Introduction
Many organisms seek a sheltered domicile or safety beneath the substratum surface as protection against harsh environments or predators, often in combination with the search for nourishment. The increased penetration of the sea floor by burrowing invertebrates is well documented during the early Phanerozoic by the trace fossil record (Crimes, Reference Crimes1992; Jensen, Reference Jensen2003; Mángano and Buatois, Reference Mángano, Buatois, Mángano and Buatois2016, Reference Mángano and Buatois2017, Reference Mángano and Buatois2020). In Greenland, as in many other areas, deep pipes of Skolithos Haldeman, Reference Haldeman1840 and other vertical burrows are a characteristic feature of many transgressive lower Cambrian sequences (Cowie and Spencer, Reference Cowie and Spencer1970; Pickerill and Peel, Reference Pickerill and Peel1990; Jensen et al., Reference Jensen, Harper and Stouge2015). The early appearance in the geological record of various groups of worms that might be responsible for the burrows is documented in fossil Lagerstätte, e.g., the Sirius Passet of North Greenland, the Chengjiang of China, and the Burgess Shale of Canada (Conway Morris, Reference Conway Morris1977; Huang et al., Reference Huang, Chen, Vannier and Salinas2004; Conway Morris and Peel, Reference Conway Morris and Peel2008; Caron et al., Reference Caron, Gaines, Mángano, Streng and Daley2010; Vinther et al., Reference Vinther, Eibye-Jacobsen and Harper2011; Eibye-Jacobsen and Vinther, Reference Eibye-Jacobsen and Vinther2012; Hou et al., Reference Hou, Steiner, Zhu, Luo, Forchielli, Keupp, Zhao and Liu2012; Parry et al., Reference Parry, Tanner and Vinther2014; Peel, Reference Peel2017a). Macrofossil groups, e.g., helcionelloid molluscs, rostroconchs, and the emerging bivalves, show morphological traits indicative of an infaunal lifestyle already in the Cambrian (Pojeta and Runnegar, Reference Pojeta and Runnegar1976; Gubanov et al., Reference Gubanov, Kouchinsky and Peel1999; Vendrasco et al., Reference Vendrasco, Kouchinsky, Porter and Fernandez2011; Peel, Reference Peel2021). Boring by invertebrates into middle Cambrian (Miaolingian) hardground surfaces in North Greenland was also described by Peel (Reference Peel2017b).
Microorganisms have a significantly older record of penetration of the substratum. In the present day, endoliths survive in hyperarid and dry Antarctic terrestrial environments that are largely incapable of supporting other life forms (Friedmann, Reference Friedmann1982; Crits-Christoph et al., Reference Crits-Christoph, Robinson, Ma, Ravel, Wierzchos, Ascaso, Artieda, Souza-Egipsy, Casero and DiRuggiero2016; Coleine et al., Reference Coleine, Stajich, Zucconi, Onofri, Pombubpa, Egidi, Franks, Buzzini and Selbmann2018; Stan-Lotter, Reference Stan-Lotter, Seckbach and Rampelotto2019), in a scenario that likely reflects the early history of life on Earth (Homann et al., Reference Homann, Sansjofre, Van Zuilen, Heubeck, Gong, Killingsworth, Foster, Airo, Van Kranendonk, Ader and Lalonde2018). Cyanobacteria have a geological record extending back > 2.5 billion years (Knoll, Reference Knoll2015; Schirrmesiter et al., Reference Schirrmeister, Sanchez-Baracaldo and Wacey2016) and descriptions of boring cyanobacteria (euendoliths) or similar organisms extend back to the Paleoproterozoic (Campbell, Reference Campbell1982; Zhang and Golubic, Reference Zhang and Golubic1987; Zhang, Reference Zhang1988). In Greenland, late Proterozoic ooids are penetrated by Eohyella Zhang and Golubic, Reference Zhang and Golubic1987, a morphological counterpart of the Recent Hyella Bornet and Flahault, Reference Bornet and Flahault1888 (Green et al., Reference Green, Knoll and Swett1988; Golubic et al., Reference Golubic, Campbell, Lee and Radtke2016). Cambrian (Miaolingian Series) occurrences were documented by Stockfors and Peel (Reference Stockfors and Peel2005a).
Fungus-like, filamentous microfossils were described by Bengtson et al. (Reference Bengtson, Rasmussen, Ivarsson, Muhling, Broman, Marone, Stampanoni and Bekker2017a) and Ivarsson et al. (Reference Ivarsson, Drake, Neubeck, Sallstedt, Bengtson, Roberts and Rasmussen2020) from vesicles and fractures in strata from the early Paleoproterozoic (2.4 Ga) and from the Ediacaran of China (Gan et al., Reference Gan, Luo, Pang, Zhou, Zhou, Wan, Li, Yi, Czaja and Xiao2021), although the microfossils themselves might have a different age. Riding (Reference Riding2006) noted that silicified cyanobacteria can be common in the Proterozoic. Calcified forms first appeared in the Neoproterozoic, but only became widespread at the transition to the Phanerozoic. In Greenland, cavity-dwelling filamentous organisms were described from the Cambrian (Miaolingian) by Stockfors and Peel (Reference Stockfors and Peel2005b). Ineson and Peel (Reference Ineson and Peel1997, fig. 12C) illustrated pendant and arborescent microbial structures in spar-filled cavities in early Cambrian (Stage 4) thrombolites of the Bistrup Land Formation similar to occurrences described by Kobluk and James (Reference Kobluk and James1979) and Kobluk (Reference Kobluk1981a, Reference Koblukb) from North America.
Peel (Reference Peel1988a) recognized diagenetically mineralized trichomes (threads composed of naked cells) and filaments (trichomes covered by an organic sheath) in specimens from the Portfjeld Formation in North Greenland that were interpreted as cyanobacteria. However, fungi and various algal groups develop similar threads and also have long geological records (Golubic et al., Reference Golubic, Campbell, Lee and Radtke2016; Bengtson et al., Reference Bengtson, Rasmussen, Ivarsson, Muhling, Broman, Marone, Stampanoni and Bekker2017a, Reference Bengtson, Sallstedt, Belivanova and Whitehouseb). Mineralization in present-day living cyanobacteria is often characteristic of freshwater forms, in which precipitation of calcium carbonate occurs by impregnation of the sheath or external encrustation of the filaments (Pentecost and Riding, Reference Pentecost, Riding, Leadbetter and Riding1986). However, Chafetz and Buczynski (Reference Chafetz and Buczynski1992) noted that bacteria in microbial mats promoted calcification of dead cyanobacteria just a few millimeters below the surface. Schroeder (Reference Schroeder1972) demonstrated diagenetic mineralization of green algal threads in cavities within marine carbonate rocks, providing a modern analogue to the present descriptions. In the fossil record, mineralization of cyanobacterial sheaths is conspicuous in Cambrian to Cretaceous strata (Wray, Reference Wray1977; Riding and Voronova, Reference Riding, Voronova, Toomey and Nitecki1985; Pentecost and Riding, Reference Pentecost, Riding, Leadbetter and Riding1986), although cyanobacteria have a much longer record (Hofmann, Reference Hofmann1976; Demoulin et al., Reference Demoulin, Lara, Cornet, François, Baurain, Wilmotte and Javaux2019).
This paper describes threads preserved by early diagenetic mineralization in cavities within Ediacaran to middle Cambrian (Miaolingian Series, Guzhangian Stage) marine dolostones and limestones from North Greenland (Fig. 1). The delicate phosphatized threads and meshworks formed in cavities within the remains of other organisms and were revealed by digestion of carbonate samples in weak acetic or formic acid (Figs. 2–5). The precise nature and thickness of the original threads is obscured by the subsequent diagenetic mineral coating, but the original threads were much narrower than the mineralized threads. Likewise, the origin of the various host cavities (crypts) is open to discussion, with crypts being formed within organisms lying on the sea floor and potentially also within hard substrata. In the present day, a variety of microorganisms, ranging from phototrophic algae and cyanobacteria to fungi and bacteria, which are known from coastal waters to the deepsea floor (Golubic et al., Reference Golubic, Campbell, Lee and Radtke2016; Stal and Cretoiu, Reference Stal and Cretoiu2016), can be present in cavity communities and were involved in their preservation (Raff et al., Reference Raff, Schollaert, Nelson, Donoghue, Thomas, Turner, Stein, Dong, Bengtson, Huldtgren, Stampanoni, Chongyu and Raff2008; Gaspard, Reference Gaspard2011; Meslier and DiRuggiero, Reference Meslier, DiRuggiero, Seckbach and Rampelotto2019; Pernice et al., Reference Pernice, Raina, Rädecker, Cárdenas, Pogoreutz and Voolstra2020).
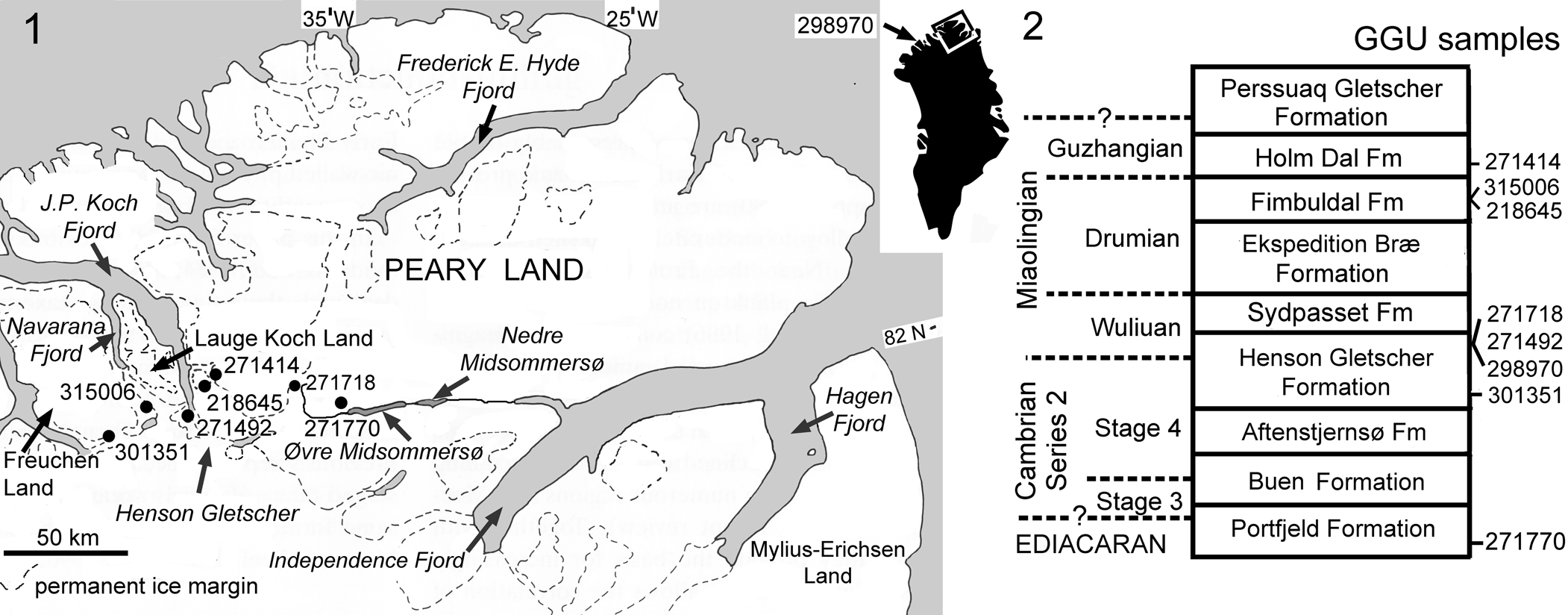
Figure 1. (1) Location of fossiliferous GGU samples. GGU sample 298970 (inset map) was collected to the west, in northern Nyeboe Land (Robison, Reference Robison1994, fig. 1). (2) Stratigraphic distribution of fossiliferous GGU samples. The Aftenstjernesø, Henson Gletscher, Sydpasset, and Ekspedition Bræ formations make up the Brønlund Fjord Group, whereas the Fimbuldal, Holm Dal, and Perssuaq Gletscher formations are assigned to the Tavsens Iskappe Group (Ineson and Peel, Reference Ineson and Peel1997).

Figure 2. Mineralized threads (SEM) in Ediacaran and Cambrian spheres: (1–3, 6) Portfjeld Formation from GGU sample 271770: (1) PMU 36874/4; (2, 6) PMU 36879/2; (3) PMU 36866/4; (4, 5) Henson Gletscher Formation, PMU 38167, from GGU sample 301351. Scale bars = 100 μm (4), 50 μm (3), 20 μm (1, 2, 5, 6).
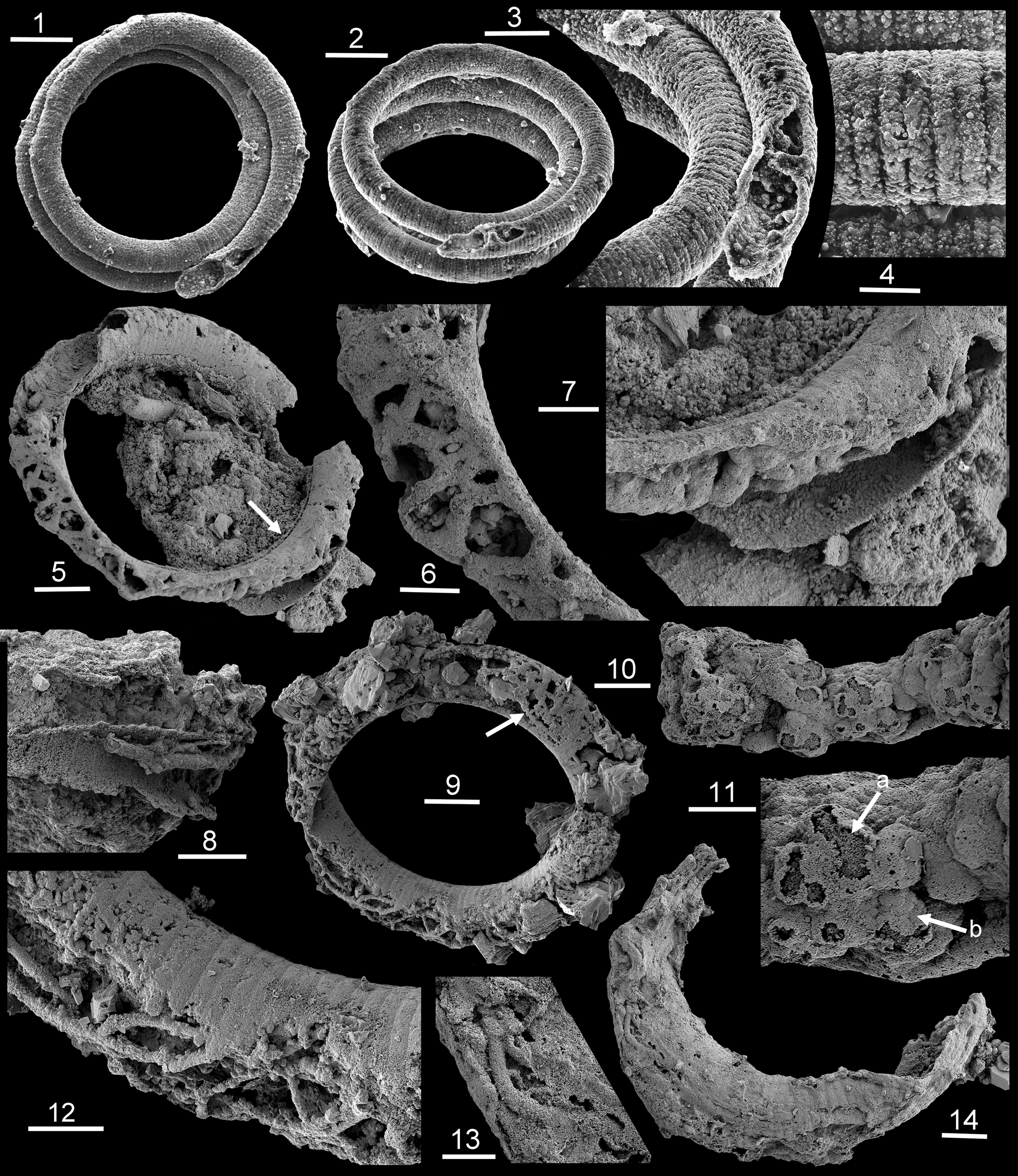
Figure 3. Mineralized threads (SEM) within Ediacaran Jiangispirellus, Portfjeld Formation, GGU sample 271770: (1–4) MGUH 17571, holotype, J. groenlandicus Peel, Reference Peel1988a from GGU sample 271769 (= 271770); (5–7) PMU 38168, with arrow in (5) locating outer encrustration detailed in (7); (8) PMU 36868/3, fragment with threads of two sizes; (9, 12) PMU 36866/5 with spherulites from threads adjacent to inner surface (9, arrow); note branching in (12); (10, 11) PMU 38169, seemingly detached fragment of a mass of cavity-dwelling threads with acicular termination of spherulite crystals (arrow a) covered by smooth layer of coalesced thin crystal plates (arrow b); (13, 14) PMU 36869/6. Scale bars = 100 μm (1, 2), 50 μm (3, 5, 8, 9), 20 μm (4, 6, 7, 10, 12, 14), 10 μm (11, 13).

Figure 4. Mineralized threads (SEM) of Cambrian cavity dwellers; Henson Gletscher Formation, GGU sample 271492, southern Lauge Koch Land; Miaolingian Series, Wuliuan Stage: (1, 8) phosphatocopid arthropod, PMU 38170, with dorsal surface broken, revealing cavity-dwelling threads; (2, 5) phosphatocopid arthropod, PMU 38171/1; (3, 4) cnidarian, PMU 38171/2, fragment of dendroid colony showing mineralized threads within corallites; (6, 7) fasiculate cnidarian, PMU 38172 from GGU sample 271718; (9) hyolithid protoconch, PMU 38173 from GGU sample 271718, with phosphatic coating on interior and exterior of the original calcareous shell, now dissolved, and internal threads (arrow). Scale bars = 500 μm (6), 200 μm (3), 100 μm (1, 2, 7–9), 50 μm (4, 5).

Figure 5. Mineralized threads (SEM) within Cambrian acrotretoid brachiopods: (1−3) Fimbuldal Formation, GGU sample 315006: (1) PMU 38174; (2) PMU 38175; (3) PMU 38176; (5) Fimbuldal Formation, PMU 38177 from GGU sample 218645; (4, 6, 8) Henson Gletscher Formation, PMU 38178 from GGU sample 298970, with arrow in (4) showing location of (6, 8); (7, 9–11) Holm Dal Formation, GGU sample 271414, westernmost Peary Land: (7, 9) MGUH 27491; (10) MGUH 27490; (11) PMU 23163. Scale bars = 300 μm (4, 11), 200 μm (2, 3, 5, 10), 100 μm (1), 50 μm (6, 8, 9), 20 μm (7).
Geological setting
The carbonate-dominated Portfjeld Formation is the lowermost formation of the transarctic Franklinian Basin in southern Peary Land (Higgins et al., Reference Higgins, Ineson, Peel, Surlyk, Sønderholm and Trettin1991a, Reference Higgins, Ineson, Peel, Surlyk and Sønderholmb; Ineson et al., Reference Ineson, Surlyk, Higgins and Peel1994; Ineson and Peel, Reference Ineson and Peel1997; Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020; Fig. 1.2). It rests unconformably on Mesoproterozoic sandstones of the Independence Fjord Group, which contain conspicuous dolerite intrusions and outliers of Neoproterozoic tillites and associated carbonates inferred to be of Marinoan age (Sønderholm and Jepsen, Reference Sønderholm and Jepsen1991; Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020). The Portfjeld Formation is unconformably overlain by transgressive siliciclastics of the Buen Formation, with the junction marked by a profound karstic surface. The sandstone-dominated lower member of the Buen Formation yields trace fossils of early Cambrian age, whereas the mudstone-dominated upper member contains rich faunas of Cambrian Series 2 (Stages 3–4) age (Peel and Willman, Reference Peel and Willman2018; Slater et al., Reference Slater, Willman, Budd and Peel2018; Wallet et al., Reference Wallet, Slater, Willman and Peel2021). The Buen Formation is overlain by a carbonate-dominated complex of prograding Cambrian (Series 2, Stage 4)–Early Ordovician formations (Fig. 1.2) assigned to the Brønlund Fjord and Tavsens Iskappe groups (Ineson and Peel, Reference Ineson and Peel1997)
The Portfjeld Formation in southern Peary Land consists of two stratigraphic units that are separated by a karstic unconformity (Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020). The lower unit (thickness ~170 m) of dolostones and rare limestones has yielded an Ediacaran biota (Peel, Reference Peel1988a; Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020). The transgressive upper Portfjeld Formation (70–90 m thick) is dominated by fluvial sandstones and high-energy, shallow marine sediments, likely of early Cambrian age (Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020), which are truncated by karstic collapse structures at the boundary with the overlying Buen Formation.
The Henson Gletscher Formation (Fig. 1.2) is distributed from western Peary Land westward across North Greenland (Higgins et al., Reference Higgins, Ineson, Peel, Surlyk, Sønderholm and Trettin1991a, Reference Higgins, Ineson, Peel, Surlyk and Sønderholmb; Ineson et al., Reference Ineson, Surlyk, Higgins and Peel1994; Ineson and Peel, Reference Ineson and Peel1997). The recessive, dark gray or black, thin-bedded, outer shelf carbonates and shales attain a maximum thickness of 112 m in southern Freuchen Land, with a prominent median sandstone unit, but thin to the north with a decreasing carbonate content. The formation is richly fossiliferous, ranging in age from Cambrian Series 2, Stage 4 to Miaolingian Series, Guzhangian Stage (Robison, Reference Robison1984, Reference Robison1994; Babcock, Reference Babcock1994; Blaker and Peel, Reference Blaker and Peel1997; Geyer and Peel, Reference Geyer and Peel2011; Peel et al., Reference Peel, Streng, Geyer, Kouchinsky and Skovsted2016; Streng et al., Reference Streng, Butler, Peel, Garwood and Caron2016; Peel, Reference Peel2017d).
The Fimbuldal Formation (80–180 m) is dominated by cliff-forming pale limestones and dolostones with a central unit of recessive limestones yielding a fauna of Drumian age (Ineson and Peel, Reference Ineson and Peel1997; Peel, Reference Peel2021; Fig. 1.2). The formation is overlain by recessive, thin-bedded limestones, dolostones, and mudstones of the Holm Dal Formation with rich faunas of Guzhangian age (Peel, Reference Peel1988b, Reference Peel2017b, Reference Peelc; Robison, Reference Robison1988; Zell and Rowell, Reference Zell and Rowell1988).
Material and methods
Material
Samples (Fig. 1) were collected during regional mapping programs (1978–1985) of Grønlands Geologiske Undersøgelse (GGU, Geological Survey of Greenland), Copenhagen, Denmark.
GGU sample 218645 was collected by J.R. Ineson on 7 July 1979 from the western side of Gustav Holm Dal, western Peary Land (82°20.3′N, 39°45′W) from just above the middle of a unit of dark, bituminous limestones forming the recessive middle unit of the Fimbuldal Formation in its type section (Ineson and Peel, Reference Ineson and Peel1997, figs. 44, 45). Cambrian, Miaolingian Series, Drumian Stage.
GGU sample 271414 was collected by P. Frykman and J.S. Peel on 19 June 1978 from the Holm Dal Formation in Gustav Holm Dal, western Peary Land (82°22′N, 39°20′W). Cambrian, Miaolingian Series, Guzhangian Stage.
GGU sample 271492 was collected by J.S. Peel on 25 June 1978 from ~7 m below the top of the upper Henson Gletscher Formation in its type section in southeastern Lauge Koch Land (82°10′N, 40°24′W) immediately above a prominent debris flow (Ineson and Peel, Reference Ineson and Peel1997, fig. 31). Cambrian, Miaolingian Series, Wuliuan Stage.
GGU sample 271718 was collected by J.S. Peel on 15 July 1978 from a thin-bedded, phosphatized, dolomitic limestone occurring ~1 m below the top of the formation on the western side of Løndal (82°18′N, 37°00′W; Clausen and Peel, Reference Clausen and Peel2012, fig. 1). Cambrian, Miaolingian Series, Wuliuan Stage.
GGU sample 271770 was collected by J.S. Peel and P. Frykman on 22 July 1978 from ~70 m above the base of the Portfjeld Formation on the northern side of Wandel Dal, west of Øvre Midsommersø (82°14′N, 36°06′W). Ediacaran microfossils from this sample were described by Willman et al. (Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020) in which the equivalent horizon was placed at ~50 m above the base in the presented measured section located a few kilometers to the east (Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020, fig. 2). The sample is a split of GGU sample 271769 from which cyanobacteria were described by Peel (Reference Peel1988a).
GGU sample 298970 was collected by J.S. Peel on 27 July 1985 from ~55 m above the base of the Henson Gletscher Formation, Hand Bugt, northern Nyeboe Land (82°11′N, 57°12′W, Fig. 1.1, inset). Trilobites from this sample were described by Babcock (Reference Babcock1994) and Robison (Reference Robison1994). Cambrian, Miaolingian Series, Wuliuan Stage.
GGU sample 301351 was collected by M.R. Blaker and J.S. Peel on 17 August 1985 from ~15 m above the base of the lower member of the Henson Gletscher Formation (Cambrian Series 2, Stage 4) on a nunatak in southern Freuchen Land (82°09′N, 42°25′W). Cambrian Series 2, Stage 4.
GGU sample 315006 was collected by J.S. Peel on 2 July 1984 from the recessive interval in the middle of the Fimbuldal Formation on the southwestern side of the glacier draining into Navarana Fjord, eastern Freuchen Land (82°17′N, 41°22′W). Miaolingian Series, Drumian Stage, Ptychagnostus punctuosus Biozone (Robison, Reference Robison1984).
Methods
Carbonate samples were dissolved in weak acetic or formic acid and wet-sieved in fractions (40 μm and coarser). Selected specimens were gold coated prior to scanning electron microscopy (SEM). Images were assembled in Adobe Photoshop CS4.
Terminology
Golubic et al. (Reference Golubic, Friedmann and Schneider1981) proposed ‘cryptoendoliths’ as the collective name for cavity-dwelling organisms within hard substrata in a nomenclature that also introduced the term ‘euendolith’ for borers into hard surfaces. Cryptoendolith is widely used in a restricted sense to describe organisms inhabiting the pore spaces of sediments, mainly sandstones, in studies of present-day extreme environments, e.g., hyperarid and dry Antarctic terrestrial environments (Friedmann, Reference Friedmann1982; Crits-Christoph et al., Reference Crits-Christoph, Robinson, Ma, Ravel, Wierzchos, Ascaso, Artieda, Souza-Egipsy, Casero and DiRuggiero2016; Coleine et al., Reference Coleine, Stajich, Zucconi, Onofri, Pombubpa, Egidi, Franks, Buzzini and Selbmann2018; Stan-Lotter, Reference Stan-Lotter, Seckbach and Rampelotto2019).
The terminology of Golubic et al. (Reference Golubic, Friedmann and Schneider1981) was employed by Bengtson et al. (Reference Bengtson, Rasmussen, Ivarsson, Muhling, Broman, Marone, Stampanoni and Bekker2017a), with regard to fungus-like threads in amygdales to 1.5 mm in diameter within Paleoproterozoic rocks and was reviewed by McLoughlin et al. (Reference McLoughlin, Staudigel, Furnes, Eickmann and Ivarsson2010). Stockfors and Peel (Reference Stockfors and Peel2005b) also used euendoliths and cryptoendoliths in describing bored shell walls and phosphatized threads within the internal cavity (crypt) in conjoined Miaolingian acrotretoid brachiopods from North Greenland. It is now known that the supposed euendoliths described by Stockfors and Peel (Reference Stockfors and Peel2005b) in the phosphatic shell material are mainly abiotic microtunnels, so-called ambient inclusion trails (McLoughlin et al., Reference McLoughlin, Staudigel, Furnes, Eickmann and Ivarsson2010; Olempska and Wacey, Reference Olempska and Wacey2016; Yang et al., Reference Yang, Han, Wang, Schiffbauer, Uesugi, Sasaki and Komiya2017), although numerous borings are present in the shell surfaces. Stockfors and Peel (Reference Stockfors and Peel2005b) considered the cavity within the conjoined hard brachiopod shells to be equivalent to the cavities within hard substrata described by Golubic et al. (Reference Golubic, Friedmann and Schneider1981); both were occupied by similar microorganisms growing inward from the walls. Álvaro and Clausen (Reference Álvaro and Clausen2010) also employed cryptoendolith for cavity-dwelling organisms within calcitic skeletons lying on the substratum from the Cambrian of southern France.
Kobluk (Reference Kobluk1981a, Reference Koblukb), Kobluk and James (Reference Kobluk and James1979), and Rowland (Reference Rowland1983) used coelobionts to describe organisms living within cavities, but in Kobluk's (Reference Kobluk1988) embracive terminology for these cavities (crypts), the term cryptobiont was employed for organisms living in a cavity (crypt). Kobluk (Reference Kobluk1988, p. 381) defined crypts as “habitats within all kinds of cavities or completely or partially enclosed void spaces” and cryptic organisms are the inhabitants of crypts. Taylor and Wilson (Reference Taylor and Wilson2003) gave an extensive review of communities through time associated with hard substrata, including cryptic occurrences.
Taylor and Wilson (Reference Taylor and Wilson2002) used cryptobiont to describe organisms colonizing hidden surfaces and regarded coelobionts as a subgroup of these for organisms living in small cavities. However, cryptobiont is also applied in a quite different context, as the descriptor of any organism capable of cryptobiosis, a reduced metabolic state entered in response to adverse environmental conditions, e.g., tardigrades (Wright et al., Reference Wright, Westh and Ramløv1992). Consequently, cryptobiont is not employed herein.
Wilson (Reference Wilson1986) and others have used coelobites (attributed to Ginzburg and Schroeder, Reference Ginsburg and Schroeder1973) as the appelation for cavity-dwelling organisms, but this was regarded as synonymous with coelobionts by Taylor and Wilson (Reference Taylor and Wilson2002) as a subgroup of cryptobiont.
Kobluk (Reference Kobluk1988, p. 381) proposed intraskeletal crypts for “habitats within open spaces in the skeleton (rigid or nonrigid) or body of a single sessile organism or organism colony.” All of the cavity-dwelling microorganisms described herein occur in cavities within other organisms, e.g., spheres (Fig. 2), helical cyanobacteria (Fig. 3), or a variety of shelled Cambrian invertebrates (Figs. 4, 5). As such, they could be described collectively as intraskeletal cryptos in the sense of Kobluk (Reference Kobluk1988), although there is overlap with the terminology of Golubic et al. (Reference Golubic, Friedmann and Schneider1981) when such crypts are incorporated into consolidated sediments.
Repositories and institutional abbreviations
GGU indicates a sample collected by Grønlands Geologiske Undersøgelse (Geological Survey of Greenland), now a part of the Geological Survey of Denmark and Greenland (GEUS), Copenhagen, Denmark. MGUH indicates a specimen deposited in the paleontological type collection of the Natural History Museum of Denmark, Copenhagen. PMU indicates a specimen deposited in the paleontological type collection of the Museum of Evolution, Uppsala University, Sweden.
Results
Spherical microfossils
Spherical phosphatized microfossils dominate GGU sample 271770 from the Portfjeld Formation at a locality west of Øvre Midsommersø in southern Peary Land, North Greenland (Fig. 1). This is currently the only occurrence of an Ediacaran biota comparable to the Doushantuo Lagerstätte of South China known from Laurentia (Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020). Many of the spheres in the Portfjeld biota are interpreted as eggs, but rare possible embryos show different stages of organismal development. Coiled cyanobacteria, acanthomorphic and leiosphaeric acritarchs, as well as possible red algal thalli complete the well-preserved, three-dimensional assemblage.
The putative eggs generally range in size from 100−200 μm and as such they are smaller than their Doushantuo assemblage counterparts. The outer surfaces can be smooth without structure or dimpled (Fig. 2.1) and folded. The spheres show a great range in preservation from pristine spheres, through collapsed, wrinkled spheres to corroded or degraded specimens with disfigured forms (Fig. 2.2). Broken surfaces indicate that many spheres are hollow, although granular, crystalline, phosphatic material is commonly present within others (Willman et al., Reference Willman, Peel, Ineson, Schovsbo, Rugen and Frei2020).
The granular surface of one dimpled specimen (Fig. 2.1) could reflect etching by acid during preparation, although some of the numerous small pits or depressions could represent the point of entry of euendoliths. Smooth surfaces in a second specimen (Fig. 2.2) indicate probable contact with the internal surface of the sphere, in part supported by the internal mineralized threads. In detail, the phosphatized threads have a granular crystalline texture (Fig. 2.6). Threads in these two specimens can separate (or more likely join, touch, or cross over each other) to form an irregular meshwork, but details are obscured by mineralization. The third specimen displays a dense mat of spherulites and short threads on the internal surface of the preserved fragment of the sphere and could represent mineralization of a bacterial layer produced during decomposition of soft parts.
Similar, larger, better-preserved spheres with phosphatized threads, 10–20 μm in diameter, and arranged in radial or anastomosing patterns were described from the Ediacaran Doushantuo Formation of South China by Xiao and Knoll (Reference Xiao and Knoll1999) who reviewed both the occurrence of such threads in the fossil record and their possible derivation. Fine threads were interpreted as probable filamentous bacteria and this interpretation could apply to the narrow threads on the sphere internal surface (Fig. 2.3). In detail, most threads show fibrous, radial spherulitic structure (Xiao and Knoll, Reference Xiao and Knoll1999, fig. 7; Xiao and Schiffbauer, Reference Xiao, Schiffbauer, Seckbach and Walsh2008, fig. 5C, D) not observed in the few available specimens from the Portfjeld Formation, although Cambrian material from North Greenland can show this spherulitic texture (Fig. 4.8).
Smooth, featureless, phosphatized spheres ~200 μm in diameter are widespread, but uncommon, in processed limestone samples from the early–middle Cambrian (Series 2, Miaolingian) of North Greenland. Such spheres are usually identified in the literature as the earliest growth stages of Olivooides Qian, Reference Qian1977 or Markuelia Val'kov, Reference Val'kov, Khomentovsky, Yakshin and Karlova1983 based on established developmental sequences (but see also Steiner et al., Reference Steiner, Zhu, Li, Qian and Erdtmann2004). Olivooides is currently interpreted as a cnidarian (Dong et al., Reference Dong, Vargas, Cunningham, Zhang, Liu, Chen, Liu, Bengtson and Donoghue2016), whereas Markuelia is usually considered to be a scalidophoran (Dong et al., Reference Dong, Bengtson, Gostling, Cunningham, Harvey, Kouchinsky, Val'Kov, Repetski, Stampanoni, Marone and Donoghue2010).
A sphere ~300 μm in diameter from the Henson Gletscher Formation (Cambrian Series 2, Stage 4) of southern Freuchen Land, referred to Olivooides, has broken to reveal numerous threads arranged radially within the crypt that intersect in a clotted mass at the center (Fig. 2.4, 2.5). As preserved, the threads display granular crystalline texture without information concerning their central core. Several thinner individual threads attached to the inner surface of the sphere can combine to form a single, more robust thread that extends toward the central mass of entangled threads (Fig. 2.5). A discounted alternative interpretation is that the threads arose from the central mass and divided as they approached the inner wall. The outer surface of the sphere is smooth and seemingly well preserved (Fig. 2.4) but pits indicative of the point of penetration of euendolith organisms, as they passed through the original outer surface of the sphere into its interior, have not been observed. The absence of pits, together with the presence of a discontinuous thin outer layer (Fig. 2.4, arrow) and the spherulite-bearing inner surface of the sphere (Fig. 2.5), could suggest that the dominant smooth outer surface is a mold of the interior of the sphere.
Specimens from Doushantuo illustrated by Xiao and Knoll (Reference Xiao and Knoll1999, fig. 7) show prominent internal bodies draped by fine bacterial threads and connected to the inner surface of the sphere by robust threads, but these are not closely similar to the radial pattern described here (Fig. 2.4).
The granular texture of the crystalline coating to the threads is similar to that seen in one Ediacaran specimen (Fig. 2.2, 2.6), with no indication of the spherulitic, botryoidal coating seen in specimens of Olivooides illustrated by Yue and Bengtson (Reference Yue and Bengtson1999, fig. 9) from the Terreneuvian of China. Because of indifferent preservation, the radiating crystal pattern has not been clearly distinguished in threads from the Portfjeld Formation (Fig. 3) and some Cambrian specimens (Fig. 4). It is seen, however, within acrotretoid brachiopods from the Henson Gletscher Formation (Fig. 5.6, 5.8; Wuliuan Stage) and Holm Dal Formation (Guzhangian Stage) of North Greenland (Fig. 5.7, 5.9, 5.10; Stockfors and Peel, Reference Stockfors and Peel2005b).
Olivooides specimens illustrated by Pyle et al. (Reference Pyle, Narbonne, Nowlan, Xiao and James2006) from the earliest Cambrian (Terreneuvian Series) Ingta Formation of Yukon Territory, Canada, display concentric and radial threads within the spheres that have been coated by spherulitic, botryoidal apatite; the mineralized threads (22 μm in diameter) contain a central core (10 μm in diameter) interpreted as the original organic thread. Concentric and radial threads were also described in similar spheres from the Manykay Formation (Terreneuvian, Fortunian Stage) of northern Yakutia (Sakha), Siberia, by Kouchinsky et al. (Reference Kouchinsky, Bengtson and Gershwin1999). Spheres of Markuelia infested with threads from the Dengying Formation (Terreneuvian) of Hunan Province, China, were illustrated by Bengtson and Yue (Reference Bengtson and Yue1997); Dong et al. (Reference Dong, Bengtson, Gostling, Cunningham, Harvey, Kouchinsky, Val'Kov, Repetski, Stampanoni, Marone and Donoghue2010, text-figs. 3G, 6J) illustrated similar specimens from Yakutia and China.
Cavity dwellers within Jiangispirellus
Phosphatized, helically coiled, oscillatoriacean cyanobacteria are the most conspicuous fossils in GGU sample 271770 from the Portfjeld Formation. Peel (Reference Peel1988a) assigned forms with a smooth sheath, with or without external calcification, to Spirellus Jiang in Luo et al., Reference Luo, Jiang, Wu, Song and Ouyang1982, originally described from China and the Indian Himalaya (Singh and Shukla, Reference Singh and Shukla1981; Brasier and Singh, Reference Brasier and Singh1987). Phosphatized trichomes without a sheath enclosing the trichome were assigned to a new genus, Jiangispirellus Peel, Reference Peel1988a, characterized by transverse annulation reflecting cell structure (Peel, Reference Peel1988a; Fig. 3.1–3.4), although some subsequent authors have considered Jiangispirellus and a variety of other taxa from Proterozoic-Phanerozoic boundary strata to be junior synonyms of a widely drawn Obruchevella Reitlinger, Reference Reitlinger1948 (Sharma and Shukla, Reference Sharma and Shukla2012).
Specimens of Jiangispirellus groenlandicus Peel, Reference Peel1988a show considerable variation in their preservation. Pristine specimens can be coiled through several whorls in helices with parallel sides. However, the maximum number of whorls is not known because of breakage, most probably during sample preparation, so that fragments of approximately one whorl are most common in the available residue. Successive whorls can be in contact or slightly separated but postmortal degradation results in slight slippage of the whorls relative to each other as the helix collapses (Fig. 3.1).
The holotype of Jiangispirellus groenlandicus preserves a naked trichome composed of a series of cylindrical cells, 50 μm in diameter (the width of the trichome) and ~8 μm in length (Fig. 3.1–3.4). Deep grooves between adjacent cells (Fig. 3.4) represent the cell walls and the infilling to each cell is composed of uniform, ovoid crystallites likely representing phosphatized bacteria (Peel, Reference Peel1988a). The cell infill was clearly formed prior to the degradation of the cell walls, although phosphatization did not extend far into the interior of the trichome, which is now a void with irregular spherulites (Fig. 3.3).
Other specimens lack such precise indications of cell walls and their preserved annulation represents mineralization of the outer surface of the trichome (Fig. 3.9, 3.12) or possibly casts from external molds of the trichome. The former can show a thin diagenetic coating of crystalline phosphate on the outside and inside of the original mineralized outer surface (Fig. 3.4) similar to the encrustation described by Xiao and Schiffbauer (Reference Xiao, Schiffbauer, Seckbach and Walsh2008, figs. 2–4) in microfossils from the Doushantuo Formation. The interior of the helical tubes is filled to varying degrees by diagenetic phosphate, so that the inner surface is often crystalline or granular in texture, whereas the annulated outer surface is sharply defined (Fig. 3.3, 3.12). Compact apatite internal molds of the type documented by Creveling et al. (Reference Creveling, Knoll and Johnston2014) in the Thorntonia Limestone (Cambrian, Miaolingian Series of Australia), and widespread in Cambrian samples from North Greenland and elsewhere, are not present in Portfjeld Formation samples.
In rare Jiangispirellus helices, the interior contains anastomosing phosphatized threads, usually with a botryoidal, probably spherulitic, texture (Fig. 3.5, 3.6), although the apparent anastomosing pattern can in part result from cross-overs or chance contacts between threads obscured by subsequent mineralization. However, true branching of threads is present (Fig. 3.7, 3.12). The outer surface of the spherulitic covering to the phosphatized threads can be covered with a smooth layer of coalesced, thin, plate-like crystals covering the sharp terminations of the acicular needles of the underlying spherulites (Fig. 3.10, 3.11). Individual short segments of the threads can be perpendicular to the walls or extend greater distances through the tubular helix (Fig. 3.13, 3.14). In one specimen (Fig. 3.10, 3.11), the botryoidal threads occur within a dense coccoid mass, which seems to have a filamentous foundation. The mineralized threads have a relatively uniform diameter of ~5–8 μm, suggesting that the original, much narrower organic threads could have a fungal origin. However, one Jiangispirellus fragment (Fig. 3.8) preserves short segments of thicker and thinner threads.
Cavity dwellers in Cambrian invertebrates
In North Greenland, Cambrian cavity dwellers are most common within articulated acrotretoid brachiopods, although examples are known from a number of other invertebrate groups.
The cryptic organisms on the inner surface of Cambrian bradoriid and phosphatocopid carapaces might have gained sustenance from decaying mantle tissues prior to mineralization (Fig. 4.2, 4.5) or developed within the cavity at a later diagenetic stage (Fig. 4.1, 4.8). The interior of some phosphatocopid arthropods displays thin mineralized sheets (Fig. 4.2) interpreted as remnants of the phosphatized internal lamella (Maas et al., Reference Maas, Waloszek and Müller2003, pls. 18B, 33C). In other phosphatocopids (Fig. 4.1, 4.8), the carapace, as preserved, is formed solely by thin phosphatic crusts deposited on the outside and inside of the original organic material.
Phosphatized threads are seen within the tip of a hyolithid (Fig. 4.9, arrow) in which the phosphatic crust deposited on the interior of the now dissolved shell has fractured. Similar structures were illustrated in an orthothecid hyolith from the Cambrian (Stage 3) of Bornholm, Denmark by Berg-Madsen et al. (Reference Berg-Madsen, Valent and Ebbestad2018, fig. 11A). In a fasciculate cnidarian (Fig. 4.6, 4.7), numerous branching phosphatized threads with botryoidal structure occur within the calices. Similar structures were described by Álvaro and Clausen (Reference Álvaro and Clausen2010) within hyolithids from the Cambrian of the Montaigne Noire, France. The preserved phosphatized corallites are slightly separated, with growth lines indicating that the outermost layer of the original calcareous walls of the calices is preserved. The inner surface of each phosphatized corallite is composed of spherulites continuous with those covering the threads. As indicated by the preserved growth lines, the outer wall is phosphatized in a dendroid cnidarian that contains mainly radial phosphatized threads (Fig. 4.3, 4.4). In these cnidarians, morphology of the mineralized threads is reminiscent of that seen in the tubes of the Ediacaran Jiangispirellus groenlandicaus (Fig. 3.7, 3.12).
Cavity dwellers in Cambrian brachiopods
Cavity-dwelling organisms within conjoined linguliformean acrotretoid brachiopods were described from the Holm Dal Formation (Miaolingian, Guzhangian) of western Peary Land by Stockfors and Peel (Reference Stockfors and Peel2005b). They are associated with longitudinally striated galleries within the acrotretoid shells that were interpreted as euendolithic borings, but these are now considered to be ambient inclusion trails of inorganic origin (McLoughlin et al., Reference McLoughlin, Staudigel, Furnes, Eickmann and Ivarsson2010; Olempska and Wacey, Reference Olempska and Wacey2016; Yang et al., Reference Yang, Han, Wang, Schiffbauer, Uesugi, Sasaki and Komiya2017). However, numerous euendolithic borings penetrate the outer surfaces of shells (Fig. 5.10), ooliths, and bioclasts, with some from the upper Henson Gletscher Formation (Wuliuan) in western Peary Land (GGU sample 271718; Fig. 1) referred to Eohyella by Stockfors and Peel (Reference Stockfors and Peel2005a).
The mineralized threads illustrated by Stockfors and Peel (Reference Stockfors and Peel2005b, fig. 3G, 5A) are composed of spherulites, with crystals radiating from an inner core. Several concentric episodes of mineralization can be present (Stockfors and Peel, Reference Stockfors and Peel2005b, figs. 2D, 3G, 5A). The threads have a botryoidal form (Fig. 5.6, 5.10) and vary from simple threads to networks and tangled masses, but true branching is common (Fig. 5.1, 5.3, 5.9). Brachiopods incorporated within the microbial lamination of phosphatic hardgrounds near the base of the Holm Dal Formation often show a dense pattern of subradial threads (Peel, Reference Peel2017c; Fig. 5.11).
Within brachiopods from the Henson Gletscher and Holm Dal formations, the spherulitic mineralization on threads can predate or occur simultaneously with the formation of phosphatic crusts on the shell inner surfaces (Fig. 5.4, 5.6, 5.8). Coated threads are frequently overgrown by layers with conspicuous fibrous structure perpendicular to the surface (Fig. 5.8). Similar spherulitic overgrowth of the original organic threads is seen in brachiopods from the Henson Gletscher Formation in GGU sample 298970 (Wuliuan) from Nyeboe Land (Figs. 1, 5.6, 5.8), whereas the botryoidal form of the mineralized threads is evident in specimens from the Fimbuldal Formation (Drumian; Fig. 5.1–5.3, 5.5).
Mineralized threads from the Fimbuldal Formation are often arranged in the plane of the commissure (Fig. 5.2, 5.5), suggesting entry into the inner cavity through the junction between the ventral and dorsal valves. However, some of the planar networks might be geopetal in origin, reflecting growth of the original threads across the upper surface of partial infilling by sediment or spar. In other specimens, there is a clear association between the mineralized threads and the posterior foramen of the brachiopod shell (Fig. 5.3).
Discussion
Modern studies have demonstrated the speed of decay and mineralization in present-day settings (Briggs, Reference Briggs1995, Reference Briggs, Krumbein, Paterson and Zavarzin2003; Sansom et al., Reference Sansom, Laflamme, Schiffbauer and Darroch2014; Briggs and McMahon, Reference Briggs and McMahon2015). Exquisite preservation in Cambrian Lagertstätten, e.g., the Orsten of Sweden, indicates fidelity of the mineralization process (Maas et al., Reference Maas, Waloszek and Müller2003; Eriksson et al., Reference Eriksson, Terfelt, Elofsson and Marone2012). Ediacaran fossils from the Doushantuo Biota of China, equivalent to the Portfjeld biota of North Greenland, are preserved by encrustation and direct replication of tissues by apatite (Xiao and Knoll, Reference Xiao and Knoll1999, Reference Xiao and Knoll2000), although the latter can be modified by later diagenesis (Schiffbauer et al., Reference Schiffbauer, Xiao, Sen Sharma and Wang2012). Direct replication by apatite is not documented here; encrustation is the dominant process. However, phosphatization was not a single, uniform event in the Greenland material, as noted also by Xiao and Schiffbauer (Reference Xiao, Schiffbauer, Seckbach and Walsh2008) in their detailed study of the preservation of phosphatized spheres from the Doushantuo Formation of China. Álvaro and Clausen (Reference Álvaro and Clausen2010) recognized several styles of phosphatization in Cambrian crypts from southern France, but the growth of botryoidal structure both on threads and surfaces is prevalent in the material at hand (Fig. 5.6, 5.9). A grainy, crystalline texture is preserved on threads within the sphere from the Henson Gletscher Formation (Fig. 2.4–2.6).
It is uncertain at which point the various cryptic threads in the North Greenland material were formed and subsequently mineralized in the taphonomic history of the individuals enclosing host organisms. Crypts can form and become occupied in the walls of thick-shelled hosts, e.g., corals, while the organism is still alive (Scoffin and Bradshaw, Reference Scoffin and Bradshaw2000), although such examples are not documented here. Bacterial and fungal growth resulting from the degradation of original organic tissues within the host crypt and their mineralization likely proceeded prior to invasion and mineralization by other cryptic organisms.
The relatively soft eggs and spiral cyanobacteria degraded rapidly and various stages in the degradation and deformation are preserved in samples. However, phosphatization of outer surfaces and probably the initial growth of cryptic communities were rapid. Evidence of breakage of phosphatized surfaces by diagenetic compaction is not widespread, indicating that degradation occurred prior to mineralization.
It is not known how and when free-lying organisms with intraskeletal crypts were incorporated into the sediment on the sea floor. In environments with well-developed microbial mats, loose-lying shells could have been enveloped quickly (Fig. 5.12 shows cryptic threads and coccoid bodies within an acrotretoid brachiopod in a microbially laminated hard ground; Peel, Reference Peel2017c). Crypts that were formed and occupied by cryptic organisms immediately after death, with the enclosing organisms lying on the sediment surface, could have been maintained and their cryptic components supplemented after burial beneath the sea floor. The hard-shelled organophosphatic brachiopods (Fig. 5) and calcified cnidarian skeletons (Fig. 4.3, 4.4, 4.6, 4.7) are resistant to compaction and the internal cavities occupied by the cryptic organisms could have persisted unfilled by calcium carbonate spar for some time after demise and burial of the animal.
Habitation by phototrophs, e.g., cyanobacteria, might not have been possible as crypts that were initially formed at or near the surface became more deeply buried, although Chafetz and Buczynski (Reference Chafetz and Buczynski1992) documented that dead cyanobacteria below the upper phototrophic zone in algal mats were more readily mineralized (and therefore preserved) through bacterial mediation than living threads. It is also possible that dead cyanobacteria might be preferentially preserved in bacteria-rich environments within crypts, but leave no trace in surrounding sediments. Other organisms comprising the deep biosphere (Ivarsson et al. Reference Ivarsson, Drake, Neubeck, Sallstedt, Bengtson, Roberts and Rasmussen2020) are known from depths in excess of 1 km beneath the sea floor.
Apart from bacteria and fungi resulting from the decay of soft parts, the intraskeletal crypt organisms must have gained access to the crypts that they inhabited. Often, as in the various invertebrates described herein (Figs. 4, 5), access was provided by natural openings, e.g., the aperture, plane of commissure, or the pedicular opening. Schroeder (Reference Schroeder1972) and Golubic et al. (Reference Golubic, Friedmann and Schneider1981) envisaged penetration to cryptoendolith cavities by euendoliths passing through surrounding hard sediments, a mechanism similar to the passage of euendolithic threads through organism walls in invertebrates (Bentis et al. Reference Bentis, Kaufman and Golubic2000; Stockfors and Peel, Reference Stockfors and Peel2005b). So-called ‘passive’ crypts originated by overgrowth of infestations by the host coral with subsequent growth, and often maintained an open connection (Scoffin and Bradshaw, Reference Scoffin and Bradshaw2000). Ivarsson et al. (Reference Ivarsson, Drake, Neubeck, Sallstedt, Bengtson, Roberts and Rasmussen2020) noted the importance water transport in hydrologically active environments.
The affinity of cryptic organisms is not known due to the subsequent heavy phosphatization. Fungi often have slender threads with diameters of 1–5 μm, whereas those of algae and cyanobacteria are more substantial. Thus, cryptic threads in Jiangispirellus (Fig. 3.5, 3.9) were likely fungi, but even the robust mineralized threads in described brachiopods, which attained 30 μm in diameter (Fig. 5.6–5.8), formed around slender threads. Threads interpreted as cyanobacteria would have formed in the photic zone but the penetration of light was likely reduced by the enclosing host. In present-day microbial mats, the photic zone is often only a few millimeters thick, but bacterially induced mineralization of dead cyanobacteria can be conspicuous immediately below this thin photic layer (Chafetz and Buczynski, Reference Chafetz and Buczynski1992). The mineralization of dead cyanobacteria reported by Chafetz and Buczynski (Reference Chafetz and Buczynski1992) could distort the interpretation of fossil occurrences. Present-day green algae have a greater depth tolerance, extending into deep water by fungi (Schroeder, Reference Schroeder1972; Golubic et al., Reference Golubic, Radtke and Le Campion-Alsumard2005, Reference Golubic, Campbell, Lee and Radtke2016). It is evident, however, that mineralization quickly followed thread formation, often on several occasions (Stockfors and Peel, Reference Stockfors and Peel2005b; Fig. 5.7), although earlier (and later) generations of unmineralized cryptic threads might have passed without record.
Conclusions
The morphology of diagenetically mineralized threads of cavity-dwelling microorganisms from the Portfjeld Formation (Ediacaran) of North Greenland is similar to that of more widespread occurrences in Cambrian (Series 2 and Stage 4, Miaolingian Series) strata from the same area. The cavities formed postmortally within other organisms, e.g., cyanobacteria, brachiopods, cnidarians, and possibly eggs.
Spherulitic phosphatic coating to the threads obscures their identity, although it is likely that many of the slender original threads were fungi. Some threads might have resulted from degradation of pre-existing soft parts immediately after death of the host. Others resulted from penetration of the outer hard surface of the original organisms into internal cavities through natural openings, e.g., the plane of commissure or pedicular opening in brachiopods. A third group entered by way of euendolithic borings through the cavity wall, although the point of entry of these is generally obscured by diagenetic encrustation. Preservational constraints prevent assessment of the diversity of the original threads, but it is expected that specialist cavity dwellers, decomposers recycling nutrients, and even members of the surrounding sea-floor microbial communities were present.
Encrustation of the threads could have taken place in several phases and it is not unlikely that several periods of thread formation occurred, with some events remaining unmineralized. Initial thread formation and mineralization probably commenced soon after death of the host organisms, as they lay on the sea floor, but both processes might have been supplemented during and after burial before the cavities were finally closed by carbonate deposition.
Acknowledgments
Cooperation with J.R. Ineson (Copenhagen) concerning North Greenland geology is gratefully acknowledged. J.O.R. Ebbestad (Uppsala) assisted with the curation of specimens. Comments from T.H.P. Harvey (Leicester), S. Xiao (Blacksburg, Virginia), an anonymous reviewer, and the journal editors are gratefully acknowledged.








