The coronavirus disease 2019 (COVID-19) pandemic has had devastating effects on society and has placed a significant burden on healthcare personnel (HCP). Reference Bandyopadhyay, Baticulon and Kadhum1,Reference Mehta, Machado and Kwizera2 HCP have reported mental health concerns and burnout, frequently citing fear of SARS-CoV-2 infection. Reference Mehta, Machado and Kwizera2–Reference Asghar, Yasmin and Alvi4 Cross-sectional studies have shown that the risk of SARS-CoV-2 infection may increase for HCP who have specific, direct, patient-care roles, Reference Shepard, Kling and Lee5,Reference Wilkins, Gray and Wallia6 or who work in high-risk locations, including COVID-19 units. Reference Eyre, Lumley and O’Donnell7–Reference Sydney, Kishore, Laniado, Rucker, Bajaj and Zinaman11 However, this risk has been inconsistently validated in published studies. Reference Baker, Nelson and Overton12–Reference Steensels, Oris and Coninx16 Performing aerosol-generating procedures (AGPs) has also been emphasized as a potential risk factor for SARS-CoV-2, primarily based on experience with SARS-CoV-1. Reference Chan, Ng and Rahman17,Reference Tran, Cimon, Severn, Pessoa-Silva and Conly18
Although cross-sectional studies can provide hypothesis-generating data, they do not directly support causal inferences. Prospective cohort studies can provide data for inferring causes of infection and are therefore preferred for identifying predictors of disease. Reference Mann19,20 Current data on occupational risks for HCP are obtained from cross-sectional analyses, and a critical need remains for prospective cohort studies as the COVID-19 pandemic continues. The COVID-19 Prevention in Emory Healthcare Personnel (COPE) study was a serosurveillance cohort study to estimate the incidence of SARS-CoV-2 infection in HCP from May–December 2020 that sought to identify occupational factors associated with SARS-CoV-2 seroconversion. In our initial cross-sectional analysis at enrollment, spending >50% of a typical shift at a patient’s bedside was associated with SARS-CoV-2 seroprevalence, whereas working in a COVID-19 unit or performing AGPs was not. Reference Howard-Anderson, Adams and Sherman21 Here, we report the incident SARS-CoV-2 infections over 6 months; we assessed whether occupational activities increase the risk for SARS-CoV-2 infection.
Methods
Study recruitment, setting, and data collection
As previously described, we recruited HCP from 4 university-affiliated hospitals and associated clinics in Atlanta, Georgia. Reference Howard-Anderson, Adams and Sherman21 Hospitals included a 961-bed, academic, safety-net hospital; a 751-bed, academic, referral hospital; a 529-bed, academic, community hospital; and a 410-bed community hospital. HCP were enrolled over 5 weeks starting in May 2020. At enrollment, HCP had to (1) be >18 years old, (2) have worked ≥1 shift in the previous 2 weeks, (3) have no self-reported COVID-19 symptoms in the week prior, and (4) have no anticipated employment changes. Several COVID-19–specific infection prevention practices (ie, visitor restrictions, universal masking of HCP, symptom and temperature screening at entry, and universal testing of admitted patients) had been implemented at each hospital prior to the start of study enrollment and remained in place throughout the 6-month study period. Universal eye protection during all patient care encounters was recommended ∼2.5 months after the start of enrollment. Designated COVID-19 units were opened and closed as needed based on the volume of COVID-19 patients in each hospital. The Emory University Institutional Review Board approved the study (IRB no. 00000505). This activity was reviewed by the CDC and was conducted consistent with applicable federal law and CDC policy (See eg, 45 CFR part 46, 21 CFR part 56; 42 USC §241(d); 5 USC §552a; 44 USC §3501 et seq).
Participants were followed for 6 months and were provided serum for SARS-CoV-2 serology testing at enrollment, at 3 months, and at 6 months. All visits were completed prior to COVID-19 vaccine rollout. Participants were sent a monthly Research Electronic Data Capture (REDCap) Reference Harris, Taylor, Thielke, Payne, Gonzalez and Conde22 survey by e-mail to report occupational and community activities. Surveys focused on the prior 2 weeks of activity to maximize accuracy of recall with the assumption that these 2 weeks were representative of the entire month. The initial enrollment survey also captured information on demographics, medical history, primary occupational role, and work setting. At 6 months, participants were also asked to summarize key occupational and community activities (Supplementary Table 1 online).
Table 1. Characteristics of Healthcare Personnel and Association With SARS-CoV-2 Seroconversion Status Over 6 Months (May–December 2020)

Note. All variables are no. (%) unless otherwise stated. AGP, aerosol-generating procedure; CDC, Centers for Disease Control and Prevention; CI, confidence interval; HCP, healthcare personnel; ICU, intensive care unit; IQR, interquartile range; PPE, personal protective equipment; OR, odds ratio.
a Survey options for race included American Indian or Alaska Native, Asian, Black or African American, Native Hawaiian or other Pacific Islander, White, other race, or prefer not to answer. Due to small numbers, American Indian or Alaska Native, Native Hawaiian or other Pacific Islander and Other were combined. We excluded participants who preferred not to answer. Ethnicity was examined separately from race in part because of the small number of HCP that self-reported as Hispanic or Latino (all of whom did not seroconvert) and to maintain consistency with our previously published analysis addressing seroprevalence in this sample of HCP at enrollment. Reference Chan, Ng and Rahman17
b HCP were considered immunocompromised if they had an autoimmune or rheumatologic disorder, active malignancy, solid-organ transplant, hematologic stem cell transplant, or other self-reported immunosuppressive condition or medication.
c Participants who stated they did not have patient care roles were not asked this question on the baseline survey.
d Excludes HCP where primary location was not able to be determined due to multiple locations being reported.
e Includes participants who performed no patient care activities or worked zero shifts in the 2 weeks prior to survey completion.
f Participants without patient care were included in the reference group.
g The following procedures were specifically included as AGPs: airway suctioning, noninvasive positive pressure ventilation, manual (bag) ventilation, nebulizer treatments, intubation, cardiopulmonary resuscitation, chest physiotherapy, mini-bronchoalveolar lavage, breaking ventilation circuit, sputum induction, bronchoscopy, high-flow oxygen delivery.
h A high-risk occupational exposure to SARS-CoV-2 was defined based on the CDC guidance as having prolonged close contact with a patient(s) with SARS-CoV-2 infection while (1) the HCP was not wearing a respirator or face mask; (2) the HCP was not wearing eye protection while the patient was not wearing a face mask or intubated; or (3) the HCP was not wearing all recommended PPE (gown, gloves, eye protection, and respirator) while performing an AGP. 39
i The cumulative incidence of COVID-19 per residential ZIP code was calculated using data from the Georgia Department of Public Health (GDPH) and the US Census Bureau and includes all reported cases of COVID-19 (confirmed and probable) from 1 week prior to enrollment to 1 week prior to 6-month blood draws for each participant.
Serologic testing
A laboratory-developed, indirect enzyme-linked immunoassay (ELISA) detecting IgG antibodies against the receptor-binding domain (RBD) of SARS-CoV-2 was developed and validated similarly to previous descriptions. Reference Howard-Anderson, Adams and Sherman21,Reference Suthar, Zimmerman and Kauffman23 Sera were tested in duplicate at a dilution of 1:100. A monoclonal antibody (CR3022) Reference Yuan, Wu and Zhu24 that binds to SARS-CoV-2 was included in each plate at 200 ng per well as an internal reference to ensure comparability across plates. Longitudinal specimens from the same individual were run on the same plate. The mean optical density of test samples was divided by the mean optical density of CR3022 from the same plate to obtain the normalized ratio (NR). Samples that tested positive on the initial run were considered seropositive only if the result was confirmed on a second independent run. The threshold for detecting SARS-CoV-2 antibodies was determined by receiver operating characteristic (ROC) curve analysis to be NR ≥0.2.
Sample size calculation
This study was designed to detect differential proportions of seroconversion at 80% power with an α of .05, comparing HCP who reported high-risk occupational activities (ie, spending >50% of a shift directly at bedside, working in COVID-19 units or performing AGPs) to those who did not. We assumed an equal distribution between groups and that 30% of HCP with a high-risk occupational activity and 15% without a high-risk occupational activity would seroconvert, corresponding to a sample size of 244. Reference Dean, Sullivan and Soe25
Variable definitions and statistical analysis
The primary outcome was SARS-CoV-2 seroconversion, defined as having a negative SARS-CoV-2 ELISA result at enrollment followed by a positive result at either 3 months or 6 months. Participants with SARS-CoV-2 antibodies at enrollment were excluded from the primary analysis. Participants with negative serology results at 3 and 6 months, or with a missing 3-month result and negative 6-month result, were categorized as nonseroconverters. Participants who missed both the 3- and 6-month serologic assessments, or who had a negative result at 3 months but missed the 6-month assessment, were excluded from analyses. For the regression analysis, we also excluded participants who were not employed at a study hospital for an entire 3-month assessment period (ie, 0–3 months or 3–6 months).
We examined the following time-invariant variables: age, sex, race, ethnicity, immunocompromised status, primary hospital of employment, and occupation. HCP were considered immunocompromised if they reported an autoimmune or rheumatologic disorder, active malignancy, solid organ transplant, hematologic stem cell transplant, or taking immunosuppressive medications. Participants were classified as administrators, nurses, physicians or advanced practice providers (APPs), or other HCP based on the enrollment survey. Time-variant variables included both occupational and community activities (ie, attending gatherings with >10 people and using public transport) as well as whether participants had close contact (ie, >20 minutes of caring for, speaking with, or touching another person) with individuals with laboratory-confirmed SARS-CoV-2. Because seroconversion information was not measured monthly and because participants could only seroconvert once, longitudinal data analysis methods (ie, repeated measures analysis) could not be performed. Furthermore, while seroconversion was assessed at 2 time points, exact dates of seroconversion were unknown. Therefore, survival analysis was not appropriate for these data. To summarize the time-variant variables across the 6 surveys, we assigned the most commonly reported value for each variable. If no value was most common, then the most common and recently reported value was used (Supplementary Table 1 online).
We used proportions for categorical variables and medians with interquartile ranges (IQR) for continuous variables to characterize the study cohort and to assess changes in occupational activities over time. Univariable logistic regression was used to examine unadjusted associations between all independent variables and SARS-CoV-2 seroconversion. Multivariable logistic regression was used to examine associations between occupational variables determined a priori and SARS-CoV-2 seroconversion, adjusted for potential confounders. The primary occupational variables investigated were average proportion of each shift spent at a patient’s bedside, proportion of shifts worked in COVID-19 units, and performing or being present for ≥1 AGP. We included potential confounders related to demographics, community risk factors, and known SARS-CoV-2 exposures based on directed acyclic graph theory (Supplementary Fig. 1 online). We also included any significant variables identified in the univariable analysis (P ≤ .10) in the final model. In addition to the primary occupational exposure variables, the final model included age, race, immunocompromised status, practicing universal masking at work, having close contact with a SARS-CoV-2–infected coworker or individual in the community, and COVID-19 cumulative incidence per residential ZIP code. The incidence was calculated using ZIP code tabulation areas 26 and COVID-19 surveillance data from the Georgia Department of Public Health and includes all reported cases of COVID-19 from 1 week prior to enrollment to 1 week prior to 6-month assessment for each participant. We considered interactions among the proportion of each shift spent at the bedside, proportion of shifts worked in COVID-19 units, and race. Interactions with race could not be examined due to insufficient sample size. We detected no evidence of an interaction between proportion of shift spent at bedside and proportion of shifts worked in COVID-19 units. All analyses were performed using R version 4.0.2 software (R Foundation for Statistical Computing, Vienna, Austria).

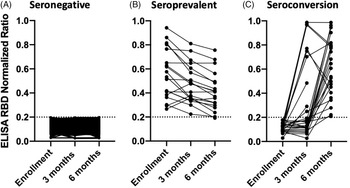
Fig. 1. Results of the enzyme-linked immunoassay (ELISA) assessing SARS-CoV-2 serology status. A participant was considered to have detectable SARS-CoV-2 antibodies if the normalized ratio was ≥0.2 (dotted horizontal line). (A) All 278 participants who did not seroconvert over the 6 months. (B) The 19 participants who had SARS-CoV-2 antibodies at enrollment and were not eligible for the seroconversion outcome. (C) The 26 participants who were seronegative for SARS-CoV-2 at enrollment and had detectable SARS-CoV-2 antibodies at 3 or 6 months.
In a post hoc analysis performed to explore reasons why we did not observe an association between AGPs and seroconversion, we examined the frequency of personal protective equipment (PPE) use among participants who performed or were present for ≥1 of the following AGPs in COVID-19 units: airway suctioning, cardiopulmonary resuscitation (CPR), intubation, and noninvasive positive pressure ventilation (NIPPV).
Results
Of the 353 enrolled COPE participants, 19 (5.4%) had SARS-CoV-2 antibodies at enrollment, 18 (95%) of whom had detectable antibodies at 6 months; 1 participant had a NR just below the limit of detection (Fig. 1). In addition to the 19 HCP with SARS-CoV-2 seroprevalent infection, we also excluded 30 participants with missed phlebotomy visits from the seroconversion analysis. Of the remaining 304 participants, 26 (9%) had SARS-CoV-2 antibodies by 6 months (9 participants by 3 months and 17 additional by 6 months). Of the 9 participants with SARS-CoV-2 seroconversion at 3 months, 7 had testing at 6 months, and all 7 remained seropositive (Fig. 1).
Of the 301 participants included in the regression analysis (3 additional participants were excluded due to no longer being employed at a study hospital), 172 (57%) were <40 years old, 231 (77%) were female, 219 (73%) were White race, and 24 (8%) were immunocompromised. Nurse was the most common occupation (n = 119, 40%), followed by physician or APP (n = 79, 26%), other HCP (n = 72, 24%), and healthcare administrator (n = 31, 10%). Also, 121 (40%) worked primarily in inpatient medical-surgical floors (Table 1).
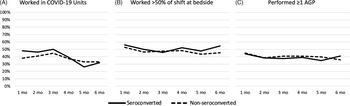
On average, 289 participants (range, 281–296) completed each survey (Supplemental Table 2). Among them, 57 participants (19%) reported no direct patient care or shifts in most of their surveys. The proportion of HCP working shifts in COVID-19 units decreased from 48% in month 1 to 32% in month 6 for HCP who seroconverted and from 38% to 33% for HCP who did not seroconvert. The proportion of participants who reported spending >50% of a typical shift at a patient’s bedside was relatively consistent over time, ranging from 46% to 56% in HCP who seroconverted and from 44% to 52% in HCP who did not seroconvert. Similarly, a relatively constant proportion of HCP reported performing or being present for ≥1 AGP on each survey (range, 35%–44% for seroconverters and 36%–45% for nonseroconverters) (Fig. 2).
Table 2. Proportion of Participants Reporting Different Frequencies of PPE Use by Type of AGP Performed in COVID-19 Units a

Note. PPE, personal protective equipment; AGP, aerosol-generating procedure; CPR, cardiopulmonary resuscitation; PAPR, powered air-purifying respirator.
a Participants were included if they reported performing AGPs in COVID-19 units only.
b Percentages were calculated for each survey (1–6) with the denominators equal to the number of participants who performed/were present for at least 1 of the specific AGP for that survey; percentages were then averaged across surveys.
c Participants reported using N95 and PAPR separately; to combine them, we used the greatest amount of time used for either N95 or PAPR.
d Average no. of participants for specific AGP per survey, rounded to the nearest whole number.

Fig. 2. Proportion of healthcare personnel reporting the following occupational activities or characteristics at each month: (A) working at least some time in COVID-19 units; (B) working >50% of a typical shift at bedside; (C) performing ≥1 AGP. Note. mo, month; AGP, aerosol generating procedure.
In univariable analyses, HCP who identified as Black race (odds ratio [OR] 3.2; 95% confidence interval [CI], 1.1–8.2) or immunocompromised (OR, 3.2; 95% CI, 1.0–8.9) were more likely to be infected with SARS-CoV-2 over the 6-month study period. Participants who did not always or nearly always perform universal masking demonstrated a trend toward increased risk of SARS-CoV-2 seroconversion (OR, 3.5; 95% CI, 0.9–11.2).
In the multivariable analysis, HCP who identified as Black race had >4 times greater odds of seroconversion than HCP who identified as White race (adjusted OR, 4.5; 95% CI, 1.3–14.2), and race was the only variable significantly associated with SARS-CoV-2 seroconversion. None of the prespecified occupational characteristics (eg, work in COVID-19 units, proportion of typical shift spent at bedside, and performing or being present for ≥1 AGP) were associated with SARS-CoV-2 seroconversion. Additionally, a similar proportion of participants who did and did not report direct patient-care activities seroconverted (8.5% vs 9.3%; OR, 0.9; 95% CI, 0.4–2.8).
The median number of AGPs that HCP performed or were present for over the 6 months was 3.0 (IQR, 0–26.0), with the majority of participants performing ≥1 AGP (n = 172, 57%). Performing or being present for ≥1 AGP was not a risk factor for SARS-CoV-2 seroconversion (adjusted OR, 1.1; 95% CI, 0.3–4.4). In an exploratory analysis, participants frequently reported wearing appropriate PPE when performing or being present for common AGPs including airway suctioning, cardiopulmonary resuscitation (CPR), intubation, and noninvasive positive-pressure ventilation (NIPPV) in COVID-19 units (Table 2). On average, >80% of participants reported always wearing respirators [ie, N95 or powered air-purifying respirator (PAPR)] when performing or being present for these AGPs (ranging from 85% for airway suctioning to 91% for intubation). Gown and glove use was also consistently high across all AGPs studied. Always using eye protection was less common, ranging from 64% for NIPPV to 79% for airway suctioning (Table 2).
Discussion
In this cohort of >300 HCP working in a large academic health system in the southern United States, 9% seroconverted for SARS-CoV-2 from May through December 2020. SARS-CoV-2 antibodies remained positive on repeated measurements from nearly all seropositive individuals for 3–6 months. We collected detailed information on monthly occupational activities, but we did not detect differences in SARS-CoV-2 seroconversion based on job title or specific healthcare-related activities.
Our results are consistent with those of prior cross-sectional studies that did not find an association between occupational role, patient care activities, or contact with SARS-CoV-2 infected patients and SARS-CoV-2 infection. Reference Baker, Nelson and Overton12,Reference Jacob, Baker and Fridkin14,Reference Steensels, Oris and Coninx16,Reference Braun, Moreno and Buys27 Community factors, including SARS-CoV-2 community incidence and close contact with SARS-CoV-2–infected individuals outside the workplace, are associated with the greatest risk of SARS-CoV-2 infection. Reference Baker, Nelson and Overton12,Reference Jacob, Baker and Fridkin14,Reference Steensels, Oris and Coninx16,Reference Thompson, Mousa and Dighe28,Reference Kayı, Madran and Keske29 One study performed genomic sequencing on SARS-CoV-2 from 95 HCP and their at-work contacts. These researchers found strong evidence for transmission from a coworker or patient in only 11% and 4% of infections, respectively. Reference Braun, Moreno and Buys27 In our study, we did not detect an association between community factors (eg, attending gatherings of >10 people, using public transport, or community incidence of COVID-19) and SARS-CoV-2 seroconversion, possibly due to a small sample size and because nearly all participants lived in the same metropolitan area. We did, however, observe an adjusted association between HCP who identify as Black race and SARS-CoV-2 infection, which may be a reflection of existing community inequities and structural racism Reference Wiley, Ross-Driscoll, Wang, Smothers, Mehta and Patzer30–Reference Scannell Bryan, Sun and Jagai33 or unmeasured disparities in workplace exposures.
In our assessment of SARS-CoV-2 seroprevalence at enrollment, we identified greater time at the bedside as a risk factor for previous SARS-CoV-2 infection; however, we detected no association with seroconversion in this study. Reference Howard-Anderson, Adams and Sherman21 This finding is likely due to healthcare-associated transmission early in the pandemic before PPE use and other mitigation practices were widely implemented. Other cross-sectional seroprevalence studies performed in the early months of the pandemic also found higher seroprevalence rates for HCP working in the emergency department, an area with high patient contact and turnover. Reference Purswani, Bucciarelli and Tiburcio10,Reference Sydney, Kishore, Laniado, Rucker, Bajaj and Zinaman11 In this study, universal testing for SARS-CoV-2 in admitted patients began just prior to COPE enrollment, and the knowledge of the test results may have modified behavior of HCP. Additionally, wearing eye protection for all patient encounters was not recommended for HCP until >2.5 months into the COPE study, which could have also contributed to the decreased risk for HCP spending most of their shift at a patient’s bedside. Reference Shah, Breeher, Hainy and Swift34 Given the low proportion of seroconversion in our study, we could not examine the impact of eye protection further.
Contrary to initial expectations, we did not detect an association between AGPs and SARS-CoV-2 infection. Assessing the true risk associated with AGPs is complicated because there is no consensus on what constitutes an AGP. Reference Klompas, Baker and Rhee35 Klompas et al Reference Klompas, Baker and Rhee35 argued that the risk of SARS-CoV-2 transmission during medical procedures likely depends on many factors, including generation of forced air, patient symptoms, distance of HCP from a patient, and the amount of time an HCP spends with a patient. Data like ours, which rely on simple AGP categorization, may not adequately capture all factors contributing to risk of SARS-CoV-2 infection. Additionally, self-reported PPE use during AGPs was relatively high, and PPE use has been shown to effectively reduce the risk of coronavirus infections. Reference Chan, Ng and Rahman17
Our findings are consistent with other studies demonstrating that anti–SARS-CoV-2 spike-protein antibodies in infected individuals including HCP are relatively stable up to 8 months. Reference Lumley, Wei and O’Donnell36,Reference Dan, Mateus and Kato37 Only 1 participant in our study who had SARS-CoV-2 antibodies at enrollment seroreverted by 6 months, with an assay result that was just below the limit of detection. Our data suggest that using 3-month intervals to assess serologic status in immunocompetent adults is reasonable without significant risk of misidentification due to waning antibodies.
Obtaining iterative, detailed assessments of occupational activities with high rates of participation is a notable strength of this study. Altghough our final model summarized the monthly survey results into single variables for each exposure, the repeated assessments allowed for a more complete picture of typical occupational activities.
This study had several limitations. Our sample size was small, and the proportion of HCP with seroconversion was lower than originally anticipated, which likely resulted in inadequate power to detect small increases in SARS-CoV-2 infection risk. This factor may account for why we did not detect associations between seroconversion and factors such as inconsistent universal masking and community exposure to a SARS-CoV-2 contact. Second, we only assessed seroconversion at 3 and 6 months,which limited our ability to link specific activities to date of seroconversion. Too few HCP had seroconverted by 3 months to perform a separate analysis at that time point. Third, this study was not designed to be an in-depth assessment of community risk factors for SARS-CoV-2, and we did not have detailed information about community activities, mask use outside of the hospital, or social determinants of health. Lastly, the ELISA detecting SARS-CoV-2 was highly specific (>99%) but may have underestimated the number of true seroconversions given the sensitivity of 86%.
In summary, nearly 10% of HCP working in an academic healthcare system from May 2020 to December 2020 were infected with SARS-CoV-2. HCP with increased patient-care activities were not more likely to be infected, and we did not identify specific occupational factors that increased risk for SARS-CoV-2 infection. Our results suggest that, with high rates of PPE use and effective institutional infection prevention measures, the risk of SARS-CoV-2 for HCP was relatively low and depended more on individual behaviors or community factors. Future work could help define what PPE is needed for specific patient-care activities, especially in the context of effective SARS-CoV-2 vaccination. Reference Hall, Foulkes and Saei38
Supplementary material
To view supplementary material for this article, please visit https://doi.org/10.1017/ice.2021.518
Acknowledgments
We acknowledge the commitment of all healthcare personnel working during the COVID-19 pandemic and are grateful to those that volunteered for this study. We thank Kara Goldstone, Monica Godfrey, and Ellie Butler for their assistance with study operations and Jens Wrammert for support in establishing our serologic testing. We also acknowledge the Emory General Clinical Research Center (GCRC) and the Emory All of Us team who supported recruitment and study visits for specimen collection. We thank the Georgia Department of Public Health for supplying data for this project. The contents herein are those of the authors and do not necessarily represent the official views of, nor an endorsement by, the Georgia Department of Public Health. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention or the National Institutes of Health.
Financial support
This study was supported by the Centers for Disease Control and Prevention through a cooperative agreement with the Georgia Emerging Infection Program (grant no. U50CK000485). J.H.A. was supported by the Antibacterial Resistance Leadership Group fellowship (National Institute of Allergy and Infectious Diseases grant no. UM1AI104681). C.A. was supported by the Agency for Healthcare Research and Quality (grant no. R01 HS025987) and the National Science Foundation (grant no. 2032084)
Conflicts of interest
B.A.L. reports grants and personal fees from Takeda Pharmaceuticals and personal fees from World Health Organization outside the submitted work.