/no’ outcome. A total score of 3 or greater has been reported as being indicative of a psychiatric disorder. Reference Goldberg, Gater, Sartorius, Ustun, Piccinelli and Gureje29
The SF-12 consists of 12 questions with standardised categorical answers and is a widely used health survey relevant across all ages and disease categories. Data were scored according to the users’ manual Reference Ware, M, Turner-Bowker and Gandeck26 providing both a physical component summary score and a mental component summary score for each individual.
The WHO Well-Being Index (version 2, 1998) Reference Bonsignore, Barkow, Jessen and Heun27 is a 5-item self-report questionnaire, each question with a choice of five responses: ‘all of the time’ (scores 5); ‘most of the time’ (scores 4); ‘more than half of the time’ (scores 3); ‘less than half of the time’ (scores 2); ‘some of the time’ (scores 1); or ‘at no time’ (scores 0). The score for each question was totalled. Individuals were considered to have poor well-being and depression if their total score was <13 or if they answered 0 or 1 for any of the five items. 30
The PGI-I scale is a validated adaptation of the Clinical Global Impression Scale Reference Yalcin and Bump28 that consists of one question that was altered slightly as indicated in italics: ‘Circle the number that best describes how you have felt overall since you began taking the Vital D Study medication.’ There are seven answer choices ranging from ‘very much better’ (score, 1) to ‘very much worse’ (score, 7): this score was used in the analysis.
Sample size
The power calculations for the Vital D study were based on the primary outcome relating to a difference in fracture rates between the treatment groups. Nevertheless, given the large cohort required for the primary outcome, the study is adequately powered to detect a 50% relative difference in the proportion of participants with a score of three or greater in the GHQ questionnaire when 15% of the placebo group score three or greater (80% power and 0.05 level of significance). Reference Walter31
Statistical methods
All analyses were intention-to-treat. The physical and mental component summary scores from the SF-12 were compared between groups using t-tests. Since participants completed the GHQ, WHO Well-Being Index and the PGI-I scale on multiple occasions, a generalised estimating equation approach with robust standard errors and exchangeable correlation was used to allow for within-participant correlation. A linear link function was used for the WHO Well-Being score, and a logistic link function for the dichotomised WHO Poor Well-Being Index and the dichotomised GHQ score. For the GHQ score, baseline GHQ was adjusted for, to increase the statistical precision of the analysis. Results are presented as estimated differences in means (SF-12, WHO Well-Being, PGI-I scale) or estimated odds ratios (binary GHQ, binary WHO score) and 95% confidence intervals with Wald-test P-values.
To investigate the a priori hypothesis that the effect of vitamin D on mental well-being would differ by baseline antidepressant use, an interaction between trial arm and antidepressant was added to each of the models above. Wald tests for the interaction are presented with estimates of treatment effect within each subgroup. No adjustment was made for multiple testing. All P-values are two-sided. Analyses were performed in Stata 10.1 for Windows.
Results
Randomisation of participants, assessments and the number of participants who withdrew are shown inFig. 1. There were no differences between the vitamin D and placebo groups in age or baseline use of antidepressants/anxiety medication (Table 1). Ingestion of study medication was high; over the course of the study 58 participants (33 in the vitamin D group and 25 in the placebo group) declined study medication and study medication was not dispensed to105 participants (44 in the vitamin D group and 61 in the placebo group) as they reported taking greater than 400 IU of vitamin D supplementation. These participants were included in the intention-to-treat analysis.
Table 1 Baseline characteristics of Vital D participants

| Vitamin D group | Placebo group | |
|---|---|---|
| All participants: SF-12 at study completion (2008), n | 1001 | 1011 |
| Age at baseline, years: median (IQR) | 75.8 (72.9–79.9) | 75.9 (72.9–79.2) |
| Use of anti-anxiety/antidepressants, % (n) | 14.2 (128) | 15.4 (138) |
| Participants + GHQ (recruited in 2005), n | 728 | 706 |
| Age at baseline, years: median (IQR) | 76.4 (73.1–80.5) | 76.3 (73.1–79.8) |
| Use of anti-anxiety/antidepressants, % (n) | 14.3 (96) | 16.2 (105) |
| Baseline GHQ-12, score of ≥3, % (n) | 14.0 (102) | 15.0 (106) |
| Biochemical substudy (WHO Well-Being Index and PGI-I), n | 61 | 57 |
| Age at baseline, years: median (IQR) | 74.5 (72.6–77.9) | 75.0 (72.9–80.4) |
| 25-hydroxyvitamin D, nmol/l: median (IQR) | 53 (4065) | 45 (4057) |
| Use of anxiety/antidepressants, % (n) | 14.8 (9) | 8.8 (5) |
GHQ-12
In total, 92% (n = 1434) of the 1565 Vital D participants recruited in 2005 completed the GHQ-12 at baseline. The proportion of participants in either treatment group with a psychiatric disorder as indicated by a GHQ score of ≥3 varied between 14 and 18% across the three time points (Table 2). There was no difference in scores between the vitamin D and placebo groups at any time point (P = 0.69,Table 3). There was no evidence of an interaction between treatment arm and use of antidepressant/anxiety medication.
Table 2 Outcome measures from 12-item Short Form Health Survey (SF-12) and General Health Questionnaire (GHQ)

| Vitamin D group | Placebo group | |
|---|---|---|
| All participants: SF-12, n | 1001 | 1011 |
| SF-12 (completed in 2008) | ||
| Physical component score (normed), mean (s.d.) | 41.4 (11.3) | 41.2 (10.9) |
| Mental component score (normed), mean (s.d.) | 52.5 (9.8) | 52.6 (9.9) |
| GHQ, n | 728 | 706 |
| GHQ-12, score of ≥3, indicator for psychiatric disorder | ||
| Baseline (2005), % (n) | 14.0 (102) | 15.0 (106) |
| 12 months post-dose (2006), % (n) | 13.8 (91) | 13.7 (89) |
| 15 months post-dose (2006), % (n) | 17.7 (117) | 16.9 (108) |
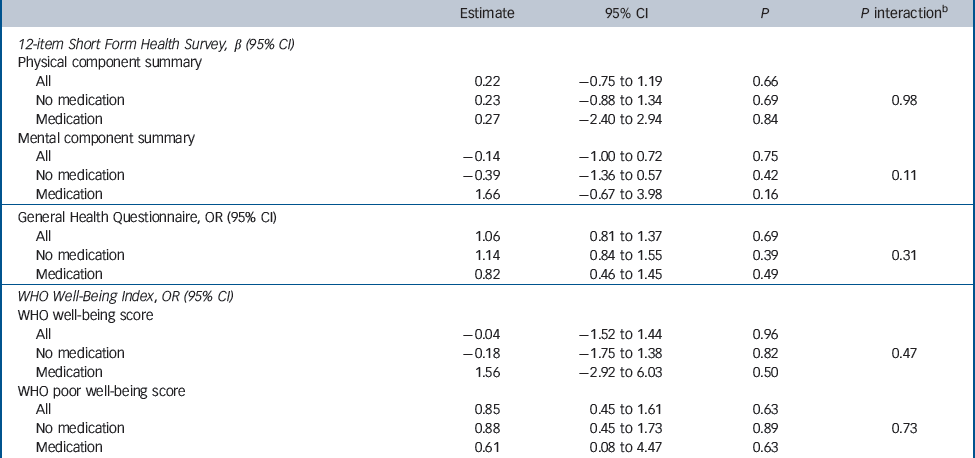
Table 3 Estimated treatment effects: results from the questionnaires of the vitamin D group compared with placebo groupa
| Estimate | 95% CI | P | P interactionb | |
|---|---|---|---|---|
| 12-item Short Form Health Survey, β (95% CI) | ||||
| Physical component summary | ||||
| All | 0.22 | – 0.75 to 1.19 | 0.66 | |
| No medication | 0.23 | – 0.88 to 1.34 | 0.69 | 0.98 |
| Medication | 0.27 | – 2.40 to 2.94 | 0.84 | |
| Mental component summary | ||||
| All | – 0.14 | – 1.00 to 0.72 | 0.75 | |
| No medication | – 0.39 | – 1.36 to 0.57 | 0.42 | 0.11 |
| Medication | 1.66 | – 0.67 to 3.98 | 0.16 | |
| General Health Questionnaire, OR (95% CI) | ||||
| All | 1.06 | 0.81 to 1.37 | 0.69 | |
| No medication | 1.14 | 0.84 to 1.55 | 0.39 | 0.31 |
| Medication | 0.82 | 0.46 to 1.45 | 0.49 | |
| WHO Well-Being Index, OR (95% CI) | ||||
| WHO well-being score | ||||
| All | – 0.04 | – 1.52 to 1.44 | 0.96 | |
| No medication | – 0.18 | – 1.75 to 1.38 | 0.82 | 0.47 |
| Medication | 1.56 | – 2.92 to 6.03 | 0.50 | |
| WHO poor well-being score | ||||
| All | 0.85 | 0.45 to 1.61 | 0.63 | |
| No medication | 0.88 | 0.45 to 1.73 | 0.89 | 0.73 |
| Medication | 0.61 | 0.08 to 4.47 | 0.63 |
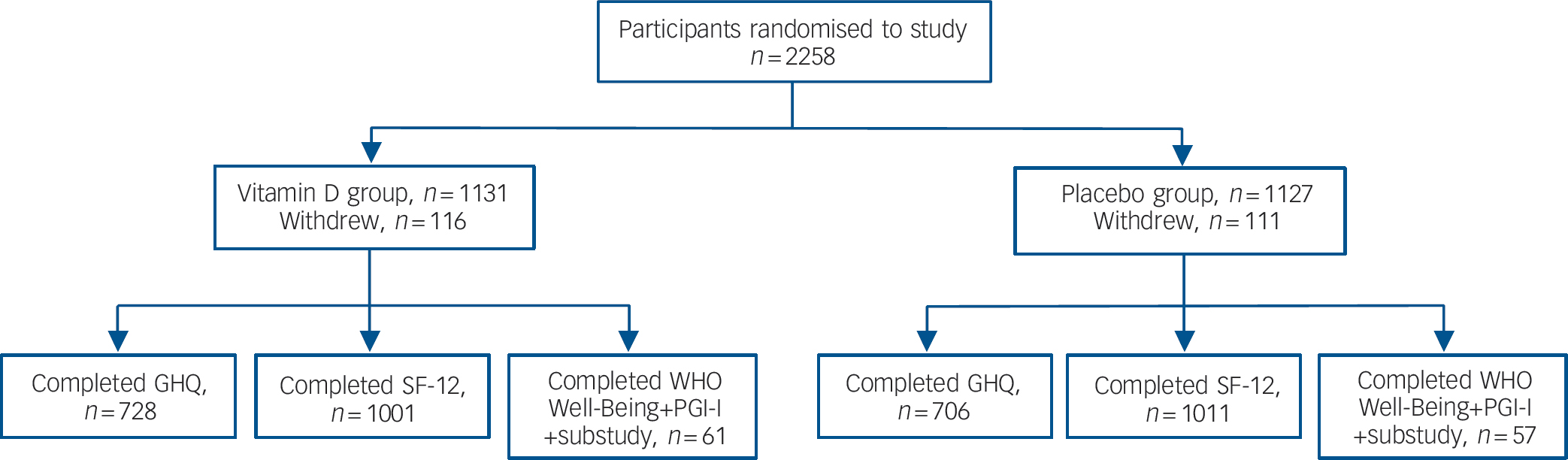
Fig. 1 Participant randomisation and completion of assessments.
GHQ, General Health Questionnaire; PGI-I, Patient Global Impression Improvement scale; SF-12, 12-item Short Form Health Survey; WHO Well-Being, World Health Organization Well-Being Index. The GHQ was completed only by participants recruited in 2005 following protocol amendment. The biochemistry substudy: 150 participants were randomly selected for the substudy; 137 accepted and complete data of biochemistry and questionnaires at the three time points were available for 118 participants.
SF-12
At study completion, 99% (n = 2016/2031) of participants completed the SF-12. There was no difference between the vitamin D and placebo groups in the mean scores from either the physical (41.4 v. 41.2, P = 0.66) or the mental component summary (52.5 v. 52.6, P = 0.75,Table 2). There was no interaction between treatment groups and use of antidepressant/anxiety medication, although there was a trend for those taking medication for treatment of anxiety or depression to score higher (i.e. better) on the SF-12 mental component summary if they were randomised to receive vitamin D rather than placebo (P = 0.11,Table 3). However, there was no significant difference between the groups in the proportion of participants from each group who either commenced antidepressant/anxiety medication during the study or ceased antidepressants during the intervention period (vitamin D v. placebo group: commenced these medications: 5.2% v. 7.1%, P = 0.12; proportion of participants taking these medications at baseline and ceased them during the intervention period: 24.6% v. 25.4%, P = 0.89).
WHO Well-Being Index and PGI-I scale
In total, 91% (137/150) of those invited consented to the biochemistry substudy. The analysis included 118 participants (61 in the vitamin D group and 57 in placebo group) who completed at least one questionnaire. There were no differences in the scores from the WHO Well-Being questionnaires at any time point (P = 0.47,Table 3). No interaction trend was found between treatment group and use of antidepressant/anxiety medication. Those taking medication for treatment of anxiety or depression did not score differently on the WHO Well-Being Index if they were randomised to receive vitamin D or placebo (Table 3 andFig. 2). There was no difference between the vitamin D and placebo groups at any of the time points corresponding to pre-dose, 1 month and 3 months post-dose. Furthermore, no differences between the treatment groups were found when using the binary scale to indicate poor well-being and depression (i.e. score <13, P = 0.73). Similarly, there was no difference on the PGI-I scale between the vitamin D and placebo groups (P = 0.52).
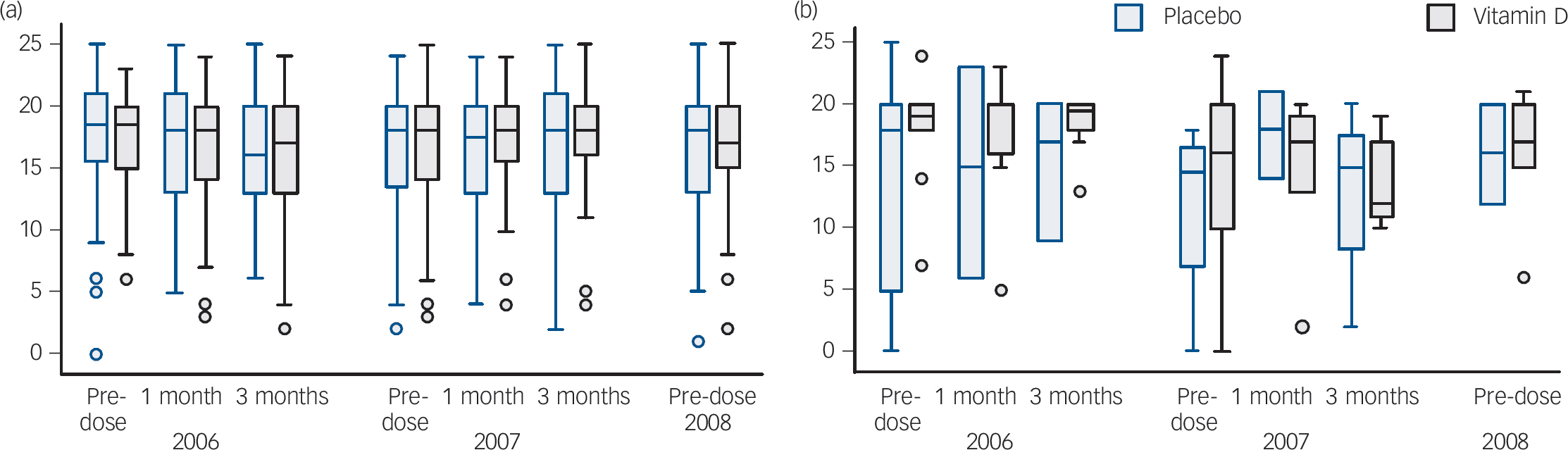
Fig. 2 WHO Well-Being Index score for (a) those not taking anti-anxiety or antidepressant medication, and (b) those taking these medications at baseline.
All participants had had at least one dose of study medication prior to 2006. The pre-dose label refers to the day participants took the current year's dose of study medication (i.e. 12 months since their last dose of study medication). Note participants completed the study in 2008 so no study medication was taken at this time point. The boxes refer to the interquartile range, the continuous lines show the range and the dots show outliers.
Biochemical changes in serum 25D
There was no difference in serum 25D between the groups at baseline (Table 1). Approximately half the substudy participants had serum 25D of 50 nmol/l or lower (46 and 61%, vitamin D and placebo groups respectively) but less than 3% had levels <25 nmol/l (1.8 and 2.7% respectively).
Detailed biochemistry data were collected on 102 (86%) of these substudy participants during 2006 and 2007, coinciding with their second to fifth annual dose of study medication. Immediately prior to the annual dose of study medication, the vitamin D group had mean serum 25D levels 41% higher than the placebo group (ratio of geometric means 1.41 95% CI 1.27–1.57; P<0.001) at approximately 70 nmol/l. One month post-dose the mean serum 25D levels of the vitamin D group was approximately 122 nmol/l (median 2006 and 2007 respectively: 121 and 124 nmol/l). By 3 months post-dose the median 25D level decreased to approximately 90 nmol/L (median 2006 and 2007 respectively: 87 and 94 nmol/l).
Relationship between serum 25D and mental well-being
There was no significant relationship between serum 25D levels and the assessments of mental well-being completed at the time of blood sampling (Fig. 3). Despite the changes in serum 25D levels (Fig. 3(a)), there was no evidence of any apparent trends between serum 25D and the scores from either the WHO Well-Being Index or the PGI-I scale (see 1-month post-dose dataFig. 4(a) and 4(b)).
Adverse events
A similar number of participants reported an adverse event in each group: 19.7% (223/1131) in the vitamin D group and 17.8% (200/1125) in the placebo group. Overall, 33 participants (2.9%) from the vitamin D group and 22 participants (1.9%) from the placebo group reported more than one adverse event. The most commonly reported adverse events were ‘injury including fracture’ (172/1131 vitamin D v. 136/1125 placebo, P = 0.06) and cardiovascular events (17/1131 vitamin D v. 13/1125 placebo).

Fig. 3 Substudy group data. (a) Serum 25-hydroxyvitamin D (25(OH)D) levels, (b) score from the World Health Organization (WHO) Well-Being Index, and (c) score from the Patient Global Impression Improvement (PGI-I) scale.
The blood sampling was done on the same day that substudy participants completed the WHO Well-Being Index and the PGI-I scale. The x-axis refers to time in months since the annual dose of study medication. The ‘0’ is 12 months since their last dose of study medication. Participants had their study medication immediately following the blood sampling. The boxes refer to the interquartile range, the continuous lines show the range and the dots show outliers.
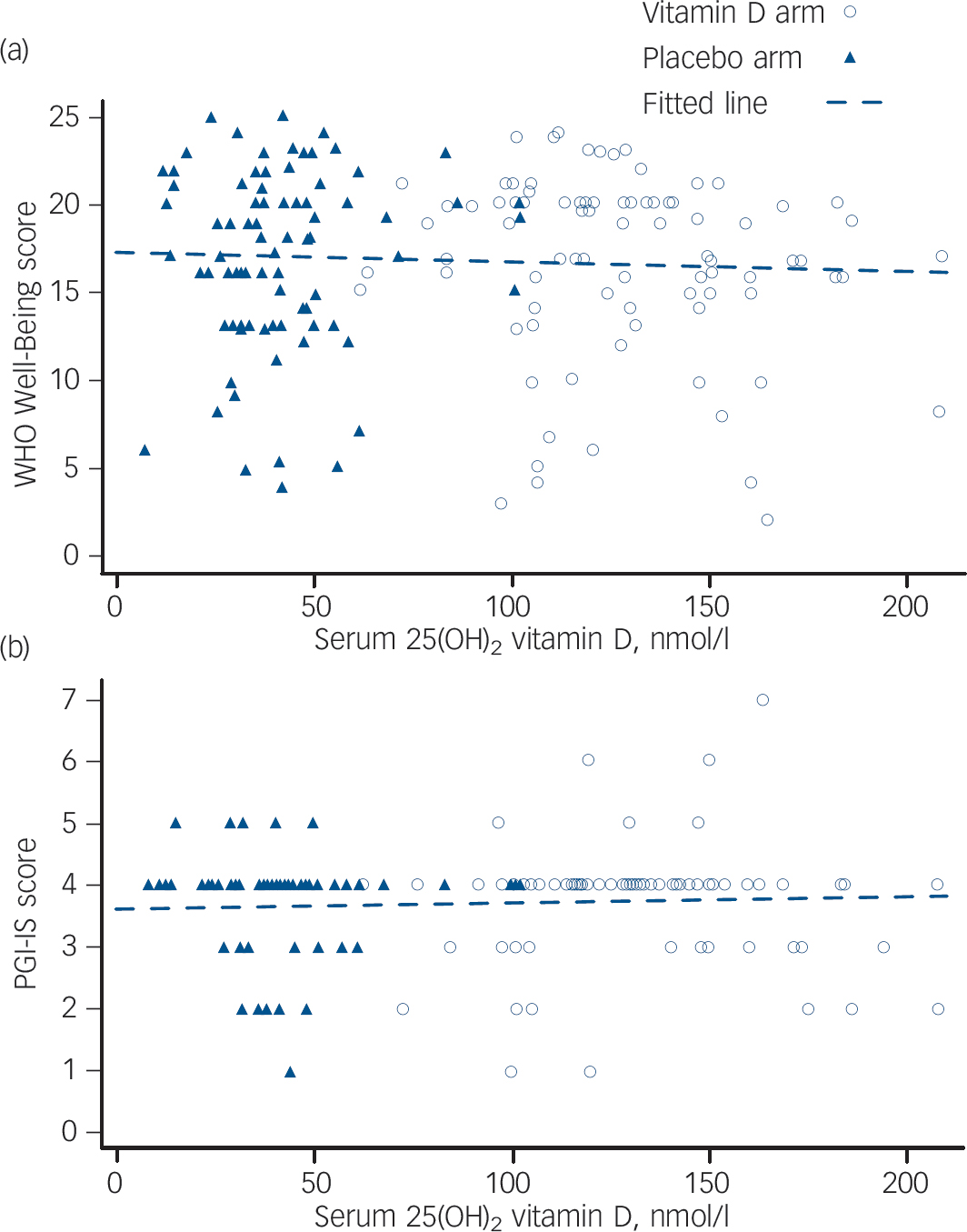
Fig. 4 Scatterplots for (a) World Health Organization (WHO) Well-Being Index score against serum vitamin D at 1 month post-dose, and (b) Patient Global Impression Improvement (PGI-I) scale score against serum vitamin D at 1 month post-dose.
There was no significant difference between groups in the number of serious adverse events: 244 vitamin D v. 207 placebo (P = 0.06). Reference Sanders, Stuart, Williamson, Simpson, Kotowicz and Young23 Over the course of the study, 87 participants died: 40 (3.5%) from the vitamin D group and 47 (4.2%) from the placebo group (P = 0.45). None of the serious adverse events were considered related to the study medication.
Discussion
Main findings
In this large randomised trial investigating the potential benefits of a single, annual dose of 500 000 IU vitamin D3, no consistent significant differences were detected between the vitamin D and placebo groups in any of the measured outcomes of mental health. Results from the GHQ-12 indicate that fewer than 20% of the cohort of almost 1600 older women had scores consistent with a non-psychotic psychiatric disorder, and this proportion did not differ between the groups at any of the three assessments (baseline, 12 months and 15 months post-dose). Similarly, the mental health and the physical health component summary scores from the SF-12 did not differ between the vitamin D and placebo groups when administered 12 months post-dose and after participants had ingested at least three annual doses of study medication. There was a non-significant trend for those taking medication for treatment of anxiety or depression to have improved scores on the SF-12 mental health component summary if they received vitamin D rather than placebo; however, this was not a consistently observed pattern.
The relationship between serum vitamin D status and mental well-being was tested in more detail in the nested subset of participants who had serial biochemistry samples taken at baseline and during the intervention period. Biochemistry results demonstrate that serum 25D were: low to moderate at baseline and typical of this population of community-dwelling older women; Reference Pasco, Henry, Nicholson, Sanders and Kotowicz2 remained low to moderate in the placebo group; initially spiked in the vitamin D group (approximately 120 nmol/l at 1-month post-dose) and remained in the high to normal range at 12-month post-dose (approximately 70 nmol/l). At each blood sampling taken 12 months post-dose, serum 25D levels were, on average, 41% higher in the vitamin D group compared with the placebo group. Despite these biochemical changes, vitamin D status was not a significant predictor of mental well-being when assessed using either the WHO Well-Being Index or the PGI-I scale scores. Since there were no significant differences in mental health outcomes measured 1, 3 or 12 months following the annual dose of study medication, the indices of mental well-being did not appear to be influenced by the abrupt changes in serum 25D levels as demonstrated in our nested substudy.
Findings from other studies
There are inconsistent findings on the association between vitamin D and mental health. Reference Garcion, Wion-Bardot, Montero-Menei, Berger and Didier12,Reference Dumville, Miles, Porthouse, Cockayne, Saxon and King18,Reference Arvold, Odean, Dornfeld, Regal, Arvold and Karwoski32 Epidemiological studies have reported associations between vitamin D insufficiency and the presence of mood disorder and depression. A low level of serum 25D was significantly associated with a high depression score in a small observational study. Similarly, no difference in questionnaire scores (Fibromyalgia Impact Questionnaire) were detected using 400 000 IU vitamin D3 over 8 weeks when given to individuals with frank vitamin D deficiency (serum 25D, less than 10 ng/ml (25 nmol/l)) nor when given to a group randomised to receive 400 000 IU vitamin D3 or placebo. Reference Arvold, Odean, Dornfeld, Regal, Arvold and Karwoski32 Although serum 25D levels increased in these two groups by a mean of 93 and 68 nmol/l respectively, there was no significant improvement in depression (including seasonal), fatigue or musculoskeletal symptoms. Our results demonstrating no improvement in mental health scores are also consistent with those of a UK randomised unmasked trial of 1621 women aged 70 years and older. Reference Dumville, Miles, Porthouse, Cockayne, Saxon and King18 Participants completed the SF-12 questionnaire both at baseline and at 6 months. Those randomised to the treatment group received 800 IU vitamin D3 and 1000 mg calcium each day. Women allocated to the control group did not receive any study medication or placebo. There was no difference in the mental component summary score between the groups at 6 months after adjusting for age and baseline mental component summary score (P = 0.262). In a small study of individuals comparing 100 000 IU of vitamin D with phototherapy for seasonal affective disorder, depressive symptoms improved in those who received vitamin D, but not the phototherapy group. Reference Gloth, Alam and Hollis17 Following the administration of vitamin D3 in late winter, improved positive affect and reduced negative affect has been reported. Reference Lansdowne and Provost33 In a randomised trial of overweight or obese people, high-dose vitamin D (20 000 or 40 000 IU per week) for 1 year had a beneficial effect on depressive symptoms compared with the placebo group; however, the study's cross-sectional analysis suggested a threshold effect. In those with baseline 25D levels > 40 nmol/l there was no apparent change in the group receiving the vitamin D. Although we only have 25D levels in a subset of 137 participants, few of these Vital D participants were clearly deficient at baseline with the interquartile range of 25D being 40–63 nmol/l. A review of the evidence linking vitamin D deficiency to brain dysfunction concludes that direct effects of vitamin D inadequacy on cognition/behaviour appear to be subtle with current experimental evidence not fully satisfying casual criteria. Reference McCann and Ames34
Although currently available evidence strongly suggest a role for 1,25(OH)2D in the nervous system, Garcion and colleagues have postulated that 1,25(OH)2D functions might overlap with other signalling pathways and may be protected in the central nervous system. Reference Garcion, Wion-Bardot, Montero-Menei, Berger and Didier12 The role of 1,25(OH)D therefore might not be apparent in the absence of other pathogenic processes. Similarly, other signalling pathways relevant to psychological end-points may overlap with no obvious phenotypic symptoms. Interestingly, a recent population-based case–control study has reported a U-shaped relationship between neonatal 25D status and risk of developing schizophrenia by the age of 24 years. Reference McGarth, Eyles, Pedersen, Anderson, Ko and Burne35 The relative risk was increased for neonates in each of the three lowest quintiles of 25D. The authors report that neonates in the highest quintile also had an increased risk of later developing schizophrenia. Our Vital D study was not powered in regards to the 25D levels of the substudy to detect a U-shaped response to the questionnaire data.
Limitations
We acknowledge that the apparent lack of mental health benefit in the vitamin D group may be confounded by the unexpected increased rate of falls and fractures in this group compared with placebo. As previously reported Reference Sanders, Stuart, Williamson, Simpson, Kotowicz and Young23 the vitamin D group had a 15% higher rate of falling (incidence rate ratio (IRR) = 1.15, 95% CI 1.02–1.30, P = 0.003) and 26% higher rate of fracture (IRR = 1.26, 95% CI 1.00–1.59, P = 0.047) compared with the placebo group. Biochemistry results from our substudy suggest that only a very small proportion of participants were clearly in the vitamin D deficient range (serum 25D <25 nmol/l) and about half of our substudy participants had baseline 25D levels above the insufficiency threshold of 50 nmol/l. Furthermore, changes in outcome measures may have occurred outside the visit schedule. Moreover, the outcome measures may not have been sufficiently sensitive to changes in mental well-being. The therapeutic response of a non-clinical sample may differ substantially from clinical effects evident in a group of individuals with depression. However, the community-based study sample provides a novel perspective of the issue. The major advantage of the Vital D study is the population-based recruitment of a large number of participants and the 3 to 5-year intervention period. The study design adequately addressed the adherence issue for vitamin D supplementation.
Implications
Whereas findings from in vitro studies suggest a plausible link between depressive mood and vitamin D insufficiency, results from observational and small intervention studies have been inconsistent and the evidence base to date is not sufficient to conclude with certainty that there is a causal connection. Results from this large randomised intervention trial do not support a therapeutic role for annual high-dose supplementation of vitamin D in older women and did not contradict a hypothesis that other compensatory neural pathways are activated in the presence of low vitamin D status. In conclusion, this study found no benefit of vitamin D supplementation for mood and general well-being in older women. The study therefore does not support the hypothesis that annual doses of the ‘sunshine’ vitamin are a practical intervention to prevent depressive symptoms in older community-dwelling women. The results of this pragmatically designed trial highlight the need for further understanding of the mechanisms and pharmacokinetics of vitamin D in relation to mental health. Clinical trials of vitamin D in clinical populations with documented insufficiency remain warranted.
Funding
This study was funded through a 1-year project grant from the Australian National Health and Medical Research Council (NHMRC No. ). Initially, the Vital D study was funded through a 5-year NHMRC project grant (NHMRC No. ), with additional funding aimed specifically at recruitment granted through the Government Department of Health and Ageing.
Acknowledgements
We thank Paul Muir (clinical trials pharmacist at Barwon Health) and others who have worked on the Vital D team. Most importantly, we gratefully acknowledge the 2031 women who participated in the Vital D study between 2003 and 2008.










eLetters
No eLetters have been published for this article.