Listeria monocytogenes is a foodborne pathogen which causes listeriosis, a systemic illness that can cause symptoms ranging from gastroenteritis to meningitis and severe sepsis. L. monocytogenes is environmentally widespread and able to contaminate a range of foods(Reference Lamont, Sobel and Mazaki-Tovi1), accounting for 19 and 17 % of the known causes of foodborne disease-related deaths in the USA and France, respectively(Reference Vaillant, de Valk and Baron2, Reference Scallan, Hoekstra and Angulo3). Overall, listeriosis was estimated to have caused 23 150 illnesses, 5463 deaths and 172 823 disability-adjusted life years worldwide in 2010(Reference Maertens de Noordhout, Devleesschauwer and Angulo4). L. monocytogenes is relatively resistant to diverse environments, which enables it to survive food processing and grow in refrigerated or ready-to-eat chilled foods(Reference Gilbert and Pini5). It is therefore difficult to control in the food production environment, and consequently listeriosis may arise from food contamination outbreaks or from sporadic consumption of contaminated food(Reference Fleming, Cochi and MacDonald6, Reference Lennon, Lewis and Mantell7). Recommendations to avoid listeriosis differ slightly between countries but generally include avoiding high-risk foods which are susceptible to L. monocytogenes contamination, such as pre-prepared or pre-packaged salads, pre-prepared foods, soft, semi-soft and surface-ripened cheeses, processed meats and unpasteurised dairy products, and practising appropriate food hygiene practices(8, 9).
Groups at higher risk of contracting listeriosis include the immunocompromised, the elderly, pregnant women and neonates. Pregnant women appear 10–20 times more likely to contract listeriosis(Reference Lamont, Sobel and Mazaki-Tovi1) compared with the general population, possibly owing to a down-regulation of cellular immunity(Reference Lamont, Sobel and Mazaki-Tovi1). Listeriosis in pregnancy is generally defined as a clinical illness in a mother and/or child in conjunction with isolation of L. monocytogenes from the mother, neonate, fetus or placental surface(Reference Dalton, Merritt and Unicomb10).
Listeriosis can frequently affect the fetus and neonate by transplacental transmission(Reference Lamont, Sobel and Mazaki-Tovi1). Pregnancy-related cases account for 20·7 % of listeriosis globally, with an overall case fatality of 14·9 %, including neonatal deaths(Reference Maertens de Noordhout, Devleesschauwer and Angulo4). Mothers with listeriosis may be asymptomatic or have influenza-like symptoms such as fever, malaise, myalgia or headache(Reference Lamont, Sobel and Mazaki-Tovi1, Reference Lennon, Lewis and Mantell7, 11). Listeriosis can result in more serious consequences in the fetus or neonate, including miscarriage, prematurity, central nervous system infections, septicaemia and death(Reference Lamont, Sobel and Mazaki-Tovi1, Reference Maertens de Noordhout, Devleesschauwer and Angulo4, Reference Tappero, Schuchat and Deaver12) due to immune system insufficiency.
The incidence of listeriosis during pregnancy has decreased recently in a number of countries, including France, Belgium and the USA(Reference Tappero, Schuchat and Deaver12–Reference Bertrand, Ceyssens and Yde14), possibly due to regulatory and industry efforts, or increased awareness following active prevention campaigns targeting pregnant women(Reference Tappero, Schuchat and Deaver12, Reference Girard, Leclercq and Laurent13). However, it may be under-reported due to variable or asymptomatic maternal presentation or under-recognition in spontaneous miscarriages or stillbirths. It is thus still important to minimise the risk of development of listeriosis during pregnancy due to the severity of the implications for fetal and neonatal health.
Recommendations for reducing nutritional exposure to high-risk or contaminated foods are the best ways to reduce the risk of listeriosis. The objective of the present systematic review was therefore to assess the association between foods and food preparation practices and listeriosis during pregnancy to guide the refinement of population-specific recommendations.
Methods
Eligibility criteria
We included case–control and cohort studies. Case reports, case series and reviews were excluded. Eligible studies included pregnant women or their newborn offspring with known listeriosis status. Exclusion criteria were reports on men or non-pregnant women and animal studies. Only articles published in English were included. The protocol was registered in the international prospective register of systematic reviews PROSPERO (CRD 42017056134). No ethics or institutional review board approval was required for this work.
The aetiological exposure was defined as nutritional exposure during pregnancy consistent with international population recommendations (such as Food Standards Australia New Zealand or the Centers for Disease Control and Prevention) for minimising listeriosis, including delicatessen-style meat, cold meats, dairy products made from unpasteurised milk, soft, semi-soft or surface-ripened cheeses, ready-to-eat foods, and appropriate food preparation and storage techniques(8, 9).
The primary outcome was a diagnosis of listeriosis during pregnancy. Secondary outcomes were maternal (pregnancy complications including miscarriage, preterm delivery, maternal sepsis, chorioamnionitis or hospitalisation), fetal (stillbirth) or neonatal (death, preterm birth, admission to hospital or infection) outcomes.
Information sources and search strategy
A comprehensive database search was conducted on 6 July 2017 to identify all articles published prior to this date. The following electronic databases were used to identify relevant published literature: MEDLINE in-process and other non-indexed citations (Ovid MEDLINE(R) In-Process & Other Non-Indexed Citations, Ovid MEDLINE(R) Daily and Ovid MEDLINE(R) 1946 to Present), Embase (Embase Classic + Embase 1947 to 3 July 2017), CINAHL Plus and Web of Science Core Collection. The search strategy for MEDLINE is documented in Supplementary Table S1 and was modified for the other databases using relevant subject headings. Additional eligible articles were identified by hand-searching the reference lists of all included articles, or by contacting clinical experts. Where abstracts were identified, these were included if a corresponding full-text article could be identified.
Study selection
One independent reviewer (L. J. M.) who was not blinded to the names of investigators or sources of publication, identified and selected the articles that met the inclusion criteria. A second independent reviewer (Y. V.) performed article selection on a subset (10 %) of articles. Disagreements between these two authors were discussed and resolved by consensus or arbitration.
Data extraction
Relevant data from included studies were extracted independently by one reviewer (L. J. M.), with a second reviewer (M. B. K.) independently checking data extraction for all of the eligible studies. Disagreements between these two authors were discussed and resolved by consensus or arbitration. The data extracted included information on author(s), year of publication, study design, study location, participant characteristics, nutritional exposures (retrospective or prospective) and outcomes.
Assessment of risk of bias
All included studies were evaluated by one independent reviewer (L. J. M.), with a second reviewer (M. B. K.) independently evaluating all of the eligible studies, with neither of the two authors blinded to the names of investigators or sources of publication. The quality of the included studies was assessed using criteria based on the Newcastle–Ottawa Scale for non-randomised studies, with a maximum score of 9(Reference Wells15). These criteria assess the selection of case and control groups, comparability of case and control groups, and the quality of outcome measurement. Studies were classified as good quality if they scored 3 or 4 in the selection domain, 1 or 2 in the comparability domain, and 2 or 3 in the outcome/exposure domain; fair quality if they scored 2 in the selection domain, 1 or 2 in the comparability domain, and 2 or 3 in the outcome/exposure domain; and poor quality if they scored 0 or 1 in the selection domain, or 0 in the comparability domain, or 0 or 1 in the outcome/exposure domain.
Data synthesis
Data were presented as subgroups of (1) studies where data were available for pregnant women and neonates as a separate group and (2) studies where data were not available for pregnant women and infants as a separate group but instead were presented as combined data. Due to clinical heterogeneity relating to differences in nutritional exposures, it was not possible to perform a meta-analysis, and thus results are described narratively. It was not possible to assess publication bias through funnel plots as no statistical data synthesis could be performed.
Results
Study selection
The database searches yielded 1429 citations, with an additional four articles identified from clinical experts. After removal of duplicates, 925 citations remained. On the basis of a priori selection criteria, screening of titles or abstracts resulted in eighty-four papers being identified for full-text assessment. Of these, seventy articles were excluded, with reasons for exclusion detailed in Supplementary Table S2. We included fourteen full-text articles (comprising eleven studies) for our final analysis (Fig. 1)(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, 16–Reference Schuchat, Deaver and Wenger27).
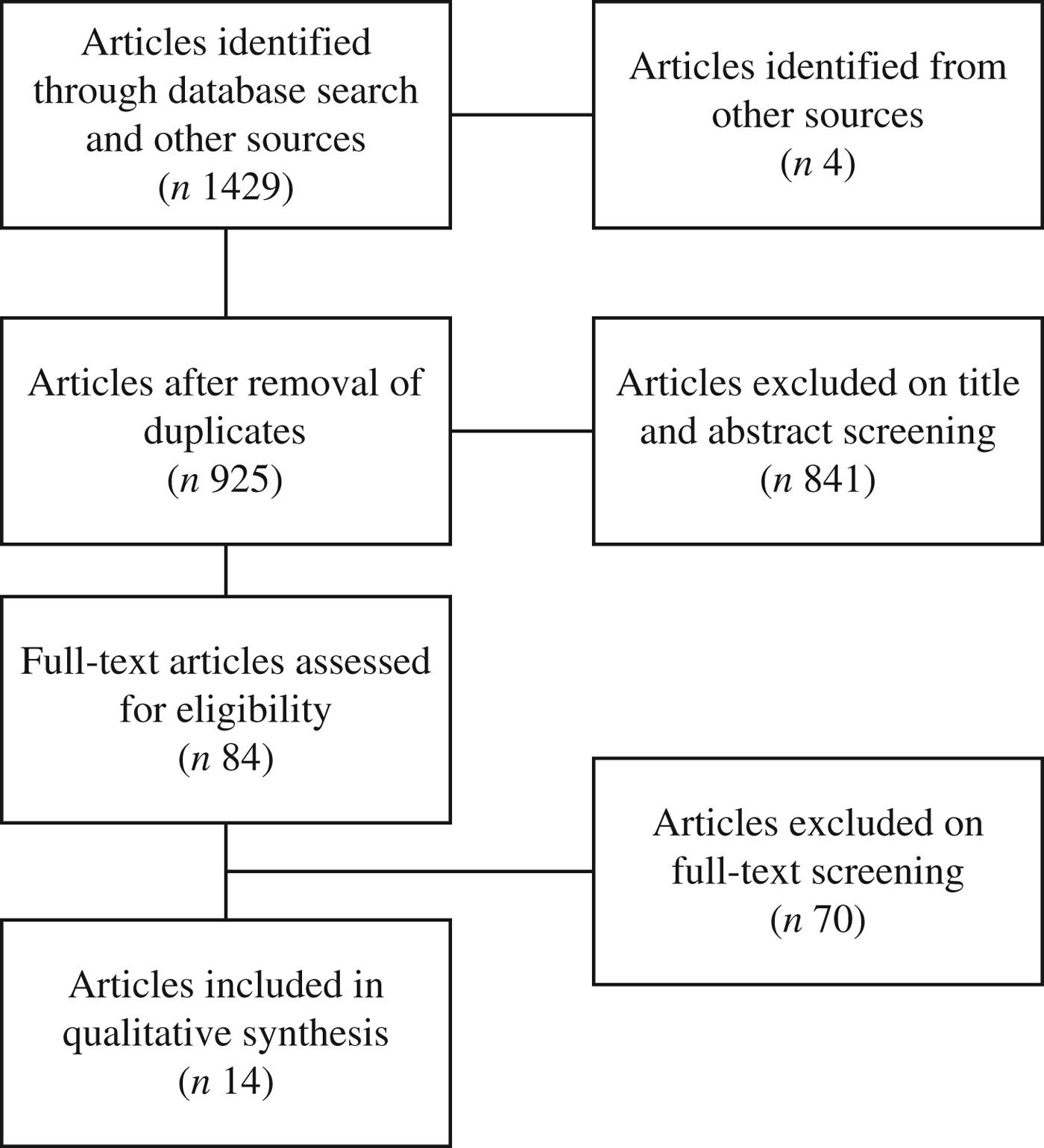
Fig. 1. Flow chart of article selection.
Study characteristics
The characteristics of included studies are presented in Table 1 and Supplementary Table S3. The studies were all case–control studies, with the exception of one cross-sectional study(Reference Pourkaveh, Ahmadi and Eslami23). The studies were conducted in the USA(Reference Fleming, Cochi and MacDonald6, Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22, Reference Schlech, Lavigne and Bortolussi24–Reference Schuchat, Deaver and Wenger27), Denmark(Reference Jensen, Frederiksen and Gernersmidt20), France(Reference Goulet, Rocourt and Rebiere19), Australia(Reference Dalton, Merritt and Unicomb10) and Iran(Reference Pourkaveh, Ahmadi and Eslami23). Five studies assessed outbreak cases(Reference Fleming, Cochi and MacDonald6, Reference Goulet, Rocourt and Rebiere19, Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22, Reference Schlech, Lavigne and Bortolussi24), five studies assessed non-outbreak cases(Reference Dalton, Merritt and Unicomb10, Reference Pourkaveh, Ahmadi and Eslami23, Reference Schwartz, Ciesielski and Broome25–Reference Schuchat, Deaver and Wenger27), and one study assessed both outbreak and non-outbreak cases(Reference Jensen, Frederiksen and Gernersmidt20) of listeriosis. The study sample sizes varied across included studies, from n 55(Reference MacDonald, Whitwam and Boggs22) to n 545(Reference Varma, Samuel and Marcus26). Where documented specifically for the cases of listeriosis during pregnancy, the participant age ranged from 16 to 40 years(Reference Goulet, Rocourt and Rebiere19, Reference Linnan, Mascola and Xiao Dong21–Reference Pourkaveh, Ahmadi and Eslami23, Reference Varma, Samuel and Marcus26). Ethnicity data were provided for a minority of studies. Of these, the majority of the study population was Caucasian(Reference Varma, Samuel and Marcus26), Hispanic(Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22), Iranian(Reference Pourkaveh, Ahmadi and Eslami23) or European(Reference Goulet, Rocourt and Rebiere19).
Table 1. Characteristics of included studies

Information on dietary exposures was collected by various methods, including a general food history(Reference Schlech, Lavigne and Bortolussi24) with an additional assessment of specific food items or food preparation methods implicated with L. monocytogenes or other foodborne diseases(Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19–Reference Pourkaveh, Ahmadi and Eslami23, Reference Varma, Samuel and Marcus26, Reference Schuchat, Deaver and Wenger27). Information was collected through methods including questionnaires, or telephone or face-to-face interviews. In all studies, nutritional information was assessed retrospectively from 1 month(Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19, Reference Jensen, Frederiksen and Gernersmidt20, Reference MacDonald, Whitwam and Boggs22, Reference Schwartz, Ciesielski and Broome25–Reference Schuchat, Deaver and Wenger27) to 3 months(Reference Pourkaveh, Ahmadi and Eslami23, Reference Schlech, Lavigne and Bortolussi24) prior to illness onset or positive culture.
Listeriosis cases were identified from medical practitioners(Reference Linnan, Mascola and Xiao Dong21, Reference Schwartz, Ciesielski and Broome25, Reference Schuchat, Deaver and Wenger27), hospital admissions(Reference MacDonald, Whitwam and Boggs22–Reference Schlech, Lavigne and Bortolussi24), medical records(Reference Linnan, Mascola and Xiao Dong21, Reference Schwartz, Ciesielski and Broome25, Reference Schuchat, Deaver and Wenger27), microbiological laboratories(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19–Reference MacDonald, Whitwam and Boggs22, Reference Varma, Samuel and Marcus26, Reference Schuchat, Deaver and Wenger27) and health departments(Reference Linnan, Mascola and Xiao Dong21, Reference Varma, Samuel and Marcus26). Controls were identified from sources including being at the same hospital(Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19–Reference Linnan, Mascola and Xiao Dong21, Reference Pourkaveh, Ahmadi and Eslami23, Reference Schlech, Lavigne and Bortolussi24, Reference Varma, Samuel and Marcus26), town or county(Reference Fleming, Cochi and MacDonald6, Reference MacDonald, Whitwam and Boggs22) or medical practitioner(Reference Fleming, Cochi and MacDonald6, Reference Goulet, Rocourt and Rebiere19, Reference Schwartz, Ciesielski and Broome25–Reference Schuchat, Deaver and Wenger27). Listeriosis during pregnancy was diagnosed as a clinical illness in a mother and/or child and/or isolation of L. monocytogenes from the mother, neonate, fetus or placental surface(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19–Reference Schlech, Lavigne and Bortolussi24, Reference Varma, Samuel and Marcus26, Reference Schuchat, Deaver and Wenger27). Specific criteria for each study included a positive culture or isolation of L. monocytogenes from mother, fetus or neonate(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19, Reference Linnan, Mascola and Xiao Dong21–Reference Schlech, Lavigne and Bortolussi24, Reference Schuchat, Deaver and Wenger27), and/or illness or death in a mother and/or child(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, Reference Jensen, Frederiksen and Gernersmidt20, Reference MacDonald, Whitwam and Boggs22, Reference Schlech, Lavigne and Bortolussi24, Reference Varma, Samuel and Marcus26), or no specific details provided(Reference Schwartz, Ciesielski and Broome25).
Risk of bias of included studies
The quality assessment of the included studies is presented in Table 1 and Supplementary Table S4. Overall, four studies were classified as good quality(Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22, Reference Schwartz, Ciesielski and Broome25, Reference Schuchat, Deaver and Wenger27), five studies were classified as fair quality(Reference Fleming, Cochi and MacDonald6, Reference Goulet, Rocourt and Rebiere19, Reference Jensen, Frederiksen and Gernersmidt20, Reference Schlech, Lavigne and Bortolussi24, Reference Varma, Samuel and Marcus26) and two studies were classified as poor quality(Reference Dalton, Merritt and Unicomb10, Reference Pourkaveh, Ahmadi and Eslami23). The case definition was adequate and was independently validated for all studies. The representativeness of the cases was adequate with the exception of one study for which it was not stated if the cases comprised all eligible cases in a certain catchment area or sample of these cases(Reference Pourkaveh, Ahmadi and Eslami23), and three studies for which the surveillance population was representative of the broader population but it was not stated how the cases were selected for the case–control study from this population(Reference Fleming, Cochi and MacDonald6, Reference Goulet, Rocourt and Rebiere19, Reference Schlech, Lavigne and Bortolussi24). The selection of the controls was adequate for all studies, with the exception of three studies where hospital controls were used(Reference Jensen, Frederiksen and Gernersmidt20, Reference Pourkaveh, Ahmadi and Eslami23, Reference Varma, Samuel and Marcus26). None of the studies stated that the controls had no history of listeriosis. For the comparability of the cases and controls on the basis of design or analysis, four studies were comparable on one factor(Reference Goulet, Rocourt and Rebiere19, Reference Schlech, Lavigne and Bortolussi24–Reference Varma, Samuel and Marcus26), and six studies(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, Reference Jensen, Frederiksen and Gernersmidt20–Reference MacDonald, Whitwam and Boggs22, Reference Schuchat, Deaver and Wenger27) were comparable on two or more factors. The cases and controls were matched for characteristics including time of birth(Reference Schlech, Lavigne and Bortolussi24), birth weight(Reference Schlech, Lavigne and Bortolussi24), gestational age(Reference Goulet, Rocourt and Rebiere19, Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22, Reference Varma, Samuel and Marcus26, Reference Schuchat, Deaver and Wenger27), maternal age(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, Reference Jensen, Frederiksen and Gernersmidt20–Reference MacDonald, Whitwam and Boggs22, Reference Schwartz, Ciesielski and Broome25, Reference Schuchat, Deaver and Wenger27), geographical area(Reference Fleming, Cochi and MacDonald6, Reference Jensen, Frederiksen and Gernersmidt20), hospital(Reference Goulet, Rocourt and Rebiere19, Reference Linnan, Mascola and Xiao Dong21), ethnicity(Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22) and health-care provider(Reference Schuchat, Deaver and Wenger27). Pourkaveh et al.(Reference Pourkaveh, Ahmadi and Eslami23) did not perform matching for any study characteristics and noted that cases were younger and were less likely to have a tertiary qualification or Internet access. The ascertainment of exposure was performed by a structured interview, where the outcome assessor was blinded to case or control status for three studies(Reference Schlech, Lavigne and Bortolussi24, Reference Schwartz, Ciesielski and Broome25, Reference Schuchat, Deaver and Wenger27), an interviewer where the outcome assessor was not blinded to case or control status for five studies(Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19, Reference Jensen, Frederiksen and Gernersmidt20, Reference MacDonald, Whitwam and Boggs22, Reference Pourkaveh, Ahmadi and Eslami23, Reference Varma, Samuel and Marcus26), and no description for two studies(Reference Fleming, Cochi and MacDonald6, Reference Linnan, Mascola and Xiao Dong21). The exposure was ascertained in the same way for cases and controls for all studies. The non-response rate was the same for cases and controls, with the exception of one study for which n 5 were missing from non-pregnancy cases(Reference Dalton, Merritt and Unicomb10), and one study where non-respondents were described(Reference Pourkaveh, Ahmadi and Eslami23).
Synthesis of results
Nutritional exposure
The dietary intake data for the included studies are presented in Table 2. Four studies reported nutritional intake data for the cases of listeriosis during pregnancy subset separately, of which two were outbreak(Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22) and two were non-outbreak(Reference Dalton, Merritt and Unicomb10, Reference Pourkaveh, Ahmadi and Eslami23). Cases were more likely to have consumed dairy products (unpasteurised dairy products(Reference Jensen, Frederiksen and Gernersmidt20), soft cheeses(Reference Pourkaveh, Ahmadi and Eslami23) or Mexican-style cheeses(Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22)), meat products (hot dogs(Reference MacDonald, Whitwam and Boggs22), semi-cooked, smoked or processed meat or smoked seafood(Reference Pourkaveh, Ahmadi and Eslami23)) or vegetables (ready-to-eat vegetables(Reference Pourkaveh, Ahmadi and Eslami23) or jicama (root vegetable)(Reference Linnan, Mascola and Xiao Dong21)). The OR for listeriosis during pregnancy after consumption of these foods ranged from 4·12 to 17·8. Conversely, there were no significant associations reported between the consumption of the following foods: rockmelon/cantaloupe, ready-to-eat fruit salad, ready-to-eat other salad, chopped liver/liverwurst, Camembert, blue-veined cheese, feta, mussels and listeriosis in one study(Reference Dalton, Merritt and Unicomb10).
Table 2. Dietary intake and maternal, neonatal and infant outcomes

For the studies reporting combined data for listeriosis during pregnancy and listeriosis not during pregnancy, three studies assessed outbreak cases(Reference Fleming, Cochi and MacDonald6, Reference Goulet, Rocourt and Rebiere19, Reference Schlech, Lavigne and Bortolussi24), three studies assessed non-outbreak cases(Reference Schwartz, Ciesielski and Broome25–Reference Schuchat, Deaver and Wenger27), and one study assessed both outbreak and non-outbreak cases(Reference Jensen, Frederiksen and Gernersmidt20) of listeriosis. Cases were more likely to have consumed dairy products (pasteurised milk(Reference Fleming, Cochi and MacDonald6, Reference Jensen, Frederiksen and Gernersmidt20), unpasteurised milk(Reference Jensen, Frederiksen and Gernersmidt20) or soft cheeses(Reference Schuchat, Deaver and Wenger27)), meat products (uncooked hot dogs, undercooked chicken(Reference Schwartz, Ciesielski and Broome25), rillettes (pork product)(Reference Goulet, Rocourt and Rebiere19) or pâté(Reference Goulet, Rocourt and Rebiere19, Reference Jensen, Frederiksen and Gernersmidt20)), fruits or vegetables (melons and hummus prepared at a commercial establishment(Reference Varma, Samuel and Marcus26) or coleslaw(Reference Schlech, Lavigne and Bortolussi24)) or food purchased from store delicatessen counters(Reference Schuchat, Deaver and Wenger27). The OR for listeriosis after consumption of these foods ranged from 1·6 to 20·5. Cases were also less likely to have consumed macaroni salad and raw apples(Reference Schuchat, Deaver and Wenger27). Only one study assessed food preparation methods and no data were reported for this outcome(Reference Schuchat, Deaver and Wenger27).
Maternal, fetal and neonatal outcomes
Secondary maternal, fetal and neonatal outcomes are reported in Table 2. These were only reported for the cases for the majority of studies(Reference Fleming, Cochi and MacDonald6, Reference Dalton, Merritt and Unicomb10, Reference Goulet, Rocourt and Rebiere19, Reference Jensen, Frederiksen and Gernersmidt20, Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22, Reference Schlech, Lavigne and Bortolussi24, Reference Varma, Samuel and Marcus26). Reported health outcomes included acute febrile illness, diarrhoea, abdominal cramps, stiff neck, vomiting, photophobia, headache, spontaneous abortion, stillbirth, premature birth, live birth of a seriously ill premature or term neonate, in utero death, meningitis and septicaemia(Reference Fleming, Cochi and MacDonald6, Reference Goulet, Rocourt and Rebiere19, Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22, Reference Schlech, Lavigne and Bortolussi24). Only one study reported outcomes (spontaneous miscarriage and gestational age at miscarriage) separately for both cases and controls(Reference Pourkaveh, Ahmadi and Eslami23). The gestational age of the current and the first spontaneous miscarriage was lower for the women with listeriosis compared with those without listeriosis.
Discussion
We report here a systematic review assessing the contribution of specific foods to listeriosis in pregnancy. Both outbreak and non-outbreak studies included in the present systematic review report that mothers with listeriosis during pregnancy were more likely to have consumed either pasteurised or unpasteurised dairy products, cooked, semi-cooked, smoked or processed meat products, or ready-to-eat foods including fruits and vegetables, although we note the OR for an increased consumption of these foods ranged from 1·6 to 20·5. With regards to secondary maternal, fetal and neonatal outcomes, these ranged from acute febrile illness and gastrointestinal discomfort to premature birth, meningitis, septicaemia and neonatal mortality. Only one study reported secondary maternal, fetal and neonatal outcomes separately for both cases and controls, and reported a lower gestational age of the current and first spontaneous abortion for cases compared with controls(Reference Pourkaveh, Ahmadi and Eslami23).
These findings support guidelines for preventing listeriosis during pregnancy. The results showed an increased risk for listeriosis during pregnancy due to the consumption of a number of dairy food products, including unpasteurised products such as raw milk, soft cheeses or Hispanic-style cheeses. These are high-risk food products for pregnant women as they either do not undergo a treatment process to remove L. monocytogenes, or L. monocytogenes can survive the production process(Reference Linton, Mackle and Upadhyay28). Furthermore, two studies comprised outbreaks associated with the consumption of Mexican-style cheese. These styles of cheeses mainly infect pregnant women with a Hispanic ethnicity in US studies(Reference Linnan, Mascola and Xiao Dong21, Reference MacDonald, Whitwam and Boggs22) and are high-risk foods for listeriosis due to the fact that they are manufactured from raw milk, undergoing processing by thermoplastification or ripening which is insufficient to kill foodborne pathogens or contamination at the post-processing stage(Reference Ibarra-Sánchez, Van Tassell and Miller29). Population recommendations should include culturally relevant information on specific higher-risk foods to avoid and safer food alternatives, in addition to safe food preparation methods(Reference Silk, Date and Jackson30). This is particularly important as there may be a lower uptake or delivery of food safety messages to ethnic minorities(Reference Mook, Grant and Little31) and higher pregnancy-associated listeriosis in ethnic minorities(Reference Filipello, Amato and Gori32, Reference Sapuan, Kortsalioudaki and Anthony33).
Outbreaks of listeriosis related to the consumption of pasteurised products can occur due to contamination after the heat-treatment process(Reference Gould, Mungai and Behravesh34). However, these result in fewer hospitalisations than outbreaks caused by unpasteurised products(Reference Gould, Mungai and Behravesh34). An association between listeriosis and pasteurised products was observed here in two studies(Reference Fleming, Cochi and MacDonald6, Reference Jensen, Frederiksen and Gernersmidt20), although we note that these were comprised predominantly of outbreak cases and that the association reported by Jensen et al.(Reference Jensen, Frederiksen and Gernersmidt20) was of borderline statistical significance. This suggests that guidelines for reducing listeriosis risk need not be modified to incorporate pasteurised products. Our results also showed an association between consumption of ready-to-eat products or products prepared in commercial establishments and a range of meat products and a higher risk of listeriosis in pregnancy including uncooked, undercooked, processed or smoked meats or pâté or smoked fish(Reference Goulet, Rocourt and Rebiere19, Reference Jensen, Frederiksen and Gernersmidt20, Reference MacDonald, Whitwam and Boggs22–Reference Schuchat, Deaver and Wenger27). These products are high risk as L. monocytogenes can grow and multiply during cold storage(Reference Chan and Wiedmann35). As these foods are often consumed without further heating, this will increase the chance of a consumer being affected by a contaminated product. This is consistent with ready-to-eat products such as delicatessen meats being classified in the very high-risk category for potential risk of listeriosis(36). This highlights the importance of physical facility, equipment design and cross-contamination controls to avoid contamination of products, and of outbreak investigations in identifying suspected food vehicles for foodborne illness.
While dietary guidelines for preventing listeriosis differ from country to country, in general they consistently cover the themes of specific high-risk foods and food handling practices to avoid(8, 9). The dietary exposures associated with listeriosis in the present systematic review were in keeping with these dietary guidelines in that unpasteurised dairy products, soft cheeses, undercooked, uncooked, smoked or processed meat, and ready-to-eat foods were associated with a higher risk of listeriosis. This highlights the utility of the current guidelines. However, there is some concern that these guidelines may overly focus on exclusion of certain foods and handling practices. It is possible that the lack of a focus on the inclusion of safer alternatives and practices may overall lead to poorer diet quality(8, 9). The uptake of the current recommendations in different populations is also unknown given variations in health literacy with demographic characteristics such as age and education(Reference van der Heide, Wang and Droomers37, Reference Berens, Vogt and Messer38). This is consistent both with our finding of younger age and lower education and Internet access for cases compared with controls(Reference Pourkaveh, Ahmadi and Eslami23). It is also consistent with previous reports of an association of listeriosis with lower age(Reference Preusel, Milde-Busch and Schmich39) or lower socio-economic status(Reference Newman, Leon and Rebolledo40), which is also of concern given potential delays in appropriate specimen collection to confirm illness for individuals living in more deprived areas(Reference Awofisayo-Okuyelu, Verlander and Amar41).
While we did not include literature reporting on listeriosis outbreaks more broadly, these were beyond the scope of the present review inclusion criteria. These focused solely on case–control and cohort studies to allow a comparison of nutritional exposures in cases compared with controls. We note important findings from the broader listeriosis outbreak literature including confirmation of high-risk foods reported here such as soft cheeses(Reference Heiman, Garalde and Gronostaj42), smoked fish(Reference Gillesberg Lassen, Ethelberg and Bjorkman43), processed meats(Reference Kvistholm Jensen, Nielsen and Bjorkman44), ready-to-eat vegetables(Reference Self, Conrad and Stroika45, Reference Cartwright, Jackson and Johnson46) and pâté(Reference Althaus, Jermini and Giannini47). We also highlight that this broader outbreak literature aids in identification of high-risk foods that were not significantly associated with listeriosis in the limited identified literature on case–control and cohort studies in pregnancy, such as rockmelon or cantaloupe(Reference McCollum, Cronquist and Silk48). These foods are well documented as being associated with outbreaks in the past which have included pregnant women(Reference McCollum, Cronquist and Silk48). Where foods identified as high risk from population outbreaks such as vegetables(Reference Self, Conrad and Stroika45, Reference Cartwright, Jackson and Johnson46) were not significantly associated with listeriosis here(Reference Dalton, Merritt and Unicomb10), the authors also comment on potential limited power to detect significant associations. This supports the inclusion of these foods in current population recommendations for minimising listeriosis(8).
We identified a relatively limited literature base of case–control studies examining the association between specific foods and listeriosis, and an even smaller number of studies that reported data for the cases during pregnancy separately(Reference Dalton, Merritt and Unicomb10, Reference Linnan, Mascola and Xiao Dong21–Reference Pourkaveh, Ahmadi and Eslami23). Furthermore, the majority of studies reported on maternal, fetal and neonatal complications associated with listeriosis just for the cases, with only one study reporting on differences in perinatal complications between cases and controls(Reference Pourkaveh, Ahmadi and Eslami23). This made it impossible for us to quantify the effects of consumption of various foods in pregnancy on adverse pregnancy outcomes mediated by listeriosis. We note that many exposures are required to cause one case of pregnancy-associated listeriosis (estimated at about 1 per 10 000 exposed pregnant women)(Reference Imanishi, Routh and Klaber49), which may limit the utility of case–control studies in assessing the relationship between outcome and exposures. Additional limitations relating to the individual studies include the possibility of recall bias due to the studies being case–control and also as dietary information was retrospectively collected 1–3 months prior to disease confirmation in all studies. The shorter time-frame may also have contributed to bias as the incubation period in pregnancy-associated listeriosis cases ranges from 17 to 67 d(Reference Goulet, King and Vaillant50). Dietary information was collected by a blinded investigator in only a limited number of studies(Reference Schlech, Lavigne and Bortolussi24, Reference Schwartz, Ciesielski and Broome25, Reference Schuchat, Deaver and Wenger27), which increases the possibility of detection bias. We also note variability in the source and matching of controls, and heterogeneity in the studies in that they comprised both outbreak and non-outbreak cases of listeriosis, which makes it difficult to interpret data more broadly for general background risk of listeriosis. The bulk of the literature (seven out of eleven studies) also focused on studies from the USA; thus research from a broader range of countries is warranted to maximise generalisability of these results. The studies also generally examined only specific food consumption, with a more limited number examining different means of food preparation(Reference Schuchat, Deaver and Wenger27), and no studies examining food hygiene practices. As all of these aspects are highlighted in recommendations for reducing listeriosis(8, 9), they also warrant assessment in future studies. Additional biases may also have occurred through identifying articles only in English.
Listeriosis is associated with severe maternal, fetal and neonatal complications and warrants consumer and health professional guidelines to minimise risk. In the present systematic review, cases of listeriosis during pregnancy were more likely to have consumed certain high-risk dairy and meat products and ready-to-eat foods in comparison with controls, with avoidance of these specific high-risk foods generally being recommended in population guidelines. Further research is warranted assessing the reach and uptake of population guidelines for preventing listeriosis in pregnant women from a range of countries, reproductive life stages and demographic backgrounds. This will aid in increasing the uptake of guidelines to reduce cases of listeriosis during pregnancy and materno-fetal morbidity and mortality.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/jns.2018.16
Acknowledgements
We acknowledge Eliza Tassone for her assistance with this manuscript.
L. J. M. is supported by a Future Leader Fellowship from the National Heart Foundation. The funding body had no role in the design, analysis or writing of this article.
L. J. M. contributed to article planning, data analysis, drafting the manuscript and critically reviewing the manuscript. Y. V. contributed to article planning, data analysis, drafting the manuscript and critically reviewing the manuscript. T. J. R. and R. C. P. contributed to article conception, data analysis and critically reviewing the manuscript. M. B. K. contributed to data analysis and critically reviewing the manuscript.
All authors declare no conflict of interest.





