Cognitive impairment has been associated with significant deficits in social and occupational functioning in bipolar disorder. Reference Martinez-Aran, Vieta, Colom, Torrent, Sanchez-Moreno and Reinares1,Reference Zubieta, Huguelet, O'Neil and Giordani2 A recent meta-analysis indicated that euthymic patients with bipolar disorder when compared with healthy controls showed deficits in executive function, verbal and nonverbal episodic memory as well as in sustained attention. Reference Torres, Boudreau and Yatham3 However, specific aspects of cognitive function such as emotional memory have not been examined in people with bipolar disorder. There is evidence that memory for emotionally charged events may require specific neuro-anatomical circuitries that include the amygdala. Previous studies have shown increased activity of amygdala in people with bipolar disorder. Given this, individuals with bipolar disorder would be expected to have enhanced perception of emotional stimuli and, as a consequence, enhanced memory for emotional content. Therefore, we investigated emotional memory using a well-validated amygdala-related cognitive task. Reference Cahill and McGaugh4
Method
Twenty consecutive consenting stable euthymic patients with bipolar I or II disorder who met DSM–IV full remission criteria were recruited from the Bipolar Disorders Program of the University Hospital at the Federal University, Porto Alegre, Brazil. Participants were interviewed with the Structured Clinical Interview for DSM–IV Axis I Disorders (SCID). 5 Participants were referred by their psychiatrist when euthymic for at least a month, and remission was confirmed by clinical interview and life charting. Those using regular β-blockers, benzodiazepines or stimulants were excluded. Occasional use of benzodiazepines was allowed (maximum 4 mg lorazepam per week or equivalent), but all participants were asked to stop taking these medications at least 24 h before the memory task was performed. None of the participants had significant concomitant medical illnesses or a history of substance misuse. The participants were matched for gender, age and schooling years with 20 healthy controls. Healthy controls were screened using the SCID (non-patient version). Those with current psychiatric morbidity or lifetime mood disorders were excluded. We also excluded people who were using psychotropic medication, those with a previous history of psychological/psychiatric treatments, family history of mental illness, drug misuse and major health problems.
We used the Heuer & Heisenberg test modified by Cahill & McGaugh Reference Cahill and McGaugh4 and others Reference Cahill, Prins, Weber and McGaugh6,Reference Quevedo, Sant'Anna, Madruga, Lovato, de-Paris and Kapczinski8 to assess emotional memory. Participants with bipolar disorder were randomly assigned to be exposed to a neutral story (n=10) or a closely matched but more emotionally arousing story (n=10). The controls were assigned to the same version of the story as their matched patients. This assignment resulted in four groups: patient neutral or patient emotional, control neutral or control emotional. All procedures for the memory test were the same as previously described. Reference Cahill and McGaugh4,Reference Cahill, Prins, Weber and McGaugh6,Reference Kazui, Mori, Hashimoto, Hirono, Imamura, Tanimukai, Hanihara and Cahill7 All participants were given an individual explanation about study objectives and informed consent was obtained. The University Hospital Ethics Committee approved the protocol. The text with the explanation about the study was the same as that used by Cahill & McGaugh Reference Cahill and McGaugh4 and others Reference Cahill, Prins, Weber and McGaugh6,Reference Quevedo, Sant'Anna, Madruga, Lovato, de-Paris and Kapczinski8 and it was read to individuals in order to ensure that all participants were informed in the same manner. Participants were told that the story they would watch might be emotionally arousing and that they would be recalled for another assessment a week later.
The stories were presented individually as a narrated slide show (comprising 11 slides, lasting a total of about 10 min), that could be neutral or emotional. Both stories were separable into three phases: the first phase including slides 1–4, the second phase including slides 5–8, and the final phase including slides 9–11. The emotional and neutral stories differed primarily in slides 5–8, when the emotional elements were introduced in the emotional story, which will be referred to from here on as phase 2. In the neutral version, no emotional element was introduced; the content was neutral across the three phases. Because the visual elements used in both stories were identical, differences in retention cannot be attributed to intrinsic differences in the visual elements. In both stories, a mother takes her young son to visit his father at the hospital where he works. In the neutral version, the son watches the staff conduct a practice disaster drill. In the emotional version, the boy is severely hurt in a car accident.
Immediately after viewing the slide show, each participant was asked to rate on a 0–10 scale how emotional they thought the story was. One week later participants were assessed on a memory-recall test. The testing session consisted of a questionnaire containing 76 multiple choice questions. The questionnaire consisted of 5–8 questions for each slide, and these were presented in the same order as the story. The questions were presented only once and the participant was asked to choose one answer and then go on to the next one.
Statistics
Statistical analyses were performed using the Statistical Package for Social Sciences version 13.0 for Windows (SPSS Inc., Chicago, Illinois, USA). Data for the self-rating emotional scale and for the multiple choice questions (percentage of correct answers) are shown as a mean (s.d.). A multivariate analysis of variance (MANOVA) with repeated measures was used to analyse the dependent variable: the percentage of correct answers in the three story phases from the multiple choice question test (memory recall). The MANOVA with repeated measures included an effect for group (bipolar disorder×control), an effect for condition (neutral ×emotional) and an effect for phase (three phases). We also examined an interaction of phase×condition as well as an interaction of phase×group×condition. A post hoc ANOVA was carried out to examine differences within groups (neutral and emotional) in the percentage of correct answers between phases of the narrated story followed by post hoc Tukey test as appropriate. The differences in self-rating emotional scale scores between the groups in various phases were analysed using ANOVA and post hoc Tukey tests. Continuous variables were tested using independent sample t-tests, as indicated. Dichotomous demographic variables were tested using chi-square tests. All tests were two-tailed with an α of 0.05 and P<0.05 was considered to indicate statistical significance.
Results
At the evaluation, all patients were taking mood stabilisers (lithium, n=3; valproate, n=7; carbamazepine, n=1; lithium and carbamazepine, n=3; lithium and valproate, n=5; valproate and carbamazepine, n=1). Half of the patients were taking antipsychotics (risperidone, n=4; haloperidol, n=2; olanzapine, n=1; chlorpromazine, n=1; sulpiride, n=1; clozapine, n=1). Other medications used included lamotrigine (n=1), biperiden (n=1) and antidepressants (i.e. sertraline, citalopram and fluoxetine; n=3). There was no significant difference between the emotional and neutral patient groups regarding number of mood stabilisers used (one mood stabiliser was used by 7 v. 4 patients and two mood stabilisers by 3 v. 6 patients respectively; P=0.37). There were 5 patients in the emotional group and 5 patients in the neutral group that used antipsychotics. Antidepressants were used by 1 patient in the emotional group and 2 in the neutral group. As to the potential effects of medications, there was no difference between various medication groups (i.e. those on lithium, divalproex or lithium plus divalproex) regarding self-rating scores (F=2.2, d.f.=2,16, P=0.14), percentage of correct answers in phase 1 (F=2.1, d.f.=2,16, P=0.15), percentage of correct answers in phase 2 (F=0.06, d.f.=2,16, P=0.93), percentage of correct answers in phase 3 (F=0.03, d.f.=2,16, P=0.99) and overall percentage of correct answers (F=0.32, d.f.=2,16, P=0.72). Similarly, there was no difference on any of these measures between those on antipsychotics or antidepressants and those who were not on these medications.
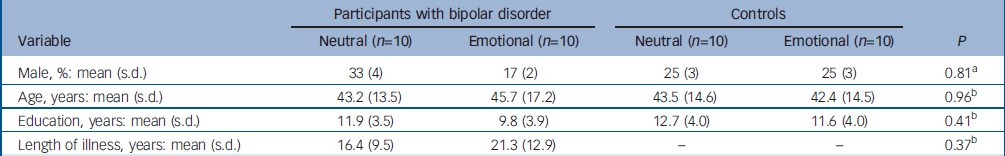
Demographic and clinical characteristics are shown in Table 1. Of the patients, 14 were female and 6 were male; their age ranged from 19 to 66 years (mean=44.5) and their mean educational level was 10.8 years. The mean length of illness of the patients was 18.8 years. They were closely matched by gender, age and schooling years to 20 healthy controls whose age ranged from 18 to 65 years (mean=42.9) and whose mean educational level was 12.1 years.
Table 1 Socio-demographic and clinical characteristics of participants in the neutral stimulus and emotional stimulus groups
| Participants with bipolar disorder | Controls | ||||||
|---|---|---|---|---|---|---|---|
| Variable | Neutral (n=10) | Emotional (n=10) | Neutral (n=10) | Emotional (n=10) | P | ||
| Male, %: mean (s.d.) | 33 (4) | 17 (2) | 25 (3) | 25 (3) | 0.81 a | ||
| Age, years: mean (s.d.) | 43.2 (13.5) | 45.7 (17.2) | 43.5 (14.6) | 42.4 (14.5) | 0.96 b | ||
| Education, years: mean (s.d.) | 11.9 (3.5) | 9.8 (3.9) | 12.7 (4.0) | 11.6 (4.0) | 0.41 b | ||
| Length of illness, years: mean (s.d.) | 16.4 (9.5) | 21.3 (12.9) | – | – | 0.37 b | ||
a. χ2-test
b. ANOVA
As expected, the overall percentage of correct answers was lower in the patients than in the matched healthy controls (t=3.3, d.f.=38, P=0.002) (Fig.1). The MANOVA with repeated measures showed an effect of phase (F=13.5, d.f.=2,72, P<0.001), effect of group (i.e. control v. bipolar disorder) (F=14.7, d.f.=1,36, P<0.001), an interaction of a phase×condition (F=4.1, d.f.=2,72, P=0.02) and an interaction of phase× group×condition (F=2.4, d.f.=6,72, P=0.03). When compared directly with controls, patients performed poorly regarding the enhancement of memory for emotional events, as indicated by scores in phase 2 of the emotional version of the story (F=5.2, d.f.=3,36, P=0.01). In addition, there was a significant difference in perception of the emotional impact of the stories between groups as indicated by significant differences in emotional impact self-rating scale scores (F=5.6, d.f.=3,36, P=0.003).
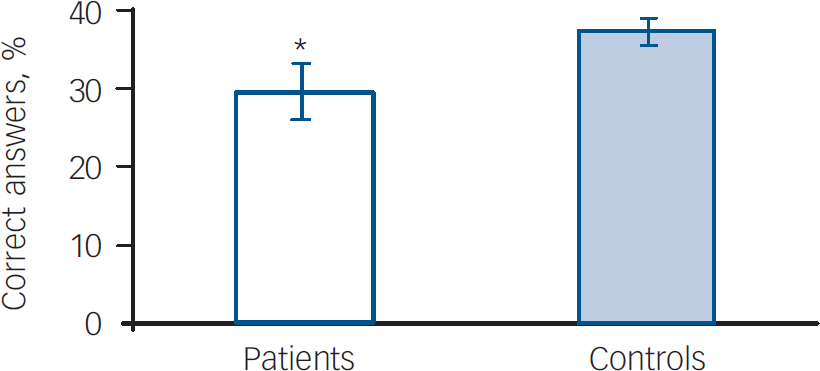
Fig. 1. Difference in overall recall performance (percentage of correct answers between patients (n=20) and healthy controls (n=20)). *Significant difference between patients and controls (t=3.3, d.f.=38, P=0.002).
The results for the controls are presented in Fig. 2. Consistent with previous studies that have used this task in healthy volunteers, Reference Cahill, Prins, Weber and McGaugh6,Reference Quevedo, Sant'Anna, Madruga, Lovato, de-Paris and Kapczinski8 the percentage of correct answers was enhanced in phase 2 of the emotional version when compared with phases 1 and 3 (F=5.7, d.f.=2,27, P=0.008). In the neutral version, there were no differences in percentage of correct answers across phases within groups (F=1.0, d.f.=2,27, P=0.35). There was a significant difference in the percentage of correct answers between the emotional and neutral groups in phase 2, the emotionally arousing phase (t=2.1, d.f.=18, P=0.04). The emotional arousal content was associated with an increase in recall as indicated by the higher percentage of correct answers. Self-rating for emotional arousal demonstrated a greater impact in the emotional version, as expected (F=5.6, d.f.=3,36, P=0.007). There was no difference between controls exposed to the emotional stimuli and those exposed to neutral stimuli regarding age (mean 42.4 v.43.5 years; P=0.86) and schooling years (mean 11.6 v. 12.7 years; P=0.98). There were more females in the group exposed to the emotional version (8 females, 2 males) compared with the group exposed to the neutral experience (6 females, 4 males), but the difference was not statistically significant (P=0.62).
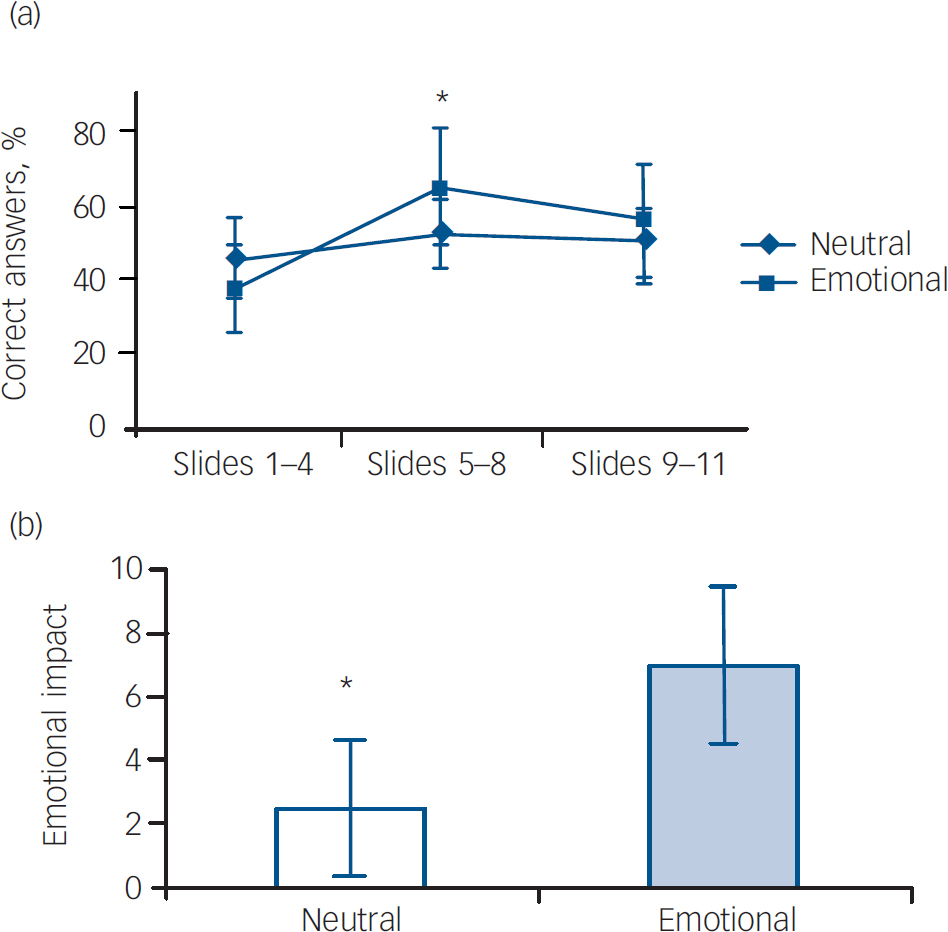
Fig. 2 (a) Recall performance of controls exposed to the emotional (n=10) and neutral (n=10) condition; (b) emotional impact of the neutral and emotional version of the story for controls (self-rating emotional scale scores). (a) *Significant difference in percentage of correct answers across phases in the emotional condition (P=0.008), but not in the neutral condition (P=0.35). (b) *Significant difference in the subjective emotional impact of the story between neutral and emotional version (P=0.007).
Results for patients exposed to the neutral and emotional stimuli are shown in Fig.3. There was no difference in the percentage of correct answers between phases in the neutral group (F=2.5, d.f.=2,27, P=0.1), as expected. Surprisingly, there was no difference between phases in the emotional group as well (F=1.3, d.f.=2,27, P=0.27). Furthermore, there was no difference in the percentage of correct answers between the emotional and neutral groups in phase 2, the emotionally arousing part (t=0.5, d.f.=18, P=0.57). The emotional arousal content was not associated with an increase in recall as reflected by the percentage of correct answers. In addition, the expected subjective emotional impact was not observed, as demonstrated by the absence of a significant difference in self-rating scale scores between the neutral and emotional groups (P=0.23) (Fig. 2(b)). Instead, participants with bipolar disorder were more likely than controls to rate the neutral stimuli as of greater emotional impact (mean=4.5 (s.d.=3.5) v. mean=2.5 (s.d.=2.5) respectively). There was no difference between the patients exposed to the emotional stimuli and those exposed to neutral stimuli regarding length of illness (mean 21.3 v. 16.4 years; P=0.37), age (mean 45.7 v. 43.2 years; P=0.72) and schooling years (mean 9.8 v. 11.9 years; P=0.22). Similar to the controls, there were more females in the emotional patient group.
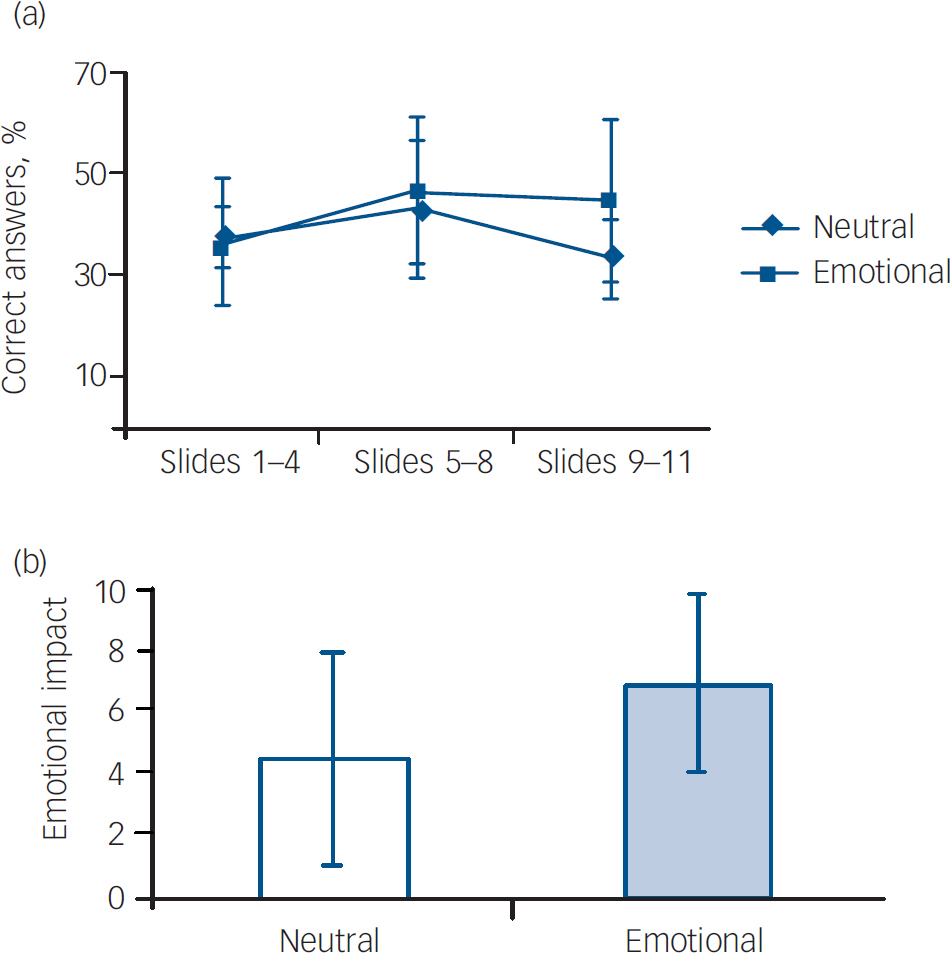
Fig. 3 (a) Recall performance of patients with bipolar disorder exposed to the emotional (n=10) and neutral (n=10) condition; (b) emotional impact of the neutral and emotional version of the story for patients (self-rating emotional scale scores). (a) No significant difference in percentage of correct answers across phases in both the neutral (P=0.1) and the emotional condition (P=0.27). (b) No significant difference in the subjective emotional impact of the story between neutral and emotional version (P=0.23).
Discussion
This is the first study to test the impact of emotion on memory formation in people with bipolar disorder in comparison with controls. As expected, controls showed a clear enhancement in memory for the emotional content of the story compared with the neutral content. Controls also perceived accurately the emotional impact of the stories, as demonstrated by difference in self-rating scores between the neutral and emotional versions.
As expected, the overall recall rate in participants with bipolar disorder was significantly lower compared with controls. However, in contrast to the findings in healthy controls and our hypothesis, our results showed that the physiological pattern of enhanced memory retrieval for emotionally bound information was blunted in patients with bipolar disorder. These findings were obtained from patients with bipolar disorder on medication, which is a potential source of bias. Drugs used to treat bipolar disorder may hamper different cognitive systems, and may impair emotional memory as well. However, it is unlikely that a non-specific flattening of cognitive function as a whole would account for the higher rates of mislabelling of neutral information as emotional, which was found in participants with bipolar disorder. Further, people with bipolar disorder perceived the neutral story as more emotionally charged, as indicated by no difference in scores on the visual analogue self-report scale between the neutral and the emotional versions. Since patients with bipolar disorder perceived the neutral content as more emotional and similar to the emotional version, one would have expected an enhancement of memory in phase 2 in both versions. However, the recall rate was similar in all phases in both versions for these individuals. This would suggest that the enhanced perception of emotional impact did not translate into enhancement of memory formation.
Amygdala-dependent memory task
When interpreting the results, the first consideration must be the neurocircuitry involved in this task. We used the Heuer & Reisburg task Reference Heuer and Reisberg9 modified by Cahill & McGaugh Reference Cahill and McGaugh4 and others, Reference Cahill, Prins, Weber and McGaugh6-Reference Quevedo, Sant'Anna, Madruga, Lovato, de-Paris and Kapczinski8 which compares memory for emotionally arousing v. neutral information. It is well-documented that the memory for an emotionally arousing event is better than for neutral stimuli. Reference Cahill10,Reference Cahill, Babinsky, Markowitsch and McGaugh11 There is evidence that the amygdala plays a crucial role in the enhancement of the strength of long-term memory for emotional events. Reference Hamann, Ely, Grafton and Kilts12 Further, previous studies that have used the modified Heuer & Reisburg test have shown that enhancement in memory associated with the emotional content is highly dependent on amygdala function. Reference Cahill, Prins, Weber and McGaugh6 For instance, memory enhancement induced by emotional arousal was absent:
-
(a) in healthy controls using β-blockers Reference Cahill, Prins, Weber and McGaugh6
-
(b) in a patient with a rare hereditary disorder that produces bilateral brain damage confined to the amygdaloid complex Reference Cahill, Babinsky, Markowitsch and McGaugh11
-
(c) in patients with bilateral amygdala damage who have had a temporal lobectomy Reference Brierley, Medford, Shaw and David13
-
(d) in short-term memory assessment. Reference Quevedo, Sant'Anna, Madruga, Lovato, de-Paris and Kapczinski8
These findings provide evidence that the amygdala might be a critical locus for emotional enhancement in memory in this task. Reference Cahill, Prins, Weber and McGaugh6,Reference Cahill, Babinsky, Markowitsch and McGaugh11 Furthermore, in healthy volunteers, the degree of activity in the left amygdala during encoding was predictive of subsequent memory and was related to the emotional intensity of the experience. Reference Canli, Zhao, Brewer, Gabrieli and Cahill14 Interestingly, patients with Alzheimer's disease with moderate overall memory impairment did perform well in this test, and the extent of memory enhancement for emotionally charged content was similar in those with Alzheimer's disease and in controls. Reference Kazui, Mori, Hashimoto, Hirono, Imamura, Tanimukai, Hanihara and Cahill7 Therefore, the poor performance of participants with bipolar disorder on this task in the present study suggests that patients with bipolar disorder have emotional memory deficits which may be related to a dysfunctional amygdala circuitry.
Amygdala circuitry in bipolar disorder
Our findings are consistent with previous literature suggesting abnormalities in amygdala circuitry in bipolar disorder. For instance, many of the symptoms experienced by patients with bipolar disorder would appear to be associated with abnormalities in emotion processing. Reference Phillips15,Reference Phillips, Drevets, Rauch and Lane16 Further, emotional hyper-reactivity is a fundamental mood characteristic of manic and mixed states. Reference Henry, Swendsen, Van den Bulke, Sorbara, Demotes-Mainard and Leboyer17 Moreover, structures known to take part in the emotional processing circuitry Reference Cahill and McGaugh4,Reference Hamann, Ely, Grafton and Kilts12 such as prefrontal cortex, subgenual anterior cingulate gyrus, the amygdala and ventral striatum have been reported to have structural and functional alterations in people with bipolar disorder. Reference Phillips, Drevets, Rauch and Lane18 Of these, the amygdala is critically involved in modulating emotional memory, attention and perception. Reference Phelps19 Interestingly, enlarged amygdala volumes have been reported in structural imaging studies in bipolar disorder. Reference Altshuler, Bartzokis, Grieder, Curran and Mintz20 Abnormal age-related increases in the amygdala volume have been found in adolescents with bipolar disorder. Reference Chen, Sassi, Axelson, Hatch, Sanches, Nicoletti, Brambilla, Keshavan, Ryan, Birmaher and Soares21 Also, magnetic resonance imaging studies have reported enlarged amygdala volumes in bipolar disorder. Reference DelBello, Zimmerman, Mills, Getz and Strakowski22 In addition to structural changes in this circuitry, functional neuroimaging studies indicate underactivity of the dorsal and ventral prefrontal cortex, and increased activity in the dorsal anterior cingulate, amygdala Reference Malhi, Lagopoulos, Ward, Kumari, Mitchell, Parker, Ivanovski and Sachdev23,Reference Malhi, Lagopoulos, Owen and Yatham24 and thalamus Reference Chen, Lennox, Jacob, Calder, Lupson, Bisbrown-Chippendale, Suckling and Bullmore25 during mood episodes. Increased metabolism within the right amygdala has been reported in people with bipolar disorder during episodes of depression. However, most of the functional imaging studies that investigated the activity of amygdala and temporal lobe in emotional processing in bipolar disorder used tests of facial expression recognition, but did not include emotional memory paradigms. The available data from these studies demonstrated that patients with bipolar disorder experiencing depressed or manic episodes identify facial expressions less accurately than do euthymic patients or healthy comparison individuals. Reference Chen, Lennox, Jacob, Calder, Lupson, Bisbrown-Chippendale, Suckling and Bullmore25 Another study showed that people with bipolar disorder in depressed or euthymic states present increased subcortical and ventral prefrontal cortical responses to both positive and negative emotional facial expressions compared with healthy controls and people with major depression. Reference Lawrence, Williams, Surguladze, Giampietro, Brammer, Andrew, Frangou, Ecker and Phillips26 In euthymic patients, enhanced disgust Reference Harmer, Grayson and Goodwin27 and impaired identification of fearful facial expressions have been demonstrated. Reference Yurgelun-Todd, Gruber, Kanayama, Killgore, Baird and Young28 The latter study also reported increased amygdala and reduced prefrontal cortical activation in response to facial expressions of fear.
Interestingly, the previously observed increased amygdala volume and increased activity of temporal lobe and amygdala during facial recognition tasks in euthymic individuals with bipolar disorder Reference DelBello, Zimmerman, Mills, Getz and Strakowski22,Reference Chen, Lennox, Jacob, Calder, Lupson, Bisbrown-Chippendale, Suckling and Bullmore25,Reference Lawrence, Williams, Surguladze, Giampietro, Brammer, Andrew, Frangou, Ecker and Phillips26,Reference Hamann and Adolphs29 did not translate into increased emotional memory formation in our study, as we expected. This is consistent with previous evidence that indicates that facial expression recognition and emotional memory formation require different neuroanatomical pathways. Reference Brierley, Medford, Shaw and David13,Reference Hamann, Stefanacci, Squire, Adolphs, Tranel, Damasio and Damasio30 For instance, a study of patients with amygdala damage due to herpes simplex encephalitis suggested that recognition of facial emotion in adults does not have an absolute dependence on the amygdala. Reference Hamann, Stefanacci, Squire, Adolphs, Tranel, Damasio and Damasio30 Further, patients with bilateral amygdala damage who have had a temporal lobectomy had superior fear face perception but their ability to form enhanced emotional memories in the Heuer & Reisburg test Reference Brierley, Medford, Shaw and David13 was severely compromised. Taken together, it would appear that the enlarged volumes and enhanced activity of the amygdala in bipolar disorder indicate an oversensitive but dysfunctional neural system for emotional processing.
Perception of emotional stimuli
Emotion, through the amygdala, can influence encoding, attention and perception. Reference Phelps19 Although the memory test used here assessed the memory formation process as a whole, we further investigated the perception of the emotional content of the stories, as indicated by self-rating scores. Participants with bipolar disorder reacted differently than controls to the emotional impact of the stories. There was no difference between the scores for the emotional and for the neutral stimuli in self-report of emotional impact among patients, whereas for controls it was clearly different. This may suggest that people with bipolar disorder may have an oversensitive emotional reaction to facts, which is not functional because it can be restrictive for the ability to focus on the real emotional content. The enhanced emotional memory is clearly adaptive, because emotional stimuli are generally more important for survival. Reference Hamann, Ely, Grafton and Kilts12 Previous studies have reported that people with bipolar disorder are less able to accurately recognise emotions in human faces than healthy controls, which was thought to be associated with their impaired social skills. Reference Chen, Lennox, Jacob, Calder, Lupson, Bisbrown-Chippendale, Suckling and Bullmore25 Our results showed that this altered perception in people with bipolar disorder might not be restricted to facial expressions as their perception for facts in a simple story was also altered. One can speculate that this altered perception may lead individuals to remember neutral stimuli as emotional, thus making them more susceptible to interpreting life events as traumatic. This is consistent with the literature showing that stressful life events are associated with mood episodes.
Limitations
Some limitations must be considered when interpreting this study. First, many factors have been reported to influence cognitive performance of people with bipolar disorder, for example number of illness episodes, number of hospitalisations and age at onset, but studies also reported absence of such associations. Reference Robinson and Ferrier31 In order to minimise the impact of these confounding factors, we included length of illness in the assessment to ensure that groups were not different in relation to illness variables. The length of illness was chosen because it has been reported to be associated with visuospatial and verbal memory impairments. Reference Robinson, Thompson, Gallagher, Goswami, Young, Ferrier and Moore32 Second, these findings were obtained in participants with bipolar disorder on medication, which can be a potential source of bias through the direct effect of medications either on perception of emotional events or on cognition. The former is unlikely because participants in this study perceived even neutral events as more emotionally charged regardless of type of medication they had been receiving, which suggests that medications did not cause flattening of perception of emotional events. It is also unlikely that cognitive deficits observed in various domains in people with bipolar disorder in this study are related to medications because previous studies suggest that medication-free euthymic individuals with bipolar disorder also have those cognitive deficits. Reference Robinson and Ferrier31 Furthermore, the only study to date that assessed the effects of a psychotropic medication sulpiride in ‘normal’ human volunteers showed that it did not significantly impair the enhancement of emotional memory. Reference Mehta, Hinton, Montgomery, Bantik and Grasby33 Thus, it is unlikely that the medications account for impairment of enhancement of emotional memory in our study. This, however, needs confirmation in future studies of people with bipolar disorder who are not on medication. Third, euthymic individuals with bipolar disorder often present subsyndromal affective symptoms, which may affect performance on cognitive measures, specifically emotional memory. We did not use scales for mood symptoms, so it was not possible to exclude the presence of this confounding factor. However, previous studies were able to demonstrate that cognitive impairment persisted in euthymic individuals with bipolar disorder after controlling for mood symptomatology. Reference Robinson, Thompson, Gallagher, Goswami, Young, Ferrier and Moore32 Fourth, there was no direct measure of attention in this study, and sustained attention deficits have been reported in euthymic individuals with bipolar disorder. However, the task consisted of a short presentation of slides and the test was administered by the investigator. Also, the self-rating scores for emotional impact suggest that patients were sensitive to the content of the story.
Acknowledgements
This work was supported by the Fundo de Incentivo a pesquisa e eventos do Hospital de Clinicas de Porto Alegre (FIPE–HCPA) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Conselho Nacional de Desenvolvimento Científico e Tecnológico (CAPES–CNPq). M.K.S.A, A.C.A., K.C., F.K.G., J.Q., I.I. and F.K. were supported by CAPES–CNPq, Brazil. L.N.Y. has been a speaker, member of advisory board and received grant funding from Lilly, Janssen, AstraZeneca, GlaxoSmithKline, Bristol Myers Squibb, Novartis and Servier.







eLetters
No eLetters have been published for this article.