Many epidemiological studies conducted in children and adolescents have shown an inverse association between eating frequency (EF) and adiposity measures( Reference Ritchie 1 – Reference Kontogianni, Farmaki and Vidra 14 ), with some exceptions( Reference Thompson, Ballew and Resnicow 15 – Reference Murakami and Livingstone 20 ). However, these results should be interpreted with caution given the methodological limitations associated with these studies. First, although the assessment of EF has often relied on a series of non-validated, self-report questions( Reference Barba, Troiano and Russo 3 , Reference Antonogeorgos, Panagiotakos and Papadimitriou 4 , Reference Cassimos, Sidiropoulos and Batzios 7 – Reference Neutzling, Taddei and Gigante 11 , Reference Toschke, Kuchenhoff and Koletzko 13 , Reference Ferreira and Marques-Vidal 17 ), only a few studies have assessed EF on the basis of information on actual dietary habits (using dietary record or 24-h recall)( Reference Ritchie 1 , Reference Lioret, Touvier and Lafay 12 , Reference Thompson, Ballew and Resnicow 15 , Reference Murakami and Livingstone 20 ). Second, the association of EF with adiposity measures may be confounded by possible under-reporting of EF concomitant with the under-reporting of energy intake (EI) by obese or overweight subjects( Reference McCrory, Howarth and Roberts 21 , Reference Bellisle, McDevitt and Prentice 22 ). For example, one study found that when subjects with implausible EI were eliminated from the analytic sample, the inverse relation between EF and BMI percentile no longer existed among children and adolescents( Reference Huang, Howarth and Lin 19 ). Third, interpreting the literature on EF is complicated by the fact that there is no consensus about what constitutes a snack, a meal or an eating occasion. Although some researchers have relied on respondents’ self-identification of meals, snacks or eating occasions( Reference Keast, Nicklas and O’Neil 2 – Reference Antonogeorgos, Panagiotakos and Papadimitriou 4 , Reference Cassimos, Sidiropoulos and Batzios 7 – Reference Toschke, Kuchenhoff and Koletzko 13 , Reference Ferreira and Marques-Vidal 17 , Reference Nicklas, Yang and Baranowski 18 ), others have attempted to use more objective criteria( Reference Ritchie 1 , Reference Kontogianni, Farmaki and Vidra 14 – Reference Jennings, Cassidy and van Sluijs 16 , Reference Huang, Howarth and Lin 19 , Reference Murakami and Livingstone 20 ) to overcome concerns over definitional differences. More importantly, potentially different effects of meal frequency (MF) and snack frequency (SF) have not been investigated simultaneously using different definitions of meals and snacks. Further, investigation of the association between MF and SF and dietary intake is extremely limited( Reference Evans, Jacques and Dallal 23 ). As a consequence of these methodological limitations, the discrepant findings are not surprising, which clearly bring into question the direction of the relationship of MF and SF with diet quality and adiposity measures and whether a relationship even exists.
An accurate distinction between meals and snacks is important, because they are hypothesised to have opposite effects on energy balance. A high MF may prevent fat mass deposition, whereas snacking may contribute to it( Reference Chapelot 24 ). This is also important for the development of science-based recommendations (of snacking) for consumers( Reference Johnson and Anderson 25 ). Moreover, in the absence of a universally accepted definition of meals and snacks, an understanding of the influence of different meal and snack definitions on the associations with diet quality and adiposity may facilitate the interpretation of the existing literature and help establish consensus on the most appropriate research definition for meals and snacks( Reference Leech, Worsley and Timperio 26 ).
Therefore, the primary aim of the present cross-sectional study was to examine the relationship of MF and SF with food and nutrient intakes, diet quality and adiposity measures in British children and adolescents, by using different definitions of meals and snacks. The secondary aim was to examine the impact of exclusion of misreporters of EI on these associations.
Methods
Survey design
The present cross-sectional study was based on data from the National Diet and Nutrition Survey (NDNS): Young People Aged 4 to 18 Years. Data from the NDNS were obtained from the UK Data Archive, University of Essex. Full details of the rationale, design and methods of the survey have been described elsewhere( Reference Gregory and Lowe 27 , Reference Murakami, McCaffrey and Livingstone 28 ). Briefly, the sample was randomly selected from 132 randomly selected postal sectors within mainland Great Britain. Eligibility was defined as being aged 4–18 years. One eligible person per private household was selected at random. Data collection was conducted during a 12-month period (January to December 1997). This study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human subjects were approved by the National Health Service Local Research Ethics Committee covering each of the postal sectors. A verbal informed consent was obtained from all subjects and their parents/guardians. Verbal consent was witnessed and formally recorded.
Anthropometric measurements
All anthropometric measurements were performed in duplicate by trained fieldworkers, and the mean value of two measurements was used in the analysis. Height (to the nearest 0·1 cm) and weight (to the nearest 0·1 kg) were measured while subjects were barefoot and wearing only light clothes. BMI (kg/m2) was calculated as weight (kg) divided by height squared (m2) and converted age- and sex-specific z score according to British growth-reference data( Reference Cole, Freeman and Preece 29 ). For subjects aged ≥11 years, waist circumference was also measured at the midpoint between the iliac crest and the lower rib (to the nearest 0·1 cm). Waist:height ratio (WHtR) was calculated as waist circumference divided by height.
Dietary assessment
Dietary data were collected through a 7-d weighed dietary record. A detailed description of the procedure has been published elsewhere( Reference Gregory and Lowe 27 , Reference Murakami, McCaffrey and Livingstone 28 ). Briefly, the subject, the parent or both, depending on the age of the subject, were asked to keep a weighed record of all food and drinks consumed by the subject, both in and out of the home, over 7 consecutive days. They were supplied with a set of digital food scales and recording diaries and given by trained interviewers both written and verbal instructions on how to weigh and record items in the diary. When weighing was not possible (e.g. eating out), the subject was asked to record as much information as possible. Generally, children aged 10 years and over were able to complete the diary themselves, whereas for children aged <10 years the parent/guardian was expected to complete the diary. Trained interviewers visited the household at least twice during the recording period and checked the completeness of food recording. All the collected diaries were checked by trained nutritionists in terms of coding, recorded weights and descriptions of items consumed. Estimates of daily intake for foods, energy and selected nutrients were calculated on the basis of the Food Standards Agency nutrient databank( Reference Smithers 30 ), which is based on McCance and Widdowson’s composition of foods series( 31 ), and manufactures’ data where applicable. For all dietary variables, mean daily values over 7 d were used in the analysis. Values of food and nutrient intake were energy-adjusted using the density method (i.e. percentage of energy for energy-providing nutrients and amount per 10 MJ of energy for foods and other nutrients).
As a measure of diet quality, the Mediterranean diet score (MDS) was calculated. The MDS represents a Mediterranean-type diet and is based on the consumption of eight different components (vegetables, legumes, fruits, cereals, fish, the ratio of unsaturated fatty acid:SFA, meat and dairy products)( Reference Trichopoulou, Orfanos and Norat 32 , Reference Struijk, Beulens and May 33 ). To modify the score for children and adolescents, the alcohol consumption component was removed from the score. For each component, subjects with an intake (g/10 MJ) above or equal to the age group- and sex-specific median were assigned a score of 1 (a score of 0 to those below), except for meat and dairy products, for which the score was assigned in the reverse manner. Scores for all eight components were summed and resulted in a total range from 0 to 8, whereby a higher score reflected better adherence to a Mediterranean-type diet.
Definition of eating frequency, meal frequency and snack frequency
Data from the 7-d dietary record were also used to calculate the mean number of eating occasions per day, that is, EF. Eating occasions were defined as any occasion when any food or drink was consumed( Reference Ritchie 1 , Reference Lioret, Touvier and Lafay 12 , Reference Kontogianni, Farmaki and Vidra 14 , Reference Jennings, Cassidy and van Sluijs 16 , Reference Murakami and Livingstone 20 , Reference Hartline-Grafton, Rose and Johnson 34 – Reference Yannakoulia, Melistas and Solomou 36 ). If two eating occasions occurred in ≤15 min, both events were counted as a single eating occasion, and when >15 min separated two eating occasions, these were considered distinct eating occasions( Reference Kontogianni, Farmaki and Vidra 14 , Reference Thompson, Ballew and Resnicow 15 , Reference Murakami and Livingstone 20 , Reference Yannakoulia, Melistas and Solomou 36 , Reference Ma, Bertone and Stanek 37 ). EF was calculated on the basis of all eating occasions except for those providing <210 kJ of energy. This calculation method has been used in several previous studies( Reference Murakami and Livingstone 20 , Reference Hartline-Grafton, Rose and Johnson 34 , Reference Ruidavets, Bongard and Bataille 38 ) and was chosen to avoid giving undue weight to eating occasions that only included water, low-energy beverages or small quantities of foods.
All eating occasions were divided into either meals or snacks with the use of two different published definitions: on the basis of (1) contribution to total EI( Reference Ritchie 1 ) and (2) clock time( Reference Duffey, Pereira and Popkin 39 ). For the first definition( Reference Ritchie 1 ), a meal was defined as any eating episode comprising ≥15 % of total EI, regardless of the time of day or composition of foods or beverages consumed. All other eating episodes were classified as a snack. For each participant, MF and SF determined on the basis of EI contribution were calculated (hereafter referred to as MFenergy% and SFenergy%, respectively). For the second definition( Reference Duffey, Pereira and Popkin 39 ), meals were defined as eating events reported during selected times of the day, that is, 06.00–10.00, 12.00–15.00 and 18.00–21.00 hours. All other eating occasions were considered snacks. For each participant, MF and SF determined on the basis of time were calculated (hereafter referred to as MFtime and SFtime, respectively).
Assessment of non-dietary variables
The socio-economic status of head of household (i.e. occupational social class) was reported and used as a proxy for children’s social class. The following three categories were used: manual (i.e. skilled manual, partly skilled and unskilled occupations: social classes III manual, IV and V), non-manual (i.e. professional, managerial, technical and skilled non-manual occupations: social classes I, II and III non-manual) or unclassified.
For subjects aged ≥7 years, a 7-d physical activity diary was completed concurrently with the dietary record. A detailed description of the procedure has been published elsewhere( Reference Gregory and Lowe 27 , Reference Murakami, McCaffrey and Livingstone 28 ). Briefly, the subject was asked to provide information on the time spent being active from a list of prompted moderate, vigorous and very vigorous activities. Information on activities that were not already listed and sleep was also provided. Trained interviewers checked the completeness of records at least twice during the recording period. Subsequently, time spent daily in sleep, very light, light, moderate, vigorous and very vigorous-intensity activities was computed for each day of recording. The number of hours spent per day on each activity was multiplied by the metabolic equivalent value of that activity (derived from a published table)( Reference Ainsworth, Haskell and Herrmann 40 ), and all metabolic equivalent-h products were summed to produce a total metabolic equivalent-h score for the day. They were then divided by 24 h to give a physical activity level value, and classified into four categories (sedentary, low active, active and very active) according to the US Dietary Reference Intakes ( 41 ). For subjects aged ≤6 years, for which the activity diary was not collected, the ‘active’ level was assigned on the basis of a result on total energy expenditure measured using the doubly labelled water in the NDNS feasibility study( Reference Rennie, Jebb and Wright 42 ).
Evaluation of energy intake reporting
We calculated each subject’s estimated energy requirement (EER) with the use of equations published in the US Dietary Reference Intakes ( 41 ). Subjects were identified as acceptable reporters, under-reporters or over-reporters of EI on the basis of their ratio of EI:EER, according to whether the individual’s ratio was within, below or above the 95 % confidence limits of the expected ratio of 1·0. On the basis of a published equation( Reference Huang, Howarth and Lin 19 ), acceptable reporters were defined as having EI:EER in the range 0·72–1·28, under-reporters as EI:EER<0·72 and over-reporters as EI:EER>1·28. A detailed description of the procedure has been published elsewhere( Reference Murakami, McCaffrey and Livingstone 28 ).
Analytic sample
Of 2672 potentially eligible people identified for the study, 2127 (80 % of the eligible sample) participated in the survey. For the present analysis, we excluded a total of 443 subjects with missing information on the variables used (n 182 for anthropometric data, n 426 for dietary data, n 125 for physical activity data, and some subjects had more than one missing value). We further excluded forty-eight underweight subjects (i.e. BMI≤3rd percentile of the age- and sex-specific growth-reference data( Reference Cole, Freeman and Preece 29 )). The final analysis sample comprised 1636 subjects aged 4–18 years (61 % of the eligible sample).
Statistical analysis
All statistical analyses were performed for children aged 4–10 years and adolescents aged 11–18 years separately, using SAS statistical software (version 9.2; SAS Institute Inc.). Separate analyses for boys and girls showed similar patterns of associations, and tests for interaction with sex were not significant (data not shown). We therefore present results for both sexes combined. Associations among various measures of MF and SF were investigated through Pearson’s correlation analyses. Associations of MF and SF with intakes of energy and selected foods and nutrients and MDS were investigated by linear regression analyses using PROC REG procedure. Linear regression analyses were also performed to explore the associations of MF and SF with BMI z score and WHtR. With the use of the PROC REG procedure, we calculated the adjusted regression coefficients with their standard errors of variation of BMI z score and WHtR by one increase of MF and SF. Potential confounding factors considered were age, sex, social class and physical activity. In adolescents, we further included EI:EER as a potential confounding factor, because EI:EER was associated not only with BMI z score (Pearson’s r −0·24) and WHtR (Pearson’s r −0·13) but also with all measures of MF and SF (Pearson’s r 0·15–0·49) (all P≤0·0002). This approach has been used in several previous studies( Reference Jennings, Cassidy and van Sluijs 16 , Reference Murakami and Livingstone 20 , Reference Murakami, McCaffrey and Livingstone 28 , Reference Johnson, Mander and Jones 43 – Reference Kelly, Rennie and Wallace 45 ). EI was not included as a potential confounding factor not only because we considered EI as a potential causal pathway between MF and SF and adiposity measures but also because there was a strong correlation between EI and EI:EER (Pearson’s r 0·73). MF and SF were analysed continuously after confirming the linearity of relations using tertile, quartile and quintile categories. The analyses were conducted not only for the entire population but also for acceptable reporters.
Data have not been weighted to take into account known socio-demographic differences between responders and non-responders not only because the impact of this adjustment – applied as a weighting factor – for nutritional variables was extremely small and not significant( Reference Gregory and Lowe 27 ) but also because we were only interested in relationships between variables, rather than estimates of prevalence( Reference Murakami and Livingstone 20 , Reference Murakami, McCaffrey and Livingstone 28 ). All reported P values are two-tailed, and P values of <0·01 were considered statistically significant to minimise the chance of a type 1 error arising from multiple testing.
Results
The mean value of BMI z score was 0·38 in children and 0·47 in adolescents (Table 1). Under-reporting of EI compared with EER was on average 16 % in children and 30 % in adolescents. The percentages of acceptable reporters and under-reporters were 80 and 19 % in children and 47 and 52 % in adolescents, respectively (only six children (0·7 %) and three adolescents (0·4 %) were classified as over-reporters).
Table 1 Characteristics of subjects (Mean values and standard deviations; percentages)
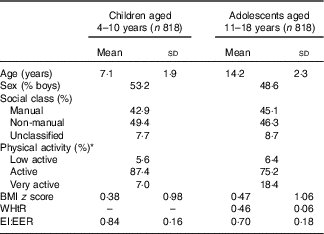
WHtR, waist:height ratio; EI:EER, ratio of energy intake to estimated energy requirement.
* There were no subjects classified into ‘sedentary’ level.
The mean values of MFenergy% and MFtime were, respectively, 2·57 and 3·39 times/d in children and 2·47 and 3·03 times/d in adolescents (Table 2). Although there was no correlation between MFenergy% and MFtime in children, MFenergy% was weakly correlated with MFtime in adolescents. The mean values of SFenergy% and SFtime were, respectively, 2·36 and 1·54 times/d in children and 2·18 and 1·62 times/d in adolescents. There were moderate correlations between two measures of SF in both children and adolescents. While MFenergy% showed a weak inverse correlation with EF in children and a weak positive correlation with EF in adolescents, there were strong correlations of MFtime, SFenergy% and SFtime with EF in both age groups.
Table 2 Descriptive statistics of meal frequency (MF), snack frequency (SF) and eating frequency (EF) (Mean values and standard deviations; percentiles; Pearson’s correlation coefficients and 95 % confidence intervals)

MFenergy%, MF determined based on percentage contribution to total energy intake; SFenergy%, SF determined based on percentage contribution to total energy intake; MFtime, MF determined based on the time consumed; SFtime, SF determined based on the time consumed.
* A meal was defined as any eating episode comprising ≥15 % of total energy intake, regardless of the time of day or composition of foods and beverages consumed; all other eating episodes were classified as a snacks.
† Meals were defined as eating events reported during selected times of the day (06.00–10.00, 12.00–15.00 and 18.00–21.00 hours); all other eating occasions were considered snacks.
In children, there were no associations between MFenergy% and any of the dietary characteristics examined (Table 3). However, MFtime, SFenergy%, SFtime and EF were associated positively with intakes of energy, dairy products, confectionery and soft drinks (except for MFtime) and inversely with legumes, cereals, fish and meat. There were inverse associations of SFtime and EF with vegetables. For nutrient intake, MFtime, SFenergy%, SFtime and EF were associated positively with SFA and total sugar and inversely with protein, PUFA, starch and dietary fibre. There was a positive association between SFtime and carbohydrate. MFtime, SFenergy%, SFtime and EF also showed inverse associations with MDS.
Table 3 Associations of meal frequency (MF), snack frequency (SF) and eating frequency (EF) with dietary characteristics in children aged 4–10 years (n 818) (Mean values and standard deviations; regression coefficients with their standard errors)

MFenergy%, MF determined based on percentage contribution to total energy intake; SFenergy%, SF determined based on percentage contribution to total energy intake; MFtime, MF determined based on the time consumed; SFtime, SF determined based on the time consumed; EF, eating frequency.
* Statistically significant at P<0·01.
† A meal was defined as any eating episode comprising ≥15 % of total energy intake, regardless of the time of day or composition of foods and beverages consumed; all other eating episodes were classified as snacks.
‡ Meals were defined as eating events reported during selected times of the day (06.00–10.00, 12.00–15.00 and 18.00–21.00 hours); all other eating occasions were considered snacks.
§ Regression coefficients mean the change of dietary variables with one additional eating occasion per day.
|| Possible score ranging from 0 to 8.
In adolescents, all measures of MF, SF and EF were positively associated with intakes of energy and carbohydrate and inversely with intakes of meat and MUFA (Table 4). MFtime, SFenergy%, SFtime and EF also showed positive associations with intakes of confectionery, soft drinks (except for MFtime) and total sugar and inverse associations with intakes of fish, cereals (except for MFtime), protein, fat, PUFA, starch and dietary fibre (except for MFtime). There were positive associations of MFtime and EF with dairy products and of SFenergy% and MFtime with fruits and an inverse association of SFtime with vegetables. Only SFtime was inversely associated with MDS. Similar results were obtained when the data for acceptable reporters were analysed separately (data not shown).
Table 4 Associations of meal frequency (MF), snack frequency (SF) and eating frequency (EF) with dietary characteristics in adolescents aged 11–18 years (n 818) (Mean values and standard deviations; regression coefficients with their standard errors)

MFenergy%, MF determined based on percentage contribution to total energy intake; SFenergy%, SF determined based on percentage contribution to total energy intake; MFtime, MF determined based on the time consumed; SFtime, SF determined based on the time consumed.
* Statistically significant at P<0·01.
† A meal was defined as any eating episode comprising ≥15 % of total energy intake, regardless of the time of day or composition of foods and beverages consumed; all other eating episodes were classified as snacks.
‡ Meals were defined as eating events reported during selected times of the day (06.00–10.00, 12.00–15.00 and 18.00–21.00 hours); all other eating occasions were considered snacks.
§ Regression coefficients mean the change of dietary variables with one additional eating occasion per day.
|| Possible score ranging from 0 to 8.
In both children and adolescents, after adjustment for age, sex, social class and physical activity (and EI:EER in adolescents), all measures of MF and SF showed no association with adiposity measures (Table 5). Similar results were obtained for those deemed to be acceptable reporters, except for the positive association between MFtime and BMI z score in adolescents.
Table 5 Associations of meal frequency (MF) and snack frequency (SF) with adiposity measuresFootnote † (Regression coefficients with their standard errors)

MFenergy%, MF determined based on percentage contribution to total energy intake; SFenergy%, SF determined based on percentage contribution to total energy intake; MFtime, MF determined based on the time consumed; SFtime, SF determined based on the time consumed; WHtR, waist:height ratio.
* Statistically significant at P<0·01.
† Adjustment was made for age (years, continuous), sex (boys or girls), social class (manual, non-manual or unclassified) and physical activity (low active, active or very active). In adolescents, further adjustment was made for ratio of energy intake:estimated energy requirement (EI:EER, continuous).
‡ Acceptable reporters were defined as subjects with EI:EER 0·72–1·28.
§ Regression coefficients mean the change of adiposity measures with one additional eating occasion per day.
|| A meal was defined as any eating episode comprising ≥15 % of total energy intake, regardless of the time of day or composition of foods and beverages consumed; all other eating episodes were classified as snacks.
¶ Meals were defined as eating events reported during selected times of the day (06.00–10.00, 12.00–15.00 and 18.00–21.00 hours); all other eating occasions were considered snacks.
Discussion
To our knowledge, this is the first study to examine the association of different measures of MF and SF with dietary intakes and adiposity measures in young populations. In this British cross-sectional study in children and adolescents, MFenergy%, MFtime, SFenergy% and SFtime were all associated with higher EI, except for MFenergy% in children. SFenergy% and SFtime were associated with higher intakes of soft drinks, confectionery and total sugar, lower intakes of cereals, fish, meat, protein, PUFA, starch and dietary fibre and a lower MDS (except for SFenergy% in adolescents). MFtime, but not MFenergy%, was associated with higher intakes of confectionery and sugar, lower intakes of fish, protein, PUFA and starch, and, only in children, a lower diet quality. All measures of MF and SF showed no association with adiposity measures in both children and adolescents. Similar results were obtained when data for subjects whose EI were deemed plausible were analysed separately. Thus, the present results demonstrated that SFenergy%, SFtime and MFtime, but not MFenergy%, were similarly associated with unfavourable dietary intake patterns, suggesting that decreasing the number of small eating occasions (<15 % of total EI) regardless of the time of day may be important to improve diet quality.
Although many epidemiological studies have investigated the association between EF (i.e. sum of MF and SF) and adiposity measures( Reference Ritchie 1 – Reference Murakami and Livingstone 20 ), none to date have attempted to investigate the effects on MF and SF separately. In the present study, MFtime, but not MFenergy%, SFenergy% and SFtime, was positively associated with BMI z score (but not WHtR) in adolescents, although this association reached statistical significance only when the analysis was limited to plausible EI reporters. This is plausible given that, although all measures of MF and SF were positively associated with EI (a potential causal factor), MFtime showed the strongest association with EI in adolescents. Having more eating occasions with the time periods 06.00–10.00, 12.00–15.00 and 18.00–21.00 hours may be specifically detrimental to BMI z score, although the results should be interpreted cautiously not only because the association did not reach statistical significance when the entire adolescent population was examined but also because of the null association for WHtR. Although there were also positive, but weaker, associations of MF and SF with EI in children, all measures of MF and SF showed no association with BMI z score. Previous studies have shown that children are generally good energy compensators, although this ability declines with age( Reference Cecil, Palmer and Wrieden 46 ), which might explain the positive association in adolescents reporting plausible EI but null association in children we observed. For dietary intake, we found that SFenergy%, SFtime and MFtime were generally associated with unfavourable dietary intake patterns in both children and adolescents, whereas MFenergy% showed no such associations. A previous US study showed that SF calculated on the basis of self-report, but not MF, was positively associated with diet quality (as assessed by the Healthy Eating Index) in children (aged 9–11 years)( Reference Evans, Jacques and Dallal 23 ). Conversely, in adolescents (aged 12–15 years) SF was inversely associated with diet quality, whereas MF showed a positive association( Reference Evans, Jacques and Dallal 23 ). These discrepant findings may be, at least partly, explained by differences in the characteristics and lifestyles of the populations, definitions of MF and SF, dietary assessment methods and potential confounding factors considered.
The advantages of the present study include the use of objective and published definitions of MF and SF based on detailed dietary information obtained from a 7-d weighed dietary record, measured anthropometric data and the use of individualised measure of EER to identify EI misreporters. However, there are also several limitations. First, the cross-sectional nature of the study does not permit the assessment of causality owing to the uncertain temporality of the association. Only a prospective study would provide better understanding of the relation between MF and SF and metabolic risk factors.
We used BMI and WHtR as proxy measures of body fatness. As BMI reflects not only body fatness but also the relative length of the legs, body frame size and fat-free body mass( Reference Prentice and Jebb 47 ), subjects with similar BMI (z score) do not necessarily have the same amount of body fat. A more valid measure of body fat mass (e.g. dual-energy X-ray absorptiometry) may be needed for further investigation.
Another limitation of the present study is that only 61 % of the eligible sample was included in the present study, although the response rate was relatively high (80 %). The subjects included in the present analysis (n 1636) differed somewhat from those excluded from the analysis (n 491). The excluded subjects were more likely to be younger and be of lower socio-economic status (all P<0·05). However, a previous analysis concluded that there was no evidence to suggest serious non-response bias in NDNS( Reference Gregory and Lowe 27 ). Additionally, although we adjusted for a variety of potential confounding variables, residual confounding could not be ruled out. In particular, adjustment for physical activity may be insufficient in the analysis of children as all subjects aged ≤6 years were categorised into the same category because of a lack of information. Also, we could not control for puberty status or parental weight status because of a lack of information, which may cause potential confounding by unknown or unmeasured factors.
Moreover, in the present study, an eating occasion was classified as any event that provided ≥210 kJ with a minimum time interval of >15 min between episodes. Although this definition has been used in several previous studies( Reference Murakami and Livingstone 20 , Reference Hartline-Grafton, Rose and Johnson 34 – Reference Ruidavets, Bongard and Bataille 38 ), some arbitrary decision (i.e. energy content and time interval) is inevitable by nature. However, there is currently no consensus about what constitutes an eating occasion, a meal or a snack. In this regard, we could not conduct the present analysis based on self-identification of eating occasions, the most common definition of meals and snacks, because of a lack of information in NDNS. Additionally, MF and SF based on time may be problematic, because eating patterns vary according to lifestyle (e.g. shift workers, individuals who consistently eat their meals at non-traditional times of day) as well as the cultural environment( Reference Johnson and Anderson 25 ). Furthermore, MF and SF based on energy contribution (≥15 or <15 %) was made on the basis of the US national averages of the distribution of energy from (self-defined) meals compared with (self-defined) snacks (breakfast: 16 %; lunch: 25 %; dinner: 37 %; and snack: 22 % from two occasions)( Reference Kant and Graubard 48 ), but this may not be suitable in the present young British population. Thus, results may possibly differ on the basis of other definitions. In any case, as research explicitly examining the impact of these different definitions is limited, it is currently difficult to decide which definition might be most appropriate for meals and snacks, and as, to our knowledge, this is the only study to examine the impact of different definitions of meals and snacks on the associations between MF and SF and diet and adiposity, similar research using different definitions of meals and snacks needs to be accumulated before reaching a consensus on what defines meals and snacks.
Finally, we assessed misreporting of EI against calculated EER with the use of published equations( 41 ). In the absence of measured total energy expenditure, these equations with high R 2 values (≥0·95)( 41 ) should serve as the best proxy. Nevertheless, the selection of physical activity category was based on self-report (i.e. 7-d physical activity diary) in subjects aged ≥7 years and fixed in subjects aged ≤6 years, which may be susceptible to systematic error. Additionally, we do not know the sensitivity and specificity of the procedure for identifying EI misreporters used. However, even though some misclassification of subjects according to EI reporting status did occur in this study, we are confident of our conclusions, because the associations of MF and SF with dietary intakes and adiposity measures observed in the entire populations were similarly observed in acceptable reporters. Nonetheless, it should be stressed that the role of misreporting was mainly evaluated only in terms of under-reporting because over-reporting occurred in such a low number of cases that no conclusions could be drawn in this regard.
In conclusion, the present cross-sectional study in Britain demonstrated that MF and SF was differentially associated with dietary intake when meals and snacks were defined on the basis of the contribution to total EI but not on the basis of time. Although all measures of MF and SF were associated with higher EI in children and adolescents (except for MFenergy% in children), SFenergy% and SFtime were associated with unfavourable dietary intake patterns, including higher intakes of soft drinks, confectionery and total sugar, lower intakes of cereals, fish, meat, protein, PUFA, starch and dietary fibre, and a lower MDS (except for SFenergy% in adolescents). MFtime, but not MFenergy%, was associated with adverse dietary profiles such as higher intakes of confectionery and sugar, lower intakes of fish, protein, PUFA and starch, and a lower MDS (only in children). Taken together, decreasing the number of small eating occasions (<15 % of total EI) regardless of the time of day may be important to improve diet quality. However, all measures of MF and SF showed no association with adiposity measures in both children and adolescents. These findings were not influenced by misreporting of EI, as similar associations were observed not only in the entire population but also in subjects with plausible EI. Further research, particularly with a prospective design, is needed so that firm conclusions can be drawn with regard to the effect of MF and SF on dietary intakes and adiposity measures.
Acknowledgements
The authors would like to thank the anonymous reviewers for their valuable comments and suggestions to improve the quality of the manuscript.
This work was supported in part by the Grants-in-Aid for Young Scientists (B) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (K. M., grant number 15K16213). The Ministry of Education, Culture, Sports, Science and Technology of Japan had no role in the design, analysis or writing of this article.
K. M. contributed to the concept and design of the study, statistical analysis, data interpretation and manuscript writing. M. B. E. L. critically reviewed the manuscript. All authors read and approved the final manuscript.
The authors declare that there are no conflicts of interest.








