Introduction
During foetal life, human oocytes often become arrested in prophase I of meiosis. The oocytes are still in the dictyate phase at birth, and each ovary has more than 500,000 healthy, primordial follicles (Varghese et al., Reference Varghese, Peter and Kamath2021). Cohorts of oocytes are taken out of this non-growing pool and start to develop during the woman’s reproductive life. The initial follicular development phase, which produces a few layers of granulosa cells (GCs) surrounding the oocyte, is primarily characterized by a rise in oocyte size. After oocytes develop their cytoplasm, follicular development focuses on the proliferation and differentiation of GCs (Tukur et al., Reference Tukur, Aljumaah, Swelum, Alowaimer and Saadeldin2020). In follicles, this differentiation promotes the development of antral cavities. In the antral phase, which is brought on by the anterior pituitary’s release of follicle-stimulating hormone (FSH), fluid gathers between GCs. A central cavity is formed, with the mural GCs situated on the perimeter (Gougeon, Reference Gougeon2010). The oocyte remains surrounded by closely related granulosa cells, referred to as cumulus cells, forming the compact cumulus–oocyte complex (Turathum et al., Reference Turathum, Gao and Chian2021). At this stage of development, gonadotrophin is needed for the follicles to continue to grow. From the early antral stage until the preovulatory stage, the follicles grow under the control of FSH (Hsueh et al., Reference Hsueh, Kawamura, Cheng and Fauser2015). The pre-ovulatory surge of luteinizing hormone (LH) triggers germinal vesicle breakdown (GVBD) at the late follicular phase (middle of the menstrual cycle), and chromosomes move from metaphase I to telophase I (Arroyo et al., Reference Arroyo, Kim and Yeh2020). The extrusion of the first polar body and the development of the secondary oocytes, both of which contain a diploid chromosomal complement, signify the end of the first meiotic division. After the first meiotic division is finished, the second one is quickly started, and the oocytes reach the metaphase II stage before ovulation (Levi et al., Reference Levi, Kaplan-Kraicer and Shalgi2011).
Oocyte maturation is basically divided into nuclear and cytoplasmic maturation. Nuclear maturation is the process of the resumption of meiosis and advancing on to metaphase II. MII Cytoplasmic maturation is known as the preparation of oocyte cytoplasm for fertilization and embryonic development (Trebichalská et al., Reference Trebichalská, Kyjovská, Kloudová, Otevřel, Hampl and Holubcová2021). These two processes, meanwhile, are not entirely distinct from one another. By controlling cytoplasmic maturation, nuclear maturation is regulated.
Despite the fact that immature oocytes taken from antral follicles may resume meiosis in vitro, cytoplasmic maturation seems to happen asynchronously with nuclear maturation (Mandelbaum et al., Reference Mandelbaum, Awadalla, Smith, Violette, Klooster, Danis, McGinnis, Ho, Bendikson and Paulson2021). This is most likely the major cause of the reduced embryo generation rates seen when oocytes are matured in vitro. Therefore, knowledge of the oocyte maturation process is crucial in order to improve the success rate of in vitro embryo formation and provide therapies for different types of infertility.
In this review, we will take a look at the mechanisms, signals mediating local control and the changes that occur during the oocyte maturation process.
Nuclear maturation
The process of nuclear maturation initiates when meiosis resumes in the GV oocyte from the diplotene stage, which is marked by chromosomal condensation and GVBD. Following GVBD, the oocyte passes through metaphase I (MI), anaphase I (AI) and telophase I (TI) until finally completing the first meiotic division. Then quickly advances through MII of the second meiotic division, where a second meiotic arrest takes place (second meiotic blockage) (He et al., Reference He, Zhang, Yang and Wang2021).
The structure of the nuclear membrane, including the GV, is preserved by proteins known as laminins. Cyclin-dependent kinase 1 (CDK1) increases nuclear envelope destabilization during GVBD by phosphorylating the laminins (Adhikari & Liu, Reference Adhikari and Liu2014). Simultaneously with the disintegration of the nuclear envelope, chromosome condensation occurs and the metaphase plate forms. The protein degradation that takes place during the transition from metaphase to anaphase is regulated by the anaphase promoter complex (APC), which is accountable for the ubiquitination of a number of different protein substrates (Landim-Alvarenga & Maziero, Reference Landim-Alvarenga and Maziero2018).
Following sperm entrance into an MII oocyte, chromosomes are segregated and the formation of the nuclear envelope brings an end to meiosis with the expulsion of the second polar bodies (PB) (Greaney et al., Reference Greaney, Wei and Homer2018). The embryonic development process begins with the union of female and male pro-nuclei after the extrusion of the second PB (Georgadaki et al., Reference Georgadaki, Khoury, Spandidos and Zoumpourlis2016).
Nuclear modifications during oocyte maturation and fertilization are required for appropriate embryonic development, which are synchronized with the movement of genetic material and organelles as well as metabolic alterations in the cytoplasm.
Meiotic arrest regulation in the GV
An essential factor in controlling the maturation of oocytes is cAMP (Ramos Leal et al., Reference Ramos Leal, Santos Monteiro, Souza-Fabjan, de Paula Vasconcelos, Garcia Nogueira, Reis Ferreira and Varella Serapião2018; Strączyńska et al., Reference Strączyńska, Papis, Morawiec, Czerwiński, Gajewski, Olejek and Bednarska-Czerwińska2022). Meiosis arrest is maintained by a high concentration of cAMP in the oocyte, whereas a reduction in cAMP levels permits meiosis to resume (Pan & Li, Reference Pan and Li2019). A balance between the enzymes adenyl cyclase (AC) and PDE, which are responsible for cAMP production and breakdown, respectively, controls the concentration of cAMP in the oocyte (Landim-Alvarenga & Maziero, Reference Landim-Alvarenga and Maziero2018). Oocyte cAMP synthesis is regulated by G protein-coupled receptors (GPR3), which are required for adenyl cyclase activation (Richards & Ascoli, Reference Richards and Ascoli2018).
Natriuretic peptide precursor C (NPPC) is present in the mural granulosa cells on the follicular wall, while natriuretic peptide receptor 2 (NPR2) is expressed by the cumulus cells surrounding the oocytes (Alam & Miyano, Reference Alam and Miyano2020). The NPPC in mural granulosa cells can bind to NPR2 receptors in cumulus cells to produce cyclic guanosine monophosphate (cGMP), which then enters oocytes through gap junctions to inhibit the activity of phosphodiesterase (PDE3A). This maintains a high level of cAMP in oocytes. Oocytes with high concentrations of cAMP inhibit maturation-promoting factor (MPF) activity through a protein kinase A (PKA)-dependent mechanism (Gilchrist et al., Reference Gilchrist, Luciano, Richani, Zeng, Wang, De Vos, Sugimura, Smitz, Richard and Thompson2016).
The MPF is a protein made up of a catalytic subunit, CDK1 and a regulatory subunit, cyclin B and is regulated by phosphorylation of CDK1 at its tyrosine 15 and threonine 14 sites. The phosphorylation is carried out by the kinase Wee1B, while the dephosphorylation is accomplished by the phosphatase Cdc25 (Landim-Alvarenga & Maziero, Reference Landim-Alvarenga and Maziero2018). Wee1B and Cdc25 activity is directly regulated by the PKA (Filatov et al., Reference Filatov, Khramova and Semenova2019) (Figure 1).
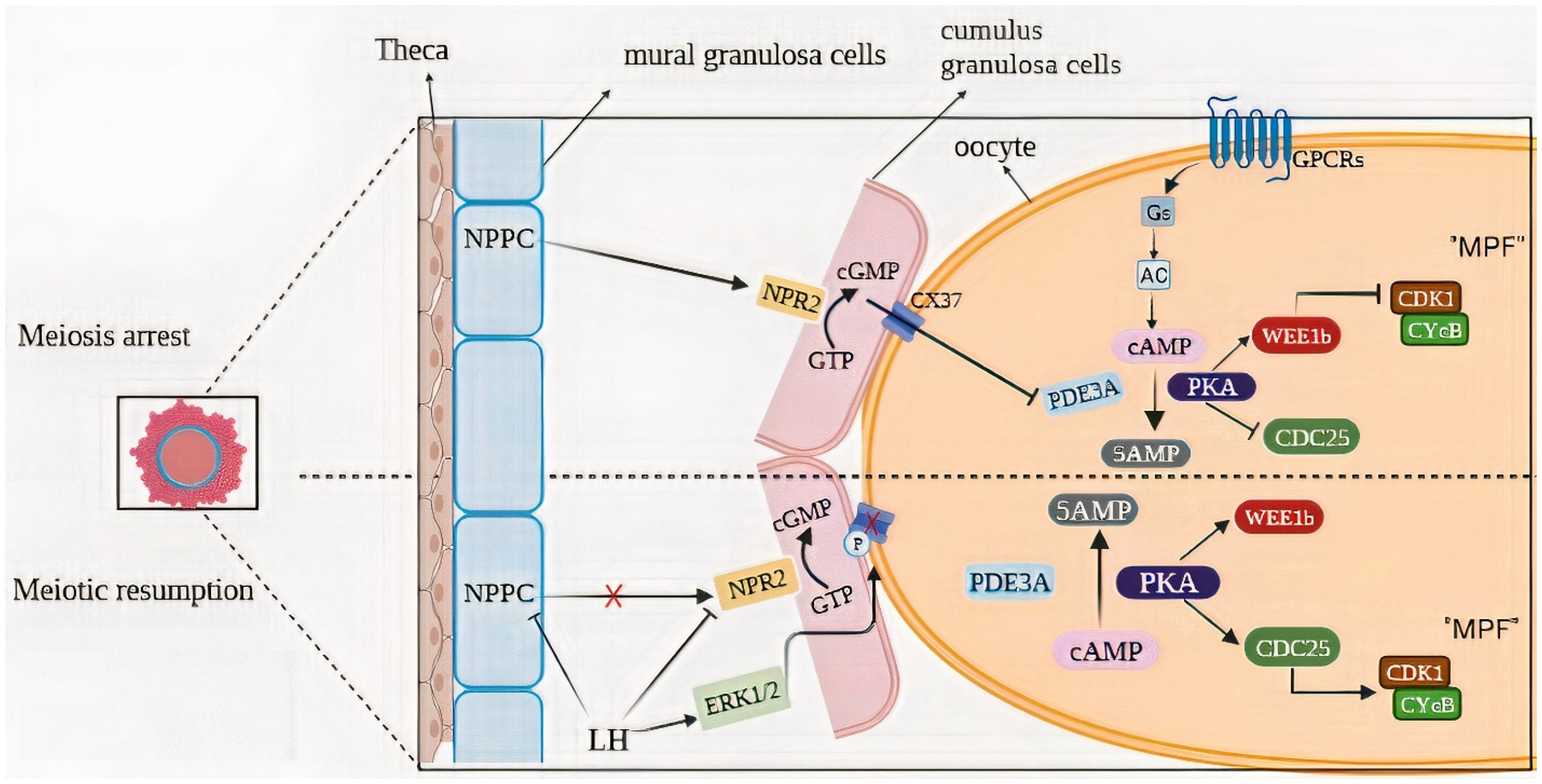
Figure 1. Mechanisms of meiotic prophase maintenance and gonadotropin-induced meiosis resumption. (A) In fully developed follicles, prophase I-arrested oocytes need the production and maintenance of high levels of cAMP, which are generated by activating the GPCR/Gs/AC pathway. Inhibition of phosphodiesterase 3A (PDE3A) prevents cAMP degradation and keeps cAMP levels high in the oocyte. Natriuretic peptide precursor type C (NPPC) is made by mural granulosa cells (GCs) and induces the production of cyclic guanosine monophosphate (cGMP) by NPR2 (a guanylyl cyclase) existing in cumulus GCs. In the oocyte, cGMP enters via Cx37 gap junctions and blocks cAMP hydrolysis by PDE3A. cAMP stimulates the activity of protein kinase A (PKA), which in turn stimulates the activity of the Wee1B kinase and inhibits the activity of the CDC25B phosphatase, resulting in the inactivation of CDK1. (B) A preovulatory level rise of LH induces gap junctions across the follicle to close, shutting off cGMP delivery to the oocyte. As a result, PDE3A’s ability to hydrolyze cAMP is subsequently enhanced. Low levels of cAMP and PKA can no longer activate WEE1B and inactivate CDC25B and CDK1 becomes dephosphorylated and catalytically active.
Meiosis resumption
Various exogenous factors are required for the resume of meiosis. LH can lead to a reduction in the expression of NPPC receptors in the granulosa cell (GC) and resulting in a lack of cGMP transfer to the oocyte (Arroyo et al., Reference Arroyo, Kim and Yeh2020). Concurrently, the LH surge leads to a disruption in the gap junctions that connect the oocyte and the GC. Epidermal growth factors such as the epiregulin, amphiregulin and beta-cells cause a breakdown in the oocyte-to-GC gap junction (Richani & Gilchrist, Reference Richani and Gilchrist2018). The decrease in cGMP causes an increase in PDE3 activity, which in turn leads to a precipitous fall in cAMP levels in the oocyte (Gershon et al., Reference Gershon, Maimon, Galiani, Elbaz, Karasenti and Dekel2019). As cAMP levels drop, PKA activity is dampened, and Cdc25 is nuclear translocated. The buildup of the phosphatase Cdc25 in the nucleus is what drives both the activation of the MPF and the delivery of Wee1B into the cytoplasm. In both the mitotic and meiotic cycles, MPF activation causes chromosomal condensation, nuclear envelope breakdown (GVBD) and cytoplasmic preparation for the M phase(Oh et al., Reference Oh, Han and Conti2010) (Figure 1).
Mitogen-activated protein Kinase (MAPKs)
Recent studies have shown that, in addition to MPF, the mitogen-activated protein kinase (MAPK) cascade is a major regulatory mechanism that drives the advancement of the meiotic cell cycle in oocytes (Sha et al., Reference Sha, Dai, Dang, Tang, Liu, Zhang and Fan2017). MAPK, which is also known as extracellular-regulated kinase (ERK), has two isoforms, ERK1 (p44) and ERK2 (p42), which are extensively expressed in the oocyte. Direct activation of MAPK occurs through phosphorylation of threonine-183 and tyrosine-185 in the activation loop by MAPK kinase (also known as MAPK-ERK kinase 1, MEK1) (Tabatabaie et al., Reference Tabatabaie, Amiri, Golestan Jahromi, Sene, Zandieh, Mehdizadeh and Amjadi2022). Phosphorylation is also required for MEK activation, and MOS, the product of the proto-oncogene c-mos, acts as an upstream activator of MEK in the oocyte. MOS was first discovered in cells transformed by the Moloney murine leukaemia virus (Cao et al., Reference Cao, Jiang and Fan2020; Papkoff et al., Reference Papkoff, Verma and Hunter1982). It is a 39-kDa Ser/Thr protein kinase that is expressed only in germ cells (Liang et al., Reference Liang, Su, Fan, Schatten and Sun2007). In CC, RAS/RAF trigger MAPK activation (Das & Arur, Reference Das and Arur2022). Through the suppression of several negative regulators and the activation of cdc25 phosphatase, the MAPK, when activated, enhances MPF stability in the oocyte. coincides with or precedes GVBD, MAPK is activated in the oocyte, and its levels rise steadily throughout oocyte maturation, staying high until meiosis II (Landim-Alvarenga & Maziero, Reference Landim-Alvarenga and Maziero2018).
Regulation of oocyte transcripts required for nuclear maturation
Oocytes actively take part in the meiotic maturation process, guaranteeing that they advance to the mature stage. During maturation, oocyte transcripts undergo a predominately large degradation; yet, this phenomenon seems to be a selective process (Su et al., Reference Su, Sugiura, Woo, Wigglesworth, Kamdar, Affourtit and Eppig2007). It is clear that transcripts and/or proteins linked to oocyte meiotic arrest at the GV stage are degraded or have significantly reduced expression levels. As an illustration, translation of CDH1, a co-activator of the anaphase-promoting complex/cyclosome (APC/C) that suppresses cyclin B1 levels by ubiquinylation and upholds meiotic arrest, is 70% lessened (Holt et al., Reference Holt, Tran, Stewart, Minahan, García-Higuera, Moreno and Jones2011).
However, transcripts implicated in signalling pathways like ERK/MAPK and PI3/AKT that are crucial for controlling oocyte meiosis and maintaining meiotic arrest during MII are still present (Su et al., Reference Su, Sugiura, Woo, Wigglesworth, Kamdar, Affourtit and Eppig2007). While it has been demonstrated that certain maternal-effect transcripts degrade during this transition, other transcripts seem to remain stable.
During oocyte maturation and/or fertilization, unusual transcript maintenance or degradation may be detrimental to oocyte quality and undermine developmental competence(Krajnik et al., Reference Krajnik, Mietkiewska, Skowronska, Kordowitzki and Skowronski2023).
However, the majority of transcripts in the oocyte are destroyed during maturation. Many additional transcripts undergo polyadenylation and connect with the polysomes to undergo translation (Chen et al., Reference Chen, Melton, Suh, Oh, Horner, Xie, Sette, Blelloch and Conti2011; Sha et al., Reference Sha, Zhang and Fan2019). Recent investigations have shown various patterns of oocyte transcripts in conjunction with the polysomes using wide-genome profiling of maternal mRNAs, showing active translational control throughout maturation. It has been proven via the use of this standpoint that during oocyte maturation, almost 7600 transcripts are actively translated while many others are repressed (Chen et al., Reference Chen, Melton, Suh, Oh, Horner, Xie, Sette, Blelloch and Conti2011).
Message ribonucleoprotein (mRNP) complexes allow for the storage of these transcripts so that they can be translated when required (Hafidh et al., Reference Hafidh, Čapková and Honys2011). Additionally, they might be spread throughout the whole oocyte’s cytoplasm or concentrated within a particular area of it. More research clarified the oocyte gene expression profile from various angles. Depending on their functions, the expression of a number of genes was associated with DNA, chromatin, RNA, transcription, kinases, membrane receptors, ion channels, mitochondria, structural nuclear proteins and phospholipases as well as to apoptosis, cell cycle, secretory pathways, exocytosis, endocytosis and other processes (Virant-Klun et al., Reference Virant-Klun, Knez, Tomazevic and Skutella2013). In addition to recognized genes like DAZL, BMP15, or GDF9, Assou et al. discovered 1,514 additional genes that were up-regulated in human oocytes. They included transcription factors like OTX2, SOX15 and SOX30, meiosis-related genes like PTTG3 (securing) and AURKC (Aurora kinase), as well as newly unreported growth factors including TNFSF13/APRIL, FGF9, FGF14 and IL4 (Assou et al., Reference Assou, Anahory, Pantesco, Le Carrour, Pellestor, Klein, Reyftmann, Dechaud, De Vos and Hamamah2006). In a similar investigation, Gasca et al. examined a collection of immature oocytes, mature MII oocytes that had not been fertilized and cumulus cells. The high gene expression levels of BRCA1 and 2, ATM, TP53, RB1, BUB1, MAD2, APC and ACTB were examined in all samples. All samples exhibited ACTB and MAD2 expression. Since MI and MII oocytes had significant levels of RBBP7, RBBP4 and RBL2 expression, it was possible that this transcription regulatory pathway may be active during oocyte maturation. Following oocyte maturation, the DNA repair marker BARD1 is expressed (Gasca et al., Reference Gasca, Pellestor, Assou, Loup, Anahory, Dechaud, De Vos and Hamamah2007). Additionally, the research by Wells and Patrizio revealed that several germ cell-specific genes, such as DAZL, and genes involved in meiosis, like SYCP2, SGOL2 and MSH2, were among those that were up-regulated in mature oocytes (Wells & Patrizio, Reference Wells and Patrizio2008).
It’s interesting to note that there seem to be two distinct methods of translational inhibition during oocyte maturation: one is transcript degradation, as was previously shown (Cui et al., Reference Cui, Li, Yin, Kang and Kim2006), and the other appears to be independent of degradation. In this latter case, translation appears to be repressed by the translocation of transcripts that remain stable, from the polysome to the subpolysome/RNP compartment (Chen et al., Reference Chen, Melton, Suh, Oh, Horner, Xie, Sette, Blelloch and Conti2011).
Well-known cell cycle regulators (Ccnb1 and Mos) have been found among the transcripts undergoing active translation during the GV-MII transition, along with elements of the anaphase-promoting complex and the spindle assembly checkpoint (Mad2, Bub1b and Sogl2). A group of transcripts that code for transcriptional regulators and chromatin remodellers are also members of this class (Sánchez & Smitz, Reference Sánchez and Smitz2012).
Recent research has shown two key regulators of translation during oocyte maturation: CPEB and DAZL. CPEB increases Dazl mRNA translation when to the MI stage advances; after that, the DAZL protein stimulates the translation of its own mRNA, creating a positive feedback loop. Although DAZL’s function has previously been suggested to have as a translational regulator during oocyte maturation (Cauffman et al., Reference Cauffman, Van de Velde, Liebaers and Van Steirteghem2005), Chen et al. (Chen et al., Reference Chen, Melton, Suh, Oh, Horner, Xie, Sette, Blelloch and Conti2011) have recently shown that DAZL has a clear and significant function during this late period.
Cytoplasmic maturation
Meiotic advancement, which is indicative of nuclear oocyte maturation, is not sufficient to ensure successful embryonic development; further alterations to the cytoplasm, or cytoplasmic maturation, are required (Trebichalská et al., Reference Trebichalská, Kyjovská, Kloudová, Otevřel, Hampl and Holubcová2021). Cytoplasmic maturation is comprised of both structural and molecular alterations that take place in the oocyte between the GV stage and the finish of the MII stage (Landim-Alvarenga & Maziero, Reference Landim-Alvarenga and Maziero2018). These changes include appropriate spatial and temporal dynamics of organelles that rearrange and store mRNA, proteins and transcription factors necessary for egg maturation, fertilization and early embryogenesis (Kang et al., Reference Kang, Wang and Yan2023). For the oocyte to attain high developmental potency, adequate alteration of the location, shape and biochemical characteristics of organelles must take place. During maturation, oocyte organelles such as the Golgi and mitochondria relocate from the oocyte’s periphery to a position in close proximity to the nucleus. The Golgi complex-derived cortical granules (CGs) travel from the centre of the oocyte to the periphery and attach to the membrane (Ferrer-Vaquer et al., Reference Ferrer-Vaquer, Barragán, Rodríguez and Vassena2019).
Rearrangement of the cytoskeleton
During oocyte development, dynamic redistribution of organelles takes place within an incredibly enormous volume (relative to somatic cells). It makes sense that the oocyte and early embryo may have developed a particular shape to aid with organelle location and distribution.
The cytoskeleton, because of its adaptability and dynamic nature, plays a crucial role in the rearrangement of oocyte organelles and may be altered to meet the oocyte’s specific requirements. Three different kinds of filaments make up this structure: microfilaments, intermediate filaments and microtubules. Microtubules in GV-stage oocytes are distributed in a rather even pattern. A similar distribution of microfilaments is also seen in the ooplasm. In the MII stage, mature oocyte microtubules and microfilaments mostly congregate in the cortical cytoplasm. Furthermore, microtubules, which may be seen as well-organized meiotic spindles, are often found in a direction perpendicular to the surface of the oocyte; they are barrel-shaped, anastral and centriole-free (Fan & Sun, Reference Fan, Sun, Leung and Adashi2019). Intermediate filaments are still not adequately comprehended in comparison to microfilaments and microtubules (Mao et al., Reference Mao, Lou, Lou, Wang and Jin2014) (Figure 2).

Figure 2. A summary of the distribution of cytoplasmic organelles and the cytoskeleton during oocyte maturation. In germinal vesicle (GV) oocytes, mitochondria are clustered around the GV; the Golgi apparatus is more concentrated in the interior than at the cortex; and the endoplasmic reticulum (ER), cortical granules, microtubules and microfilaments are evenly distributed throughout the cytoplasm. Oocytes undergo a number of morphological changes in stage germinal vesicle breakdown, including the relocation of mitochondria away from the perinuclear region, the fragmentation and aggregation of the Golgi apparatus in the central part of the oocyte, the localization of the ER in cortical regions, the condensation of microtubules around the chromosomes, and the dense accumulation of microfilaments in the subcortical region. At metaphase II, the first polar body has been extruded, spindles have formed underneath the first polar body, and intermediate filaments are uniformly dispersed throughout the cytoplasm. Also, the Golgi apparatus is further fractured and distributed throughout the oocyte; cortical granules travel towards the cortical cytoplasm and arrest in the cortex.
Also, new research shows that the cytoplasmic lattices of oocytes can play a key role in regulating the position and movement of the organelle mediated by cytoskeleton (microtubule) (Lin et al., Reference Lin, Zhu, Wang, Yang, Fan, Yi and Jiang2017). The cytoplasmic lattices (also known as cytoskeletal sheets, plaques, lamellae and fibrillar arrays) are missing in oocytes that are not developing but significantly increase in number as the oocyte grows and finally property prominently in the entirely grown oocyte (Qin et al., Reference Qin, Gao, Xiao, Zhang, Ma, Yu, Nie, Fan, Wang and Ouyang2019). Previous investigations into the molecular make-up of the lattices revealed that the lattices served as a reservoir for ribosomes or maybe intermediate filaments (Yurttas et al., Reference Yurttas, Vitale, Fitzhenry, Cohen-Gould, Wu, Gossen and Coonrod2008). More recently, studies using immunoelectron microscopy discovered that peptidylarginine deiminase 6 (PADI6), which is a very plentiful oocyte and embryo-restricted maternal protein, binds specifically to the lattices (X. Liu et al., Reference Liu, Morency, Li, Qin, Zhang, Zhang and Coonrod2017). In terms of expression, PADI6 protein is visible in the cytoplasm of oocytes as soon as they start to mature, and it is present in embryos until the blastocyst stage of development, which is resembling to that of the lattices (Innocenti et al., Reference Innocenti, Fiorentino, Cimadomo, Soscia, Garagna, Rienzi, Ubaldi and Zuccotti2022). Along with PADI6, two other maternal factors, MATER and FLOPED, have been identified to localizing to and being necessary for lattice formation (Bebbere et al., Reference Bebbere, Masala, Albertini and Ledda2016; Lu et al., Reference Lu, Gao, Qin and Li2017). Both MATER and FLOPED exhibit PADI6-like expression patterns in oocytes and early embryos (Paonessa et al., Reference Paonessa, Borini and Coticchio2021). These results suggest that the distribution of organelles via microtubules during oocyte maturation may be directly influenced by the lattices.
Endoplasmic reticulum and Mitochondria
The endoplasmic reticulum (ER) of the human oocyte serves as a reserve for the Ca2+ required for oocyte activation (Wakai et al., Reference Wakai, Mehregan and Fissore2019). Upon fertilization, the entry of sperm into the oocyte initiates a series of intricate intracellular signalling cascades, ultimately leading to oocyte activation, a pivotal process essential for the commencement of embryonic development. Specifically, the entry of sperm into the oocyte triggers the activation of phospholipase C zeta (PLCζ), an enzyme predominantly present in the sperm head (Saunders et al., Reference Saunders, Larman, Parrington, Cox, Royse, Blayney, Swann and Lai2002). PLCζ assumes a pivotal role in inducing Ca2+ oscillations within the oocyte by catalyzing the hydrolysis of phosphatidylinositol 4,5-bisphosphate (PIP2) into inositol trisphosphate (IP3) and diacylglycerol (Nomikos et al., Reference Nomikos, Kashir, Swann and Lai2013). IP3, acting as a second messenger, binds to IP3 receptors located on the ER membrane, leading to the opening of calcium channels and subsequent release of calcium ions from the ER lumen into the cytoplasm. This abrupt rise in cytoplasmic calcium levels initiates a cascade of essential downstream events crucial for oocyte activation, such as CG exocytosis, resumption of meiosis and initiation of embryo development (Stein et al., Reference Stein, Savy, Williams and Williams2020).
In contrast to oocytes in the GV stage, which exhibit a thin network of ER, mature human oocytes have ER arranged in discrete clusters (2–5 m) throughout the cell (Kang et al., Reference Kang, Wang and Yan2023; Palmerini et al., Reference Palmerini, Antonouli, Macchiarelli, Cecconi, Bianchi, Khalili and Nottola2022). Several researches showed time solidarity between the emission of Ca2+ and development of ER clusters at the cortex of mature MII-stage oocytes (FitzHarris et al., Reference FitzHarris, Marangos and Carroll2003; Kline et al., Reference Kline, Mehlmann, Fox and Terasaki1999). The redistribution of ER throughout maturation appears to be controlled by microtubules and microfilaments, similar to other organelles (mitochondria) (Kang et al., Reference Kang, Wang and Yan2023). Another restructuring characteristic connected to oocyte competence is the morphology and changing position of the mitochondria surrounding the spindle. These organelles establish a border that encloses the developing spindle and compressing chromosomes (Dalton & Carroll, Reference Dalton and Carroll2013). According to Van Blerkom, this redistribution is assumed to be essential to build high adenosine triphosphate (ATP) concentrations in areas with a high need for energy (Van Blerkom, Reference Van Blerkom2011).
Findings from electron microscopy and confocal studies of oocytes suggest that throughout maturation, mitochondria seem to undergo an orderly clustering and also move in close proximity to the ER (Takahashi et al., Reference Takahashi, Hashimoto, Yamochi, Goto, Yamanaka, Amo, Matsumoto, Inoue, Ito, Nakaoka, Suzuki and Morimoto2016). Numerous investigations have indicated that in somatic cells, near physical contacts between ER and mitochondria promote organelle interaction via concerted signalling processes. For instance, it was observed that mitochondrial-derived ATP activated ER membrane Ca2+ pumps, which then sent Ca2+ signals to the mitochondria and changed their metabolic rate. Furthermore, by controlling local Ca2+ concentrations and the rate of Ca2+ signalling events, mitochondria can function as a Ca2+ buffer (Silva et al., Reference Silva, DiGiovanni, Kumar, Carmichael, Kim and Schrader2020). Based on studies, it is probable that the ER and mitochondria in oocytes interact functionally, and that this interaction affects both ATP production and Ca2+ regulation (Wang et al., Reference Wang, Li, Meng, Wang, Wang, Hou, Schatten, Sun and Ou2020). Furthermore, the efficient interaction between these two organelles in oocytes may turn out to be a crucial factor in effective embryonic development (L. Liu et al., Reference Liu, Hammar, Smith, Inoue and Keefe2001) (Figure 2).
Golgi complex and cortical granules
The GV oocytes each have many Golgi complexes that are dispersed throughout the cytoplasm and are surrounded by numerous tiny vesicles. There are two types of vesicles that may be seen: smooth vesicles with uneven size and electron density are visible close to the trans face of the Golgi complex, whereas coated vesicles that resemble pinocytosis granules are detected next to the cis face (Landim-Alvarenga & Maziero, Reference Landim-Alvarenga and Maziero2018). During the course of oocyte maturation, the number and size of Golgi complexes present in the oocyte’s cytoplasm both decrease and become fragmented. The fragmented Golgi apparatus is dispersed throughout the oocyte in MI, and this pattern is also preserved in MII oocytes (Satouh & Sato, Reference Satouh and Sato2023).
The Golgi complex is the place where the CG were first derived (Trebichalská et al., Reference Trebichalská, Kyjovská, Kloudová, Otevřel, Hampl and Holubcová2021). At the ultrastructural level, the CGs all seem to have the same size (0.2 µm to 0.6 µm) and have a similar morphology (M. Liu, Reference Liu2011). Cortical granules are scattered in the cytoplasm of GV oocytes. At the end of the maturation process, in MII oocytes, the granules are located on the inner surface of the cell membrane. The arrangement described herein represents a pre-fertilization distribution mechanism enacted prior to sperm entry and subsequent oocyte activation (Reader et al., Reference Reader, Stanton and Juengel2017). After the fusion of sperm and oocyte, a significant surge in intracellular calcium levels ensues (as noted in section 2.2 Endoplasmic reticulum). Subsequent to this rise in calcium concentration, protein kinases such as protein kinase C (PKC) and Ca2+/calmodulin-dependent protein kinase II (CaMKII) become activated, phosphorylating various specific proteins associated with CGs within the oocyte (Tsaadon et al., Reference Tsaadon, Kaplan-Kraicer and Shalgi2008). These proteins often play pivotal roles in facilitating the activity of the exocytosis machinery or in regulating the fusion of CGs with the oocyte’s plasma membrane. Notable examples of such proteins include Soluble N-ethylmaleimide-sensitive factor Attachment Protein REceptors and Rab GTPases (M. Liu, Reference Liu2011). Within CGs, oocytes house a variety of hydrolytic enzymes, such as proteases, peroxidases and glycosaminoglycans (Georgadaki et al., Reference Georgadaki, Khoury, Spandidos and Zoumpourlis2016).
Oocytes employ specific strategies to prevent polyspermy. These strategies can generally be categorized into two types: the ‘oocyte membrane block’ to sperm penetration and the ‘zona reaction’ (Mio et al., Reference Mio, Iwata, Yumoto, Kai, Sargant, Mizoguchi, Ueda, Tsuchie, Imajo and Iba2012).
The oocyte membrane block begins with depolarization triggered by an influx of Na+ ions, altering the membrane potential from negative to positive. This depolarization then activates voltage-gated calcium channels on the oocyte membrane, allowing Ca2+ to enter from the extracellular space into the oocyte cytoplasm. The increase in intracellular calcium levels that follows serves as a pivotal signalling event for the rapid block to polyspermy. This rapid change in potential effectively inhibits sperm penetration and attachment to the oocyte membrane, providing a transient barrier against polyspermy. Referred to as the ‘fast block’ to polyspermy, this mechanism occurs within seconds. However, this ‘fast block’ mechanism is unlikely to play a significant role in preventing polyspermy during the fertilization process in humans (Carvacho et al., Reference Carvacho, Piesche, Maier and Machaca2018; Mio et al., Reference Mio, Iwata, Yumoto, Kai, Sargant, Mizoguchi, Ueda, Tsuchie, Imajo and Iba2012).
The second proposed mechanism is initiated by the attachment of sperm to the oocyte membrane, triggering a Ca2+ oscillation event. This occurrence results in an increase in intracellular Ca2+ concentration, which in turn triggers the release of CGs from beneath the oocyte membrane into the perivitelline space through exocytosis (Lin et al., Reference Lin, Zhu, Wang, Yang, Fan, Yi and Jiang2017) (Figure 2). Following the exocytosis of CGs, a ‘slow block’ to polyspermy occurs within the zona pellucida, the extracellular envelope surrounding the oocyte (Mio et al., Reference Mio, Iwata, Yumoto, Kai, Sargant, Mizoguchi, Ueda, Tsuchie, Imajo and Iba2012). The contents of these cortical granules, including proteinases such as tissue-type plasminogen activator (tPA), peroxidase and N-acetylglucosaminidase, play critical roles in this process. The proteinases modify the zona pellucida, making it less receptive to sperm binding by catalyzing the proteolysis of ZP2 and causing ZP hardening (Sun, Reference Sun2003). Similarly, peroxidase, an oocyte-specific protein stored within cortical granules, contributes to the polyspermy block by participating in the formation of the fertilization envelope post-cortical reaction (M. Liu, Reference Liu2011). Specifically, N-acetylglucosaminidase released during fertilization inactivates the sperm GalTase-binding site on ZP, effectively blocking subsequent sperm binding (Zitta et al., Reference Zitta, Wertheimer and Miranda2006). These enzymatic actions collectively modify the structure of sperm receptors like ZP2 and ZP3, creating a barrier that prevents additional sperm penetration and thus ensures monospermy. This ‘slow block’ typically occurs within 5 to 8 minutes after oocyte activation and is considered the primary mechanism for preventing polyspermy in humans (Mio et al., Reference Mio, Iwata, Yumoto, Kai, Sargant, Mizoguchi, Ueda, Tsuchie, Imajo and Iba2012).
In exploring the contentious issue of polyspermy prevention, Professor Brian Dale and colleagues have critically examined whether mechanisms actively repel supernumerary sperm from oocytes, or if the natural sperm-to-oocyte ratios inherently minimize the need for specific polyspermy prevention mechanisms. They suggest that monospermia is likely maintained through a combination of controlled sperm-oocyte encounters and specialized molecular and structural features within the oocyte (Dale & DeFelice, Reference Dale and DeFelice2011).
Professor Dale argues that the fusion of cortical granules with the plasma membrane represents a slow structural change, transforming the oocyte’s receptive external investment into a hardened protective layer for the developing embryo, rather than functioning solely to repel sperm. Additionally, he asserts that polyspermy is observed in aged oocytes both in vivo and in vitro. He contests the prevailing notion of ‘polyspermy-blocking mechanisms’ in mammalian oocytes, primarily based on in vitro experiments using oocytes stripped of their external coats, which may not accurately reflect natural fertilization processes (Dale & DeFelice, Reference Dale and DeFelice2011).
It’s important to note that Figure 2 has been adapted from a previously published study conducted by Trebichalská et al (Trebichalská et al., Reference Trebichalská, Kyjovská, Kloudová, Otevřel, Hampl and Holubcová2021). Slight modifications were made to the original figure to better align it with the focus of the current research.
Conclusion
The available information reviewed above demonstrates unequivocally that oocyte maturation is a complicated process that involves many ‘arrest and resume’ processes and is strictly controlled during the reproductive cycle. The basic paradigm of meiotic maturation in the oocyte is the precise management of intracellular cAMP’s spatial and temporal regulation to control the activation of the MPF. This process is accomplished by significant modifications in the level of expression of transcripts associated with the arrest of meiosis in the oocyte or the stabilization of transcripts that are crucial for oocyte maturation. Although oocyte nuclear and cytoplasmic maturation occur simultaneously, they are nevertheless autonomous phenomena. Cytoplasmic maturation takes place with the reorganization of organelles, most notably the active mitochondria and ER in the oocyte cytoplasm. Cytoplasmic maturation is crucial for oocytes to achieve fertilizable and post-fertilization developmental competence during maturation. Many of the processes described here are intracellular, while others are subject to paracrine regulation that is modulated by the close relationships between follicular cells and the oocyte. All of the activities that take place are essential to producing a functioning gamete that has the potential to develop into a healthy embryo after fertilization.
Acknowledgements
We would like to thank the authors of the primary studies reviewed in this article for their contributions to the field.
Author contributions
H.T.; T.A.; Sh.P.; L.T.; participated in the search and collection of articles, interpretation and manuscript writing. R.Sh.; R.M. contributed extensively to the interoperation of the data collection, study concept and design and manuscript writing. I.A.; M.M.: Manuscript writing and final approval of the manuscript. All authors read and approved the final manuscript.
Funding
There is no financial support in this study.
Competing interests
The authors declare no conflicts of interest.





