Introduction
Polar bears Ursus maritimus are distributed throughout the ice-covered marine areas of the circumpolar Arctic, especially over the shallower and more biologically productive continental shelves and inter-island channels. Nineteen subpopulations are currently recognized across the Arctic region and each is managed independently (Aars et al., Reference Aars, Lunn and Derocher2006).
The polar bear is the most iconic symbol of the Arctic and thus any perception of a threat has generally been followed by significant national and international response. For example, in 1973, following years of concern that polar bears were being overexploited, Canada, USA, USSR, Norway and Denmark (for Greenland) signed the Agreement on the Conservation of Polar Bears (Prestrud & Stirling, Reference Prestrud and Stirling1994; Aars et al., Reference Aars, Lunn and Derocher2006). Among other things, the Agreement stipulates that each of the five signatory nations ‘shall take appropriate action to protect the ecosystems of which polar bears are a part’.
In Canada threatened species are identified by the Committee on the Status of Endangered Wildlife in Canada (COSEWIC) and species are reassessed at least every 10 years. To date, polar bears have been considered a single biological unit with a shared conservation status (Stirling & Taylor, Reference Stirling and Taylor1999; COSEWIC, 2002). However, they occupy habitat that extends > 5,000 km from east to west and 3,000 km north to south (Fig. 1a) and it is now clear that the rate and consequences of climate-related sea ice declines vary in different parts of the Canadian Arctic (Stirling et al., Reference Stirling, Lunn and Iacozza1999; Derocher et al., Reference Derocher, Lunn and Stirling2004; Stirling & Parkinson, Reference Stirling and Parkinson2006; Amstrup et al., Reference Amstrup, Marcot and Douglas2007). Consequently, it may now be more appropriate to consider the status of polar bear populations in different regions independently.
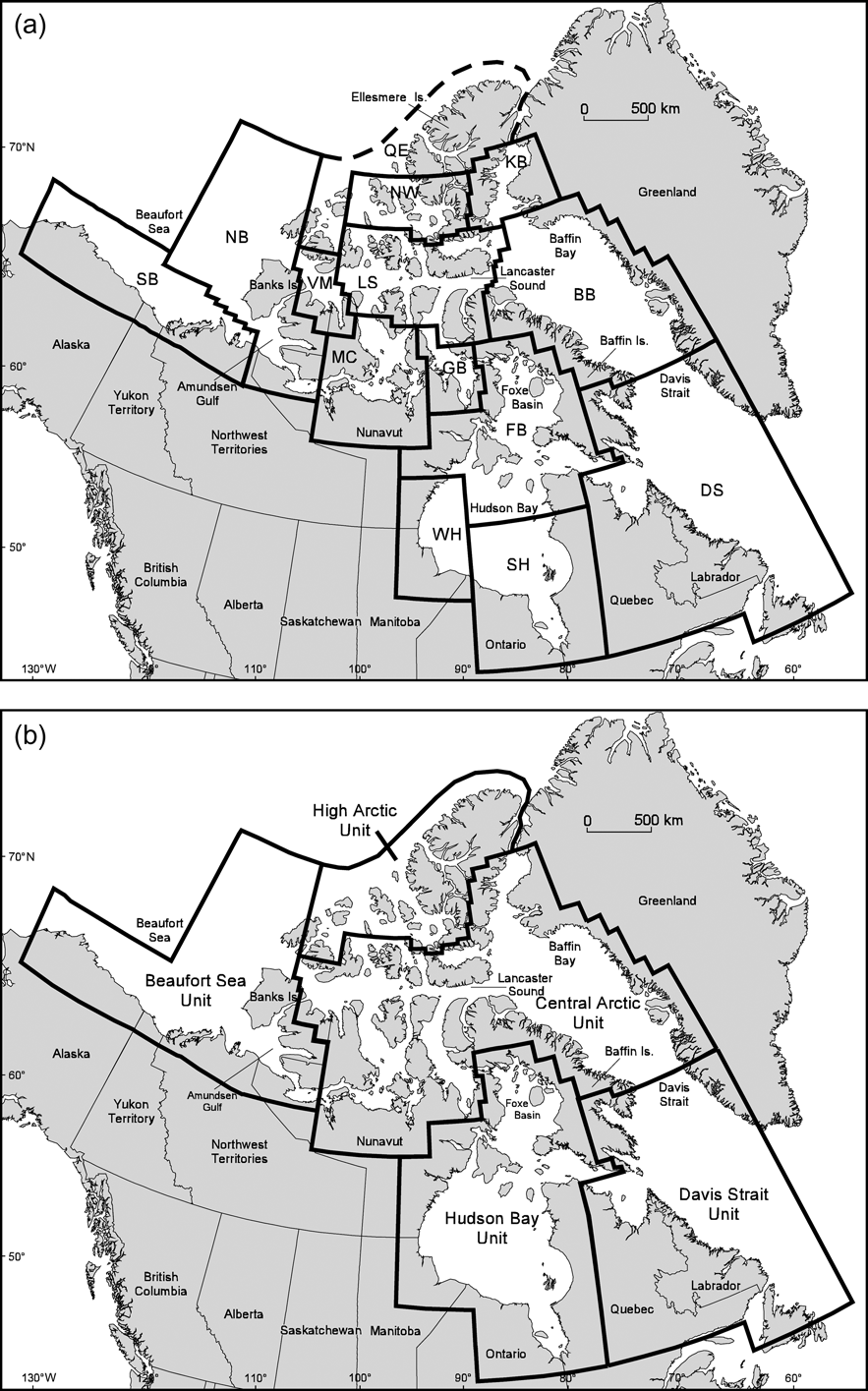
Fig. 1 (a) Current Canadian polar bear management zones as defined by the IUCN/SSC Polar Bear Specialist Group (Aars et al., Reference Aars, Lunn and Derocher2006). NB, Northern Beaufort Sea; SB, Southern Beaufort Sea; VM, Viscount Melville; LS, Lancaster Sound; MC, M'Clintock Channel; GB, Gulf of Boothia; NW, Norwegian Bay; KB, Kane Basin; BB, Baffin Bay; FB, Foxe Basin; WH, Western Hudson Bay; SH, Southern Hudson Bay; DS, Davis Strait. The Queen Elizabeth (QE) population was previously used as a geographic catch-all to account for the northern tip of Ellesmere Island (IUCN/SSC Polar Bear Specialist Group, Reference Lunn, Schliebe and Born2002) and is no longer recognized as a distinct subpopulation. (b) Proposed designatable units (DUs) for polar bear conservation based on genetic, ecological and life history data (see text for details).
In 2002 the Government of Canada passed the Species at Risk Act, the wording of which allows ‘distinct populations of wildlife’ within a species to be afforded legal protection. Green (Reference Green2005) described a systematic procedure to identify intraspecific units of conservation (designatable units, DUs) based on the hierarchical consideration of taxonomy, genetic distinction, range disjunction and biogeographical distribution. This approach was adopted by COSEWIC (2005) and intraspecific units can be given separate legal listings to protect diversity within a species. The DU approach has been applied to a variety of species including the Boreal felt lichen Erioderma pedicellatum, Blanding's turtle Emydoidea blandingii, coho salmon Oncorhynchus kisutch and woodland caribou Rangifer tarandus caribou (COSEWIC, 2005). Here we apply the DU framework to polar bears to examine the possibility that large-scale ecological processes have yielded groups of polar bears with regionally-specific conservation needs.
To examine the ecological factors relevant to polar bear conservation we incorporated four types of intrinsic data (genetic, spatial, demographic and life history), and two categories of extrinsic data (abiotic, including barriers established by land, multi-year ice and open water, and biotic, including the distribution and abundance of prey and overall biological productivity). We examined possible correlations between these physical and ecological properties and the conservation status of polar bears.
Background
Sea ice is the polar bear's primary habitat. It provides a platform upon which polar bears move, hunt, mate and sometimes den. In portions of their range where the sea ice melts completely, polar bears are forced to fast on land without access to the ringed seals Phoca hispida and bearded seals Erignathus barbatus that comprise most of their diet (Stirling et al., Reference Stirling, Jonkel, Smith, Robertson and Cross1977; Iverson et al., Reference Iverson, Stirling, Lang, Boyd, Wanless and Camphuysen2006). In some areas long-term climate warming has caused ice breakup to occur earlier and periods of open water to be extended (Stirling et al., Reference Stirling, Lunn and Iacozza1999; Stirling & Parkinson, Reference Stirling and Parkinson2006; Regehr et al., Reference Regehr, Lunn, Amstrup and Stirling2007b). Consequently, bears have less time to accumulate body fat and may be forced to fast for longer periods. In areas where polar bears remain on the ice year-round, increased melting has resulted in decreased time on the biologically productive continental shelf (Regehr et al., Reference Regehr, Hunter, Caswell, Amstrup and Stirling2007a). Climate-related habitat loss has resulted in declines in body condition, survival, reproduction and total abundance in some populations (Stirling et al., Reference Stirling, Lunn and Iacozza1999; Hunter et al., Reference Hunter, Caswell, Runge, Regehr, Amstrup and Stirling2007; Regehr et al., Reference Regehr, Hunter, Caswell, Amstrup and Stirling2007a,Reference Regehr, Lunn, Amstrup and Stirlingb).
The polar bear is categorized as Vulnerable on the IUCN Red List because of projected declines in polar bear abundance and habitat quality (Aars et al., Reference Aars, Lunn and Derocher2006; IUCN, 2007). Largely in response to evidence that climate warming has already affected the abundance and reproductive success of polar bears, the US Department of the Interior has listed the polar bear as threatened throughout its range under the Endangered Species Act (USFWS, 2008). This listing considers polar bears as a single population with a uniform conservation status. However, research used to inform the listing decision concluded that the long-term status of polar bears will probably vary across different polar ice ecoregions (Amstrup et al., Reference Amstrup, Marcot and Douglas2007). Nevertheless, the utility of distinct population segments or DUs for the legal protection of polar bears remains unexplored.
In Canada hunting of polar bears is important to both the culture and economy of indigenous peoples, primarily Inuit (Freeman & Wenzel, Reference Freeman and Wenzel2006). Although the overall management goal is to maximize the kill within sustainable limits, the success of this approach is contingent upon good scientific data and a relatively stable, predictable environment. Thus, the extensive loss of sea ice to date (Comiso, Reference Comiso2002; Stroeve et al., Reference Stroeve, Holland, Meier, Scambos and Serreze2007), and forecast future loss (Meehl et al., Reference Meehl, Stocker, Collins, Friedlingstein, Gaye, Gregory, Solomon, Qin, Manning, Chen, Marquis and Averyt2007), presents a new and serious threat to the survival of polar bears. Although adverse effects of harvesting may, in principle, be mitigated by hunting restrictions, the interaction of climate change and harvest make it more difficult to ensure a sustainable harvest.
Section 2(1) of the Species at Risk Act allows a ‘wildlife species’ to be defined as any ‘geographically or genetically distinct population’. Although the Act does not provide an explicit definition for a ‘distinct population’, COSEWIC has adopted a methodology for identifying DUs for protection under the Act (COSEWIC, 2005; Green, Reference Green2005). The first step in this method is to examine the species as a whole and determine whether a single designation would accurately reflect the conservation status of the entire species. If not, the second step is to identify potential DUs by sequentially examining: (1) named subspecies or varieties, (2) genetically distinct units, (3) geographically distinct units, and (4) biogeographically distinct units. In the third step DUs are reassessed to ensure they accurately reflect the conservation status of the individual groups.
Designatable units for polar bears
In different areas polar bears belong to ecosystems that differ fundamentally in their structure and functioning. In addition, the ecological effects of climate change vary substantially throughout the Canadian Arctic. Thus, although all polar bears in Canada belong to a single species they do not share a single, uniform conservation status. The identification and assessment of possible DUs is therefore warranted. For polar bears, no named subspecies or varieties have been recognized (Wilson, Reference Wilson, Pelton, Lentfer and Folk1976).
Genetically distinct units
Current management subpopulations (Fig. 1a) reflect polar bears’ fidelity to denning, summer refugia, and foraging areas (Jonkel, Reference Jonkel1969; Schweinsburg & Lee, Reference Schweinsburg and Lee1982; Lentfer, Reference Lentfer1983; Stirling et al., Reference Stirling, Calvert, Andriashek, Olson, Hastings and Geddes1984; Derocher & Stirling, Reference Derocher and Stirling1990; Mauritzen et al., Reference Mauritzen, Derocher and Wiig2001; Taylor et al., Reference Taylor, Akeeagok, Andriashek, Barbour, Born and Calvert2001), and long-distance migration between populations is limited (Stirling et al., Reference Stirling, Jonkel, Smith, Robertson and Cross1977; Lentfer, Reference Lentfer1983; Taylor & Lee, Reference Taylor and Lee1995). The conditions therefore exist for groups of polar bears to have developed distinct genetic adaptations to local environmental conditions.
Paetkau et al. (Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999) examined 16 microsatellite loci in polar bears from 12 of the 13 Canadian subpopulations and identified four major genetic clusters (Fig. 2). Relative to the variability between all groups, they found a high degree of similarity between the Northern and Southern Beaufort Sea subpopulations, and between the Western Hudson Bay and Foxe Basin subpopulations. Although genetic data from Southern Hudson Bay were unavailable, movement and foraging data (Stirling et al., Reference Stirling, Lunn and Iacozza1999; Thiemann, Reference Thiemann2006) suggest that Western and Southern Hudson Bay are similar. Bears from Baffin Bay and Kane Basin are genetically homogeneous and formed a larger cluster with Lancaster Sound, Gulf of Boothia, M'Clintock Channel and Viscount Melville Sound. Although Paetkau et al. (Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999) grouped Davis Strait with Foxe Basin and Western Hudson Bay subpopulations, their data indicated that Davis Strait bears are genetically intermediate between Foxe Basin and the Central Arctic groups (Fig. 2). Bears from the Norwegian Bay subpopulation are genetically distinct. The genetic relationships between subpopulations were consistent with movement patterns, i.e. the occasional movement of individuals between subpopulations appeared to be responsible for genetic similarities.
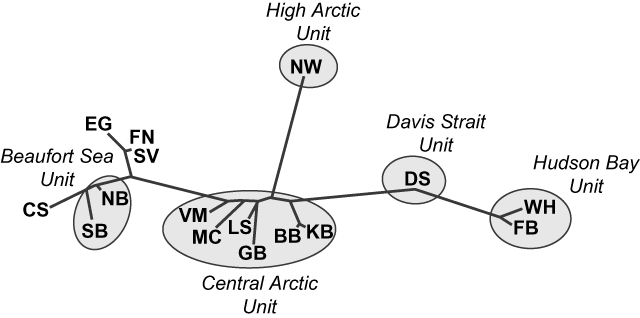
Fig. 2 Graphical representation of genetic distances between polar bear subpopulations (for abbreviations, see Fig. 1a; Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999), and the five designatable units (see text for details). Clustering is based on the genotype likelihood ratio distance and represents relative patterns of similarity among subpopulations. Overall, the genetic variability among polar bears is less than that observed in intraspecific groups of black bears Ursus americanus or brown bears Ursus arctos but patterns still reflect ecological relationships among subpopulations. The international Chukchi Sea (CS), East Greenland (EG), Franz Josef Land-Novaya Zemlya (FN), and Svalbard (SV) subpopulations are not discussed in this paper. Modified from Paetkau et al. (Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999).
Although Paetkau et al. (Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999) concluded the observed genetic differences are relatively small compared to other bear species and do not indicate multiple evolutionary significant units, they noted that differences in important adaptive traits may exist between groups of polar bears. Because microsatellite sequences are neutral genetic markers they reflect the amount of time that groups have been separated, rather than the amount of evolutionary adaptation that has occurred. Other, phenotypic, differences may reflect recent, adaptive, genetic diversity.
These results (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999) suggest that polar bears in Canada comprise five distinct genetic units that could be considered as DUs: Beaufort Sea, Central Arctic, High Arctic, Hudson Bay/Foxe Basin and Davis Strait (Fig 1b). Although microsatellite data indicate some genetic similarity between members of Hudson Bay/Foxe Basin and Davis Strait we consider these as separate DUs because of the distinct ecological conditions in the two regions (see below). To account for the northern tip of Ellesmere Island we have combined the previously recognized Queen Elizabeth subpopulation (IUCN/SSC Polar Bear Specialist Group, Reference Lunn, Schliebe and Born2002) with the Norwegian Bay population to form the High Arctic DU.
Biogeographically distinct units
Here, we incorporate larger-scale ecogeographical data and examine whether significant genetic differences are correlated with biologically significant phenotypic differences. The areas covered by the five possible DUs include substantial ecological and geographical variability (Table 1).
Table 1 Selected environmental, ecological and demographic characteristics of proposed designatable units (DUs; Fig. 1b) and subpopulations (Fig. 1a) of polar bears in Canada.
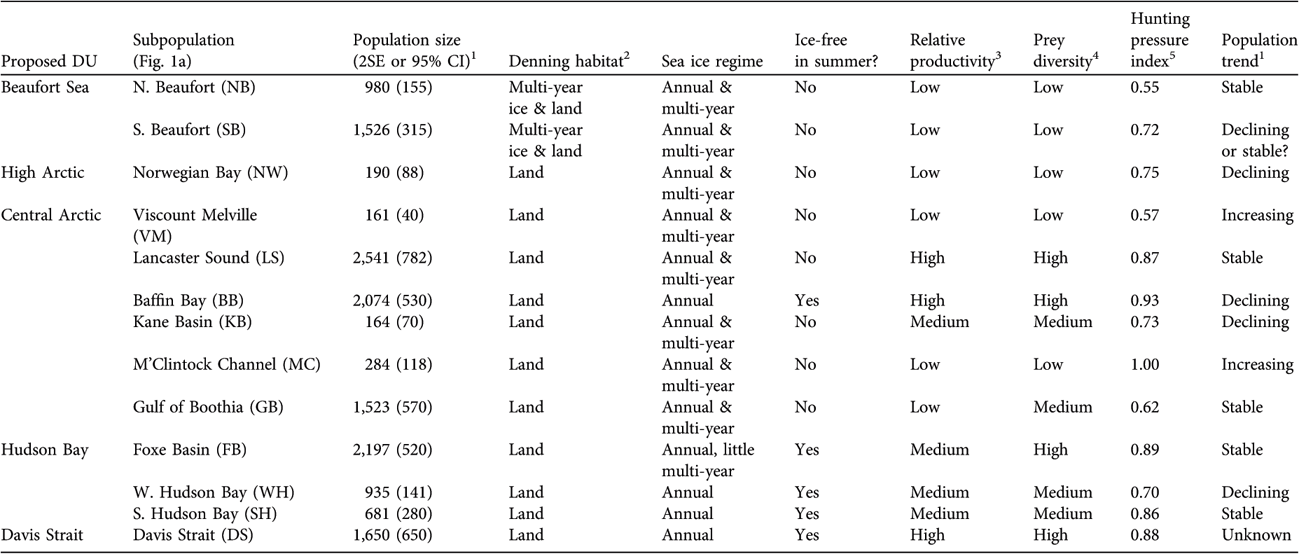
1 From IUCN/SSC Polar Bear Specialist Group (Reference Aars, Lunn and Derocher2006), Regehr et al. (Reference Regehr, Amstrup and Stirling2006), Obbard et al. (Reference Obbard, McDonald, Howe, Regehr and Richardson2007), Stirling et al. (Reference Stirling, McDonald, Richardson and Regehr2007)
2 From Stirling et al. (Reference Stirling, Calvert and Andriashek1980, Reference Stirling, Calvert, Andriashek, Olson, Hastings and Geddes1984), Schweinsburg et al. (Reference Schweinsburg, Spencer and Williams1984), Amstrup & Gardner (Reference Amstrup and Gardner1994), Messier et al. (Reference Messier, Taylor and Ramsay1994), Van de Velde et al. (Reference Van de Velde, Stirling and Richardson2003), Fischbach et al. (Reference Fischbach, Amstrup and Douglas2007)
3 From Subba Rao & Platt (Reference Subba Rao and Platt1984), Stirling & Øritsland (Reference Stirling and Øritsland1995)
4 Low, 2–3 species; Medium, 4–5 species; High, 6–8 species
5 Historical annual removals/potential maximum annual removals (provides a measure of relative harvest intensity); from IUCN/SSC Polar Bear Specialist Group (Reference Aars, Lunn and Derocher2006)
Geography The polar bears of the Hudson Bay DU (Western Hudson Bay, Southern Hudson Bay and Foxe Basin) comprise a distinct genetic, ecological and geographical group. Although bears in these three subpopulations are separated during the ice-free season, their foraging and breeding ranges may overlap substantially (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999; Stirling et al., Reference Stirling, Lunn and Iacozza1999; A.E. Derocher, unpubl. data). A small number of polar bears move through Hudson Strait (Stirling & Kiliaan, Reference Stirling and Kiliaan1980; Taylor et al., Reference Taylor, Akeeagok, Andriashek, Barbour, Born and Calvert2001), resulting in some genetic similarity between the Hudson Bay and Davis Strait DUs (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999). However, the large land masses of eastern mainland Canada and Baffin Island limit movement between Hudson Bay and the Atlantic Ocean. Similarly, Fury and Hecla Strait forms a narrow oceanographic connection between Foxe Basin and Gulf of Boothia, with little genetic exchange (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999).
Banks and Victoria Islands serve as land barriers between the Beaufort Sea DU and the polar bears of the Central Arctic. The rugged topography and ice-fields of Ellesmere and Devon Islands, and thick multi-year sea ice between the archipelago islands where seal densities are lower (Kingsley et al., Reference Kingsley, Stirling and Calvert1985), effectively separate the High Arctic and Central Arctic DUs. Within the Central Arctic DU the islands of the archipelago separate six subpopulations (Bethke et al., Reference Bethke, Taylor, Amstrup and Messier1996; Taylor et al., Reference Taylor, Akeeagok, Andriashek, Barbour, Born and Calvert2001). However, these separations appear to be relatively weak, and genetic (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999), ecological (Thiemann et al., in press), and conservation similarities (Stirling & Parkinson, Reference Stirling and Parkinson2006) between these subpopulations support their consideration as a single DU.
Ice conditions The abundance and availability of sea ice habitat is the most important environmental factor affecting the distribution and movement of polar bears (Schweinsburg & Lee, Reference Schweinsburg and Lee1982; Schweinsburg et al., Reference Schweinsburg, Lee and Latour1982; Stirling et al., Reference Stirling, Andriashek and Calvert1993, Reference Stirling, Lunn and Iacozza1999, Reference Stirling, Lunn, Iacozza, Elliott and Obbard2004; Ferguson et al., Reference Ferguson, Taylor, Born and Messier1998, Reference Ferguson, Taylor, Born, Rosing-Asvid and Messier2001; Mauritzen et al., Reference Mauritzen, Derocher and Wiig2001; Parks et al., Reference Parks, Derocher and Lunn2006). In addition to serving as a substrate for polar bears to travel, hunt, mate and den, ringed and bearded seals depend on the sea ice for reproduction and as a place to rest and moult. Consequently, the local abundance of polar bears is directly related to the availability of suitable sea ice habitat.
Optimal polar bear habitat is generally a mix of stable land-fast ice and dynamic offshore pack ice (Stirling et al., Reference Stirling, Andriashek and Calvert1993). Seal abundance is reduced in areas of multi-year ice (Kingsley et al., Reference Kingsley, Stirling and Calvert1985) and because polar bears avoid areas where hunting is poor, multi-year ice can form a significant barrier to movement (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999; Taylor et al., Reference Taylor, Akeeagok, Andriashek, Barbour, Born and Calvert2001). The fast ice that forms over the biologically productive continental shelf is prime ringed seal habitat. This annual ice is thin enough for ringed seals to maintain breathing holes yet stable enough for the construction of birth and haul-out lairs (McLaren, Reference McLaren1958; Smith & Stirling, Reference Smith and Stirling1975; Hammill & Smith, Reference Hammill and Smith1989). Polar bears, particularly adult females with cubs (Stirling et al., Reference Stirling, Andriashek and Calvert1993), detect these lairs by smell and dig into them to capture the seals within. Other age and sex classes of polar bears often forage farther offshore near the floe edge (Stirling et al., Reference Stirling, Andriashek and Calvert1993).
Within the Central Arctic DU sea ice provides both a barrier and a connection between subpopulations. For instance, multi-year ice in eastern Viscount Melville Sound separates this subpopulation from the Lancaster Sound subpopulation. However, bears from both areas may use the multi-year ice as a summer refuge (Taylor et al., Reference Taylor, Akeeagok, Andriashek, Barbour, Born and Calvert2001), which contributes to their genetic similarity (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999). Lancaster Sound and Baffin Bay polar bears are separated, mainly during the summer, by the North Water Polynya, a large area of annually-recurring open water (Stirling, Reference Stirling1997). Although polar bears would be unlikely to swim across such an expanse of water, the sea ice surrounding the North Water is probably dynamic enough to provide a connection between Lancaster Sound and Baffin Bay. Consistent with these oceanographic connections, polar bears within the Central Arctic DU display strong genetic (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999) and foraging similarities (Thiemann et al., in press; see below).
The Baffin Bay and Davis Strait subpopulations are separated for management purposes by a line that extends from Cape Dyer on Baffin Island to Disko Island, Greenland. In winter and early spring this area is generally covered with consolidated pack ice that could easily be traversed by polar bears (Stirling et al., Reference Stirling, Calvert and Andriashek1980). However, ocean currents and patterns of sea ice breakup and freeze-up contribute to the separation of Baffin Bay and area of Davis Strait. Specifically, the West Greenland current flows northward along the western coast of Greenland before a portion of the current is shifted westward by the submarine Davis Strait Ridge (Dunbar, Reference Dunbar1951). The result is a counterclockwise gyre in the area of the Davis Strait subpopulation (Stirling et al., Reference Stirling, Calvert and Andriashek1980). Within the Baffin Bay region the West Greenland Current continues northward before joining the south-flowing Baffin Bay Current. This south-flowing cold water current, along with the deep fiords and bays of eastern Baffin Island, results in earlier freeze-up and later breakup in Baffin Bay than in Davis Strait. Therefore, despite ocean currents that flow across the subpopulation boundary, different ecological conditions in Baffin Bay and Davis Strait appear to discourage movement between these subpopulations (Stirling et al., Reference Stirling, Calvert and Andriashek1980) and contribute to a degree of spatial separation that has resulted in significant genetic differences (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999). Fisheries and Oceans Canada has also identified the areas of Baffin Bay and Davis Strait as distinct ecoregions, with the boundary occurring at Cape Dyer (Powles et al., Reference Powles, Vendette, Siron and O'Boyle2004).
Consistent with previous analyses of Canada's ocean ecosystems (Powles et al., Reference Powles, Vendette, Siron and O'Boyle2004; Spalding et al., Reference Spalding, Fox, Allen, Davidson, Ferdańa and Finlayson2007), we identified the polar bear subpopulations of Foxe Basin, Western Hudson Bay and Southern Hudson Bay as comprising a single DU. This area is heavily influenced by a large terrestrial freshwater input. A weak ocean current enters the area through Fury and Hecla Strait and another current leaves via Hudson Strait (Dunbar, Reference Dunbar1951). The entire region becomes ice-free in summer and polar bears throughout the DU spend significant amounts of time on land without access to prey (Stirling et al., Reference Stirling, Jonkel, Smith, Robertson and Cross1977; Stirling & Parkinson, Reference Stirling and Parkinson2006). Although the ice-free season is shorter in Foxe Basin than in Hudson Bay, the overall sea ice regime and geographical isolation of the area appears to contribute to the genetic (Paetkau et al., Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999) and ecological similarities (Thiemann, Reference Thiemann2006) among bears within the Hudson Bay DU.
Productivity and prey diversity Sea ice plays a significant role in determining productivity and ecosystem structure, which show distinct variation among the proposed DUs. In the Beaufort Sea primary productivity is generally lower than in the eastern Canadian Arctic. Primary production in the nearshore Beaufort Sea is 10–15 g m-2 yr-1 (Alexander, Reference Alexander, Reed and Sater1974) compared with 60 g m-2 yr-1 in Lancaster Sound (Welch et al., Reference Welch, Bergmann, Siferd, Martin, Curtis and Crawford1992) and 40–75 g m-2 yr-1 in Baffin Bay (Grainger, Reference Grainger, Cameron and Billingsly1975). The relatively low productivity of the Beaufort Sea results in lower ringed seal densities (Stirling & Øritsland, Reference Stirling and Øritsland1995) and a marine mammal community limited to ringed seals, bearded seals, and beluga Delphinapterus leucas and bowhead whales Balaena mysticetus.
Productivity in polynyas may be several times higher than in adjacent ice-covered water and these annually-recurring areas of open water provide important habitat for Arctic sea birds and marine mammals (Stirling, Reference Stirling1980). One of the most biologically productive areas of the Beaufort Sea is the Cape Bathurst Polynya, in Amundsen Gulf between Banks Island and the mainland coast (Fig. 1; Stirling, Reference Stirling1997). This area is important foraging habitat for bears of both Beaufort Sea subpopulations (Stirling, Reference Stirling2002) and the shared use of this important biological feature supports the consideration of a single Beaufort Sea DU.
In the High Arctic DU primary productivity may be reduced because less light penetrates the abundant multi-year ice than through annual ice. However, there is considerable annual ice in the south of this area and primary productivity may be underestimated (Pomeroy, Reference Pomeroy1997).
The relatively high biological productivity of the Lancaster Sound region (Welch et al., Reference Welch, Bergmann, Siferd, Martin, Curtis and Crawford1992) supports a rich diversity of marine mammals that may be preyed upon by bears in adjacent subpopulations. Ringed seals, bearded seals, and beluga whales are abundant and polar bears may also hunt narwhals Monodon monoceros, walrus Odobenus rosmarus and harp seals Pagophilus groenlandicus in some areas of the Arctic archipelago (Thiemann, Reference Thiemann2006). Bears in Lancaster Sound, Kane Basin, and Baffin Bay share access to a variety of marine mammal prey that occur in and around the North Water Polynya, and smaller recurring polynyas and shoreleads occur throughout the archipelago (Stirling, Reference Stirling1997). The combination of shallow, productive seas and a mix of annual and multi-year ice and open water create ecological conditions that are similar across the Central Arctic DU.
The relatively warm waters of the North Atlantic in the Davis Strait region provide high biological productivity and a diversity of marine mammal prey. There are major whelping patches of both harp seal and hooded seal Cystophora cristata in this area and both species are predated by polar bears in Davis Strait (Iverson et al., Reference Iverson, Stirling, Lang, Boyd, Wanless and Camphuysen2006; Thiemann et al., in press). The unique prey assemblage and oceanographic characteristics of the Davis Strait region support its consideration as a separate DU.
The overall productivity of Hudson Bay is generally intermediate between the Arctic Basin and the North Atlantic (Stirling et al., Reference Stirling, Jonkel, Smith, Robertson and Cross1977). In addition to ringed seals and bearded seals, polar bears in the Hudson Bay DU have access to beluga whales and localized or seasonal access to harp seals, harbour seals Phoca vitulina, walrus and narwhals (Thiemann, Reference Thiemann2006).
Morphology, ecology and life history Inherited characters such as morphology, life history and behaviour provide evidence of genetic distinctiveness between groups (COSEWIC, 2005). Because they are affected by factors such as food supply and habitat quality, such phenotypic traits also reflect localized ecological differences. Thiemann et al. (in press) examined the fatty acid signatures of polar bears across their Canadian range to generate quantitative estimates of diet composition (Iverson et al., Reference Iverson, Field, Bowen and Blanchard2004, Reference Iverson, Stirling, Lang, Boyd, Wanless and Camphuysen2006). The overall diet patterns mirrored the genetic relationships identified by Paetkau et al. (Reference Paetkau, Amstrup, Born, Calvert, Derocher and Garner1999; Figs 3 & 4). Specifically, polar bears appear to forage beyond their own subpopulation boundaries but prey resources and polar bear diets are similar within the proposed DUs.
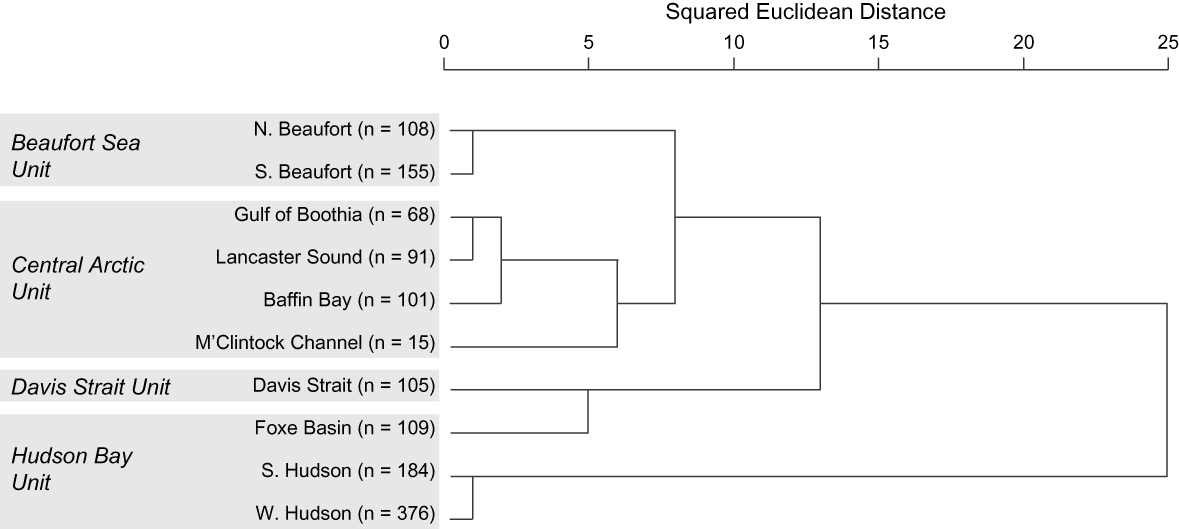
Fig. 3 Hierarchical cluster analysis of average fatty acid signatures of polar bears (≥ 2.5 years old) in 10 Canadian subpopulations, with designatable units (see text for details) indicated. Clusters were formed based on squared Euclidean distance, using 64 fatty acids and the between-groups linkage method. Modified from Thiemann et al. (in press).
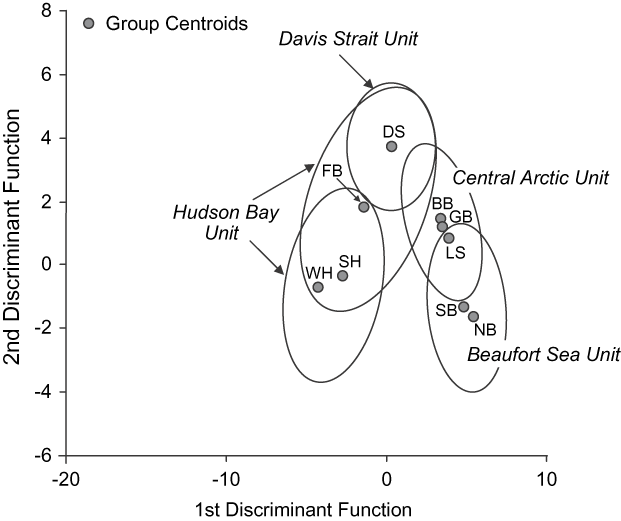
Fig. 4 Discriminant analysis of polar bears (≥ 2.5 years old) in nine Canadian subpopulations (for abbreviations see Fig. 1a), with designatable units (see text for details) indicated, using 17 fatty acids and the same samples as Fig. 3. The first and second discriminant functions accounted for 88% of total variance. Discriminant analysis classified 80.9% of original cases and 79.8% of cross-validated cases to their correct region. Ellipses represent 95% data point clouds. Modified from Thiemann et al. (in press).
Consistent with the relatively low ecosystem productivity, polar bears in the Beaufort Sea DU are generally smaller (Derocher & Stirling, Reference Derocher and Stirling1998) and the age of females at first reproduction is later (Stirling, Reference Stirling2002) than bears in the eastern Canadian Arctic. Females in the Beaufort Sea do not begin breeding until 5 years of age, 1 year later than in Western Hudson Bay, Davis Strait or Lancaster Sound (Stirling et al., Reference Stirling, Pearson and Bunnell1976, Reference Stirling and Kiliaan1980, Reference Stirling, Calvert, Andriashek, Olson, Hastings and Geddes1984; Ramsay & Stirling, Reference Ramsay and Stirling1988; Derocher & Stirling, Reference Derocher and Stirling1998). Multi-year ice is available to bears in the Beaufort Sea year-round as the floe edge recedes to the north in summer but does not melt away completely. Therefore, unlike most other areas, females in the Beaufort Sea DU can excavate maternity dens on the sea ice (Lentfer, Reference Lentfer1975; Amstrup & Gardner, Reference Amstrup and Gardner1994). However, when the sea ice retreats past the edge of the continental shelf it lies over deep water that is relatively unproductive (Regehr et al., Reference Regehr, Hunter, Caswell, Amstrup and Stirling2007a).
Data on the ecology and life history of polar bears in the High Arctic DU are limited. Considering the preponderance of multi-year ice and the low densities of ringed seals (Kingsley et al., Reference Kingsley, Stirling and Calvert1985), bears in this DU are probably small and slow-growing. The limited data suggest that reproductive rates are low even among prime-age females (> 6 years old; IUCN/SSC Polar Bear Specialist Group, Reference Aars, Lunn and Derocher2006).
Within the Central Arctic DU ecological and reproductive productivity are locally variable. Low ecosystem productivity contributes to the small size (Derocher & Stirling, Reference Derocher and Stirling1998) and slow reproduction (IUCN/SSC Polar Bear Specialist Group, Reference Aars, Lunn and Derocher2006; Taylor et al., Reference Taylor, Laake, McLoughlin, Cluff and Messier2006) of bears in the Gulf of Boothia and M'Clintock Channel relative to other areas. In contrast, primary productivity may be high in the Lancaster Sound portion of the Central Arctic DU and polar bears may grow larger there (Derocher & Stirling, Reference Derocher and Stirling1998) and have higher litter production rates (IUCN/SSC Polar Bear Specialist Group, Reference Aars, Lunn and Derocher2006). Although growth rates and productivity may show localized differences within the Central Arctic DU, there are general similarities in denning habitat, sea ice regime, and foraging patterns (Figs 3 & 4) across this region.
Reproductive rates in the Davis Strait DU tend to be higher than in most other areas of the Canadian Arctic (Stirling et al., Reference Stirling, Calvert and Andriashek1980), probably because of the region's relatively warm waters, high productivity, and diverse and abundant prey fauna. In addition to ringed, bearded, and harbour seals, approximately 6 million harp seals inhabit the waters of the North-west Atlantic (DFO, 2005). Each spring, harp seals congregate in vast numbers along the coasts of Newfoundland and Labrador to pup and moult, and dietary analyses indicate that polar bears from the Davis Strait DU make extensive use of this resource (Iverson et al., Reference Iverson, Stirling, Lang, Boyd, Wanless and Camphuysen2006; Thiemann et al., in press). Satellite telemetry indicates that polar bears from south-eastern Baffin Island migrate southward to the whelping patch in spring before returning to the Island for the ice-free season (I. Stirling, unpubl. data). Predictable access to huge numbers of harp seals is a defining characteristic of the Davis Strait DU.
The life history pattern of polar bears in Hudson Bay is defined by a prolonged onshore period that begins when the sea ice of Hudson Bay melts completely in June or July (Jonkel, Reference Jonkel1969; Stirling et al., Reference Stirling, Jonkel, Smith, Robertson and Cross1977). While on land the bears fast and rely on stored fat reserves until freeze-up c. 4 months later. Pregnant females remain on shore for 8 months or more during which time they den and give birth to their young (Ramsay & Stirling, Reference Ramsay and Stirling1988). The counterclockwise currents of Hudson Bay cause the ice to breakup earliest in the western portion of the Bay, with bears coming ashore earliest in north-eastern Manitoba. Breakup occurs later along the coasts of Ontario and Quebec and the bears of Foxe Basin move ashore latest as the sea ice may persist in this area until late August. Despite localized differences in the timing of sea ice formation and breakup, polar bears throughout the Hudson Bay DU are likely to be affected by long-term climate warming (Stirling et al., Reference Stirling, Lunn and Iacozza1999; Derocher et al., Reference Derocher, Lunn and Stirling2004; Stirling & Parkinson, Reference Stirling and Parkinson2006; Obbard et al., Reference Obbard, Cattet, Moody, Walton, Potter, Inglis and Chenier2006, Reference Obbard, McDonald, Howe, Regehr and Richardson2007; Regehr et al., Reference Regehr, Lunn, Amstrup and Stirling2007b). Spatial variability in sea ice breakup may also diminish established population boundaries as bears remain on the ice longer and potentially show reduced fidelity to onshore sites (Derocher et al., Reference Derocher, Lunn and Stirling2004).
Polar bear conservation in Canada
If a species as a whole shares a single conservation status then DUs are unnecessary (Green, Reference Green2005). However, there is considerable variation in important aspects of the biology and habitat of polar bears in Canada and we therefore conclude that a DU approach to their conservation is warranted. The significance of this ecological variability has increased with the onset of rapid climate warming and degradation of the sea ice habitat. The five DUs proposed here are not intended to serve as management units, and the sustainable harvest of polar bears should continue to be managed at the level of independent subpopulations. Rather, the DUs we have proposed represent units that are genetically, geographically, and ecologically separable. Next, we examine how these groups differ in the threats they face and/or their risk of extinction.
Polar bears in the southernmost portion of their range have been those most affected by climate warming (Stirling et al., Reference Stirling, Lunn and Iacozza1999, Reference Stirling, McDonald, Richardson and Regehr2004; Derocher et al., Reference Derocher, Lunn and Stirling2004; Stirling & Parkinson, Reference Stirling and Parkinson2006; Regehr et al., Reference Regehr, Lunn, Amstrup and Stirling2007b). Because of progressively earlier sea ice breakup in Western Hudson Bay, Southern Hudson Bay and Foxe Basin (Stirling & Parkinson, Reference Stirling and Parkinson2006), bears in the Hudson Bay DU have less time to hunt seals in spring and summer when ringed seals are most available and polar bear food intake is at its peak. Consequently, the bears are forced to fast onshore for longer periods with less stored fat on their bodies (Stirling et al., Reference Stirling, Lunn and Iacozza1999; Obbard et al., Reference Obbard, Cattet, Moody, Walton, Potter, Inglis and Chenier2006). Because parturient females rely on stored fat reserves for gestation and the first 3 months of lactation, this decline in condition has resulted in declining reproductive performance and population size in Western Hudson Bay (Derocher & Stirling, Reference Derocher, Stirling, McCullough and Barrett1992, Reference Derocher and Stirling1995; Stirling et al., Reference Stirling, Lunn and Iacozza1999; Regehr et al., Reference Regehr, Lunn, Amstrup and Stirling2007b). Although the harvest quota for this population was recently reduced in response to the population decline, the impacts of climate-related habitat loss will make sustainable management of Canada's polar bear populations increasingly difficult.
Climate change is also affecting the other DU regions, albeit at different rates and in different ways. In the Beaufort Sea, Fischbach et al. (Reference Fischbach, Amstrup and Douglas2007) found that the proportion of females denning on the pack ice is declining as more bears den on land in Alaska, apparently in response to larger areas of open water. Amstrup et al. (Reference Amstrup, Stirling, Smith, Perham and Thiemann2006) documented three separate cases of polar bears hunting and cannibalizing other polar bears in 2004, and Stirling et al. (Reference Stirling, Richardson, Thiemann and Derocher2008) discovered 14 sites where polar bears had clawed through solid ice in attempts to capture ringed seals below sheets of rafted ice. In both reports, the authors suspected that reductions in suitable sea ice habitat had played a role. Recent studies have found that population growth in Southern Beaufort Sea becomes negative when the ice-free period exceeds c. 125 days (Hunter et al., Reference Hunter, Caswell, Runge, Regehr, Amstrup and Stirling2007; Regehr et al., Reference Regehr, Hunter, Caswell, Amstrup and Stirling2007a). Polar bear populations will probably decline rapidly if recent patterns of ice distribution and open water persist (Hunter et al., Reference Hunter, Caswell, Runge, Regehr, Amstrup and Stirling2007; Regehr et al., Reference Regehr, Hunter, Caswell, Amstrup and Stirling2007a).
Analysis of new Davis Strait DU inventory data is currently underway but existing information is scarce (IUCN/SSC Polar Bear Specialist Group, Reference Aars, Lunn and Derocher2006). However, this area has undergone substantial warming and sea ice decline over the last 2 decades (Comiso & Parkinson, Reference Comiso and Parkinson2004; Moore, Reference Moore2006). The extent of sea ice may affect the survival and abundance of harp seal pups (Johnston et al., Reference Johnston, Friedlaender, Torres and Lavigne2005) and thus affect polar bear food supply (Derocher et al., Reference Derocher, Wiig and Andersen2002; Iverson et al., Reference Iverson, Stirling, Lang, Boyd, Wanless and Camphuysen2006). In the Central Arctic DU multi-year ice provides a more predictable environment for polar bears relative to southern populations, where the annual ice-free season largely defines life history patterns. However, it seems likely that continued climate warming will negatively affect bears in the Central Arctic DU. Because of its extreme latitude, the High Arctic DU may be protected from the effects of climate change in the short-term. However, its small population size (200–300 individuals) makes it vulnerable to Allee effects (Molnár et al., Reference Molnár, Derocher, Lewis and Taylor2008) and genetic drift. If this DU were found to have < 250 mature individuals it may be afforded Endangered status under COSEWIC's (2006) assessment process. However, such a designation would require a full population assessment. Considering the ecological and genetic isolation of this group of polar bears, such a study would be extremely valuable.
Derocher et al. (Reference Derocher, Lunn and Stirling2004) predicted that polar bear subpopulation boundaries would weaken due to changes in sea ice distribution and habitat connectivity. In 2007, of nine female polar bears fitted with satellite collars in Western Hudson Bay, six came ashore in Southern Hudson Bay (A.E. Derocher, unpubl. data). The boundaries between management subpopulations have always been somewhat artificial because the distribution of polar bears is defined more by a continuum of home ranges than by absolute barriers to movement (Taylor & Lee, Reference Taylor and Lee1995; Amstrup et al., Reference Amstrup, Durner, Stirling and McDonald2005). It therefore seems unreasonable to expect these divisions to hold up in the face of rapid ecological change in Arctic marine ecosystems. A broad approach that accounts for large-scale ecological forces may provide a better framework for studying polar bear biology.
The five biological units we have identified differ in their conservation status and therefore warrant consideration as separate DUs under the Canadian Species at Risk Act. These DUs reflect biologically significant genetic, phenotypic, and biogeographical differences in polar bears across their Canadian range. Considering the vast geographical distribution of the species and the spatially variable ecological impacts of climate change, the continued consideration of polar bears as a single biological unit is untenable. Such an approach necessitates a single conservation strategy that within some regions may either be too conservative or too permissive. The DUs we have proposed are consistent with COSEWIC's assessment guidelines and would allow this iconic species to be more prudently conserved in the face of a rapidly deteriorating Arctic environment. We urge COSEWIC to assess the status of polar bears using the more biologically-sound approach of separate units of biodiversity.
Acknowledgements
This work was supported by the Canadian Wildlife Federation, Natural Sciences and Engineering Research Council of Canada, Alberta Ingenuity Fund, the Killam Trusts, the University of Alberta, the Canadian Wildlife Service, and WWF (Canada). M. Festa-Bianchet and an anonymous reviewer provided helpful comments on an earlier version of the manuscript.
Biographical sketches
Gregory Thiemann's research focuses on the foraging ecology of Arctic predators. He has studied the impact of environmental conditions on food web interactions in terrestrial, marine, and freshwater ecosystems. He is also a member of the IUCN/SSC Polar Bear Specialist Group. Andrew Derocher specializes in the ecology, conservation and management of large Arctic mammals. He has examined the limiting and regulating factors influencing polar bear populations for more than 20 years and is the current Chair of the IUCN/SSC Polar Bear Specialist Group. Ian Stirling has studied polar ecosystems, marine mammals and polar bears for more than 40 years. His research examines the behaviour, ecology, evolution, and conservation of marine mammals. He has been a member of the IUCN/SSC Polar Bear Specialist Group since 1974.







