In humans it has been well documented that transition from an environment where food is poor or adequate to one where diet is high in fat and carbohydrates is associated with higher prevalence of the metabolic syndrome, typified by type 2 diabetes, hypertension, hypercholesterolaemia and obesity(Reference Bateson, Barker and Clutton-Brock1–Reference Vickers, Breier and Cutfield6). It has been suggested that such a ‘nutritional transition’ to a Western diet may have deleterious consequences on the health of future generations by affecting early development, influencing susceptibility to disease in later life. We recently proposed that the extent of such susceptibility depends on the degree of mismatch between the developmental and later environments, for example, in nutritional content(Reference Gluckman and Hanson7).
Developmental induction of cardiovascular and metabolic risk factors has been observed in rodent models in which the pregnant dam is exposed to nutrient restriction and the offspring fed a normal chow diet(Reference Desai, Byrne and Zhang8–Reference Ozaki, Nishina and Hanson10). However, there are fewer studies that have examined adaptive and maladaptive mechanisms during fetal development when pregnant dams are exposed to overnutrition. The issue is particularly important in the light of the increasing consumption of refined foods with a high glycaemic index and fat content among men and women now consuming 30 % more saturated fats than the recommended daily intake(11). Several studies, including ours, have shown in rats that a maternal diet rich in fat during pregnancy results in features of the metabolic syndrome such as obesity, sedentary behaviour and vascular dysfunction in the offspring(Reference Bayol, Simbi and Stickland12–Reference Levin and Govek15). However, these studies were confined to short-term modifications in the maternal diet during pregnancy and/or lactation periods alone. Altering the maternal diet during critical periods of gestation(Reference Langley-Evans, Clamp and Grimble16, Reference Zambrano, Bautista and Deas17) or throughout gestation and/or the suckling period(Reference Khan, Dekou and Douglas18, Reference Zhang, Wang and Terroni19) results in a varying degree of phenotypic outcomes associated with the metabolic syndrome, suggesting the importance of the timing and duration of the nutritional insult. In humans, consumption of a high-fat diet just during pregnancy or suckling is not common, but there is increasing concern about the effects of obesogenic diets in children on the health of their future offspring. We therefore extended our animal model to investigate the consequences for their offspring of feeding dams a high-fat (HF) diet from the time that they were weaned, through their pregnancy and lactation until their pups are weaned. In addition we determined whether there are additive effects of feeding these offspring themselves an HF diet from weaning to adulthood.
Methods
Experimental protocol
All animal procedures were in accordance with the UK Animals (Scientific Procedures) Act 1986. Female C57BL/6 mice (Charles River Laboratories, Margate, Kent, UK) were maintained under a 12 h light–dark cycle at constant temperature (25 ± 2°C) with food and water available ad libitum. At age 4 weeks they were randomly allocated to either a control diet of standard laboratory chow (C; 5·3 % fat (maize oil), 21·2 % protein and 49·2 % carbohydrate; Special Diet Services, Witham, Essex, UK) or an HF experimental diet supplemented 18% (w/w) with animal lard with additional vitamins and minerals, protein and choline to correct for the dilution (final composition in g % (w/w): lard, 17·8; casein, 26·5; choline chloride, 0·3; l-cysteine, 0·4; rice starch, 28·3; cellulose, 6·1; soya oil, 4·3; sucrose, 10·4; minerals, 4·3; vitamins, 1·2; Special Diet Services diet no. 824053, Witham, Essex, UK). This HF diet is as used in previous studies(Reference Van Heek, Compton and France20). At age 10 weeks, the females were time-mated and, after confirmation of mating (i.e. presence of vaginal plug), were individually housed under a 12 h light–dark cycle at constant temperature with water available ad libitum.
After birth, pups were weighed and litter size was reduced to eight pups and, when possible, to equal numbers of males and females. From weaning (21 d post-partum) offspring from the HF and C dams were fed either HF or C diets. We refer to the offspring born to HF dams as HF-HF and HF-C according to their post-weaning diet. Similarly, offspring born to C dams are referred to as C-HF and C-C according to their post-weaning diet. Food intake and body weights were monitored weekly until the offspring reached adulthood. Adult offspring were killed at age 36 weeks by cervical dislocation. Blood was collected by cardiac puncture and fat depots (i.e. gonadal, retroperitoneal, interscapular, inguinal and peri-renal) were dissected and weighed. Cumulative fat depot weights for each animal were compared with total body weights and body fat as a percentage of total body weight was calculated. The livers were also dissected, fixed in 10 % neutral buffered formaldehyde and stored for further histological analyses.
Blood pressure measurements
Systolic arterial blood pressure (BP) was measured by tail-cuff plethysmography, as described previously by Krege et al. (Reference Krege, Hodgin and Hagaman21) who showed that BP taken by this method was highly correlated with intra-arterial BP measured by telemetry in unrestrained, unanesthetised animals(Reference Krege, Hodgin and Hagaman21). We conducted the measurements in a heated room (27–28°C) in order to get optimal BP readings at the same time during the day. All animals were made accustomed to the procedure for 7 d before each BP measurement session. At least five readings were taken from each animal per session and averaged to get a single session value. BP was measured at 13, 18, 23, 27, 30 and 36 weeks post-weaning. At each time point, we took the average BP values from eight offspring of each sex picked randomly from each of the eight litters in each treatment group.
Measurement of plasma C-reactive protein and cholesterol
C-reactive protein (CRP) in the serum was measured using a sensitive double-antibody sandwich immunoassay enzymic ELISA rabbit anti-human CRP and peroxidase-conjugated rabbit anti-mouse CRP (VITROS CRP Slides; Vitros Products, Rochester, NY, USA)(Reference Danesh, Wheeler and Hirschfield22). Monoclonal anti-CRP antibody conjugated to horseradish peroxidase served as the signal generator. The assay was linear up to 5 mg/l and logarithmic thereafter. The inter-assay CV were less than 10 % across the range of measured results. Serum total cholesterol was measured with commercially available kits (Vitros Products) using enzymic methods and measured by reflectance spectrophotometry as previously reported(Reference Levin and Govek15, Reference Khan, Dekou and Douglas18).
Histology
Fixed liver tissues were embedded in paraffin and 5–10 μm sections were cut and mounted on glass slides. Deparaffinised, fixed sections were stained with haematoxylin and eosin. Microscopic examination was performed on stained liver sections from representative animals in each group.
Data analysis
The biochemical and biophysical parameters in dams were analysed using one-way ANOVA followed by the Tukey–Kramer comparisons test. All data are expressed as mean values with their standard errors. A P value less than 0·05 was considered to be statistically significant. All statistical analysis was calculated with SPSS 14.0 (SPSS, Inc., Chicago, IL, USA).
Results
Body weight and adiposity
We did not find any significant difference in food intake among the various treatment groups during the experimental period (data not shown). HF-HF and C-HF male offspring were heavier at 36 weeks post-weaning than HF-C and C-C males (Fig. 1 (A)). These changes were also reflected in their total body fat (as percentage body weight), where the HF-HF and C-HF males had greater fat depots compared with the C-C and HF-C groups (Fig. 1 (C)). In female offspring the HF-C offspring were lighter than the HF-HF and C-HF animals but heavier compared with the C-C animals (Fig. 1 (B)). Corresponding increases in total body fat were observed in these groups compared with the C-C females (Fig. 1 (D)).
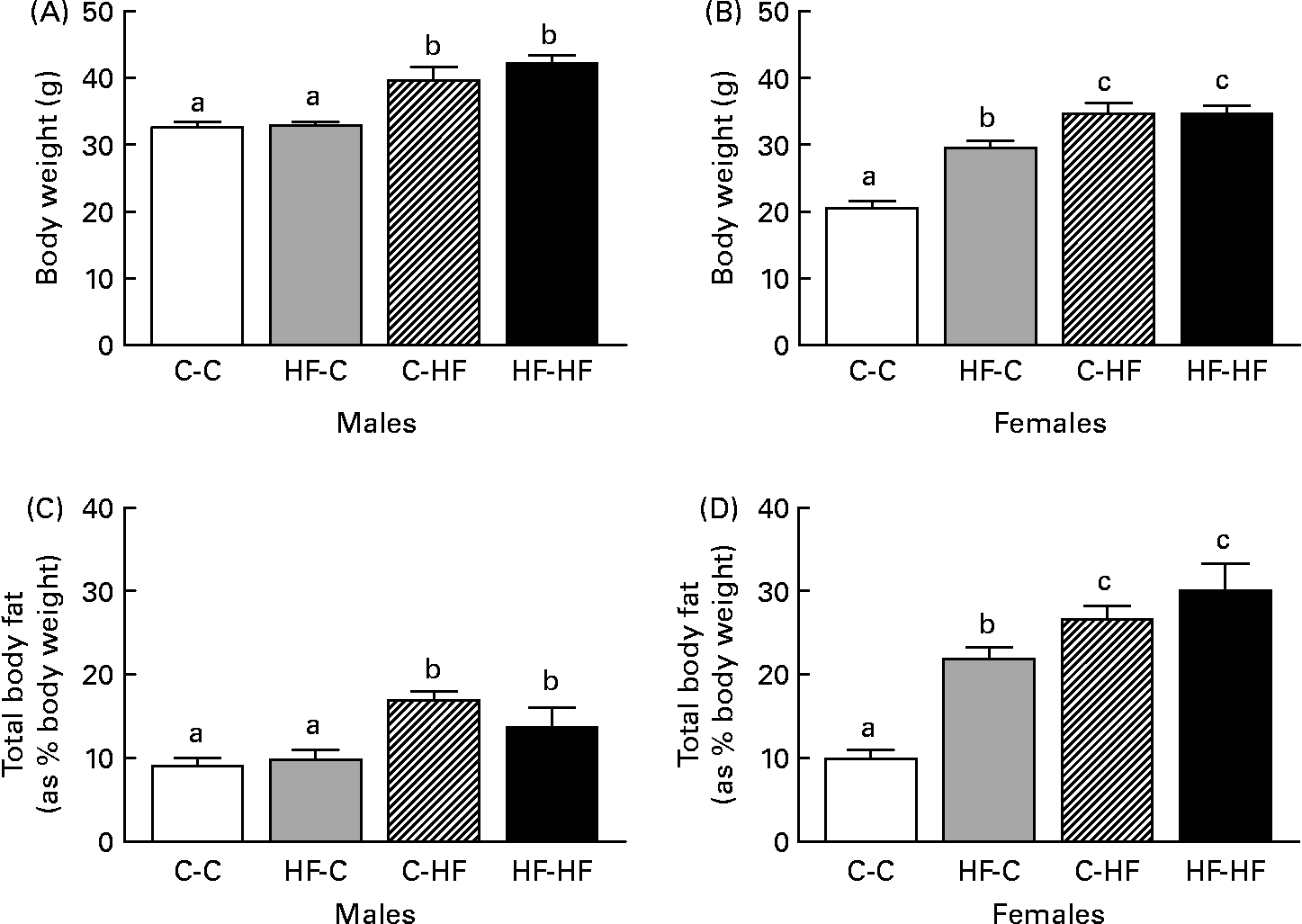
Fig. 1 Comparison of body weight (A, B) and total body fat (C, D) in male (A, C) and female (B, D) offspring from control-fed mothers that were then fed a chow diet (C-C) or a high-fat diet (C-HF) and from high-fat-fed mothers that were then fed a high-fat diet (HF-HF) or a chow diet (HF-C). Values are means (n 8–10 per group), with standard errors represented by vertical bars. a,b,c Mean values with unlike letters were significantly different (P < 0·05; Tukey–Kramer comparisons test).
Blood pressure
Systolic BP was elevated in the HF-HF, HF-C and C-HF male and female offspring at 36 weeks post-weaning compared with C-C offspring (Fig. 2 (A) and (B)). The HF-C males had the highest BP and this was significantly greater than the HF-HF and C-HF groups. In the females, the HF-HF and HF-C groups had significantly elevated BP levels compared with the C-HF offspring.
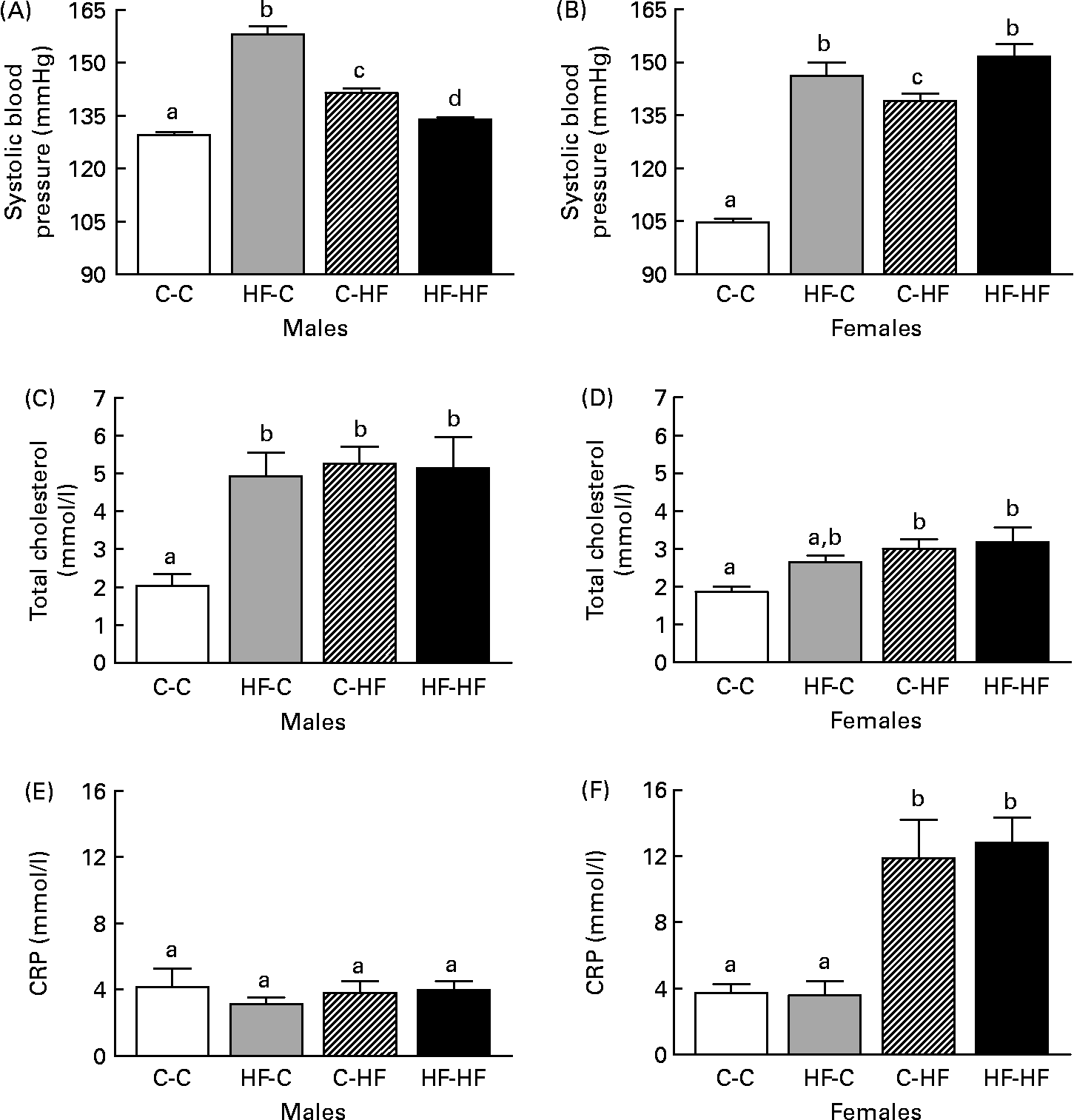
Fig. 2 Comparison of systolic blood pressure (A, B), total cholesterol (C, D) and C-reactive protein (CRP) levels (E, F) in male (A, C, E) and female (B, D, F) offspring from control-fed mothers that were then fed a chow diet (C-C) or a high-fat diet (C-HF) and from high-fat-fed mothers that were then fed a high-fat diet (HF-HF) or a chow diet (HF-C). Values are means (n 8–10 per group), with standard errors represented by vertical bars. a,b,c Mean values with unlike letters were significantly different (P < 0·05; Tukey–Kramer comparisons test).
C-reactive protein and cholesterol levels
Total cholesterol was elevated in the HF-HF, HF-C and C-HF males at 36 weeks post-weaning compared with the C-C males (Fig. 2 (C)). Interestingly, no difference was observed in CRP levels among these four treatment groups (Fig. 2 (E)). In females, total cholesterol and CRP level were elevated in the HF-HF and C-HF animals compared with the C-C offspring (Fig. 2 (D) and Fig. 2 (F), respectively). Total cholesterol in the HF-C group was also elevated but was not significantly different from the C-C or from the C-HF and HF-HF groups.
Development of fatty liver in adult offspring
Histological examination of the liver showed that the C-C offspring had normal liver structure (Fig. 3). However, we observed lipid vacuoles of various sizes within hepatocytes of the C-HF, HF-HF and HF-C offspring. Moreover, mononuclear cell infiltration, pyknotic nuclei and the rupturing of the endothelium of some central veins were observed in the livers in these groups of offspring compared with C-C offspring (data not shown).

Fig. 3 Liver histology in female offspring from control-fed mothers that were then fed a chow diet (C-C) or a high-fat diet (C-HF) and from high-fat-fed mothers that were then fed a high-fat diet (HF-HF) or a chow diet (HF-C). C-C offspring had normal liver structure. However, lipid vacuoles ( ← ) of various sizes could be observed within hepatocytes of the C-HF, HF-HF and HF-C offspring. Staining with haematoxylin and eosin; magnification × 20; bar scale = 40 μm.
Discussion
Nutritional status during critical periods of early life has important influences on development, and modification of the quality and/or quantity of maternal nutrition during pregnancy has been shown to have consequences on the later health of the offspring, changing their responses to environmental challenges and thus their predisposition to disease(Reference Langley-Evans23, Reference Oh, Gelardi and Cha24). In the present study we examined in a mouse model the consequences to the offspring of a long-term maternal HF dietary regimen, starting from when the prospective mothers were themselves weaned until weaning of their offspring. This contrasts with earlier animal experiments, which focused on the consequences for the offspring of maternal HF feeding during only gestation and/or lactation periods(Reference Bayol, Simbi and Stickland12, Reference Khan, Dekou and Hanson13, Reference Levin and Govek15). The long-term HF feeding results in changes in the dam's physiology, including increased body weight and raised circulating total and LDL-cholesterol levels compared with the C-fed group, as observed in our previous study(Reference Elahi, Cagampang and Anthony25). These changes in the HF dams may provide ‘cues’ used by the developing offspring in altering phenotype in anticipation of their postnatal environment. The long-term dietary regimen used in the present study may better represent the situation in human populations, following socio-economic transitions where consumption of high-fat food occurs very early in life and continues in women through pregnancy and lactation. The present study demonstrates that offspring of such mothers are predisposed to becoming fat, hypercholesterolaemic and hypertensive in adulthood, thus perpetuating the cycle of chronic disease.
We previously reported that prenatal and early postnatal exposure to a maternal diet rich in animal fat leads to the development of characteristics similar to the human metabolic syndrome in adult rats, even when they are reared on a balanced diet(Reference Khan, Dekou and Hanson13, Reference Khan, Taylor and Dekou14). The present study clearly shows that long-term consumption of an HF diet by the female dams predisposes their offspring to obesity, hypercholesterolaemia, hypertension and fatty liver in adult life, at least when they were also fed an HF diet. Greater adiposity was also seen in females, even when they were fed a C diet post-weaning, suggesting that predisposition to obesity had been induced during development. This was not, however, seen in the HF-C male offspring.
In males the elevation of BP was less pronounced in the HF-HF group than in the HF-C or C-HF groups. This supports the partially beneficial cardiovascular effect of reducing the dietary mismatch in the HF-HF offspring reported in our earlier study(Reference Khan, Dekou and Hanson13) although in that study the dams were only fed the HF diet during pregnancy and weaning, and endothelial dysfunction rather than elevated BP was attenuated compared with the C-fed offspring. These data are broadly in support of the predictive adaptive response concept(Reference Khan, Dekou and Hanson13). However, as outlined in the original exposition of the concept(Reference Gluckman and Hanson3), such an effect only operates within a range of postnatal environments, beyond which the risk of pathophysiology is increased. The postnatal HF used in the present study is likely to take the offspring into such a range, one which from an evolutionary point of view is novel, possibly explaining the pathophysiological effects observed in them.
We observed a substantial number of lipid vacuoles within hepatocytes of the HF-HF, HF-C and C-HF but not the C-C offspring. Previous studies in rats have demonstrated that offspring of fat-fed dams have profound metabolic defects such as increased liver weight and liver TAG content(Reference Guo and Jen26, Reference Koukkou, Ghosh and Lowy27). It has been reported in humans that there is a strong association between hypercholesterolaemia, steatohepatitis (non-alcoholic fatty liver) and increased risk of CVD(Reference Reid28) but it is unclear whether certain diets are more likely to produce these effects than others. Steatohepatitis and atherosclerosis are both produced in apo-E lipoprotein-deficient mice, suggesting a pathophysiological effects on both the liver and aorta of cholesterol-enriched diets(Reference Shiri-Sverdlov, Wouters and van Gorp29, Reference Tous, Ferre and Camps30). The accumulation of lipids in hepatocytes suggests possible interference with mitochondrial and microsomal function leading to disruption in lipoprotein transport and fatty acid accumulation v. metabolism(Reference Khan, Taylor and Dekou14). The accumulation of lipids in hepatocytes in the HF-C offspring suggests that susceptibility to development of fatty liver can be induced during early development.
Chronic inflammation is a major contributor to atherosclerosis and CVD(Reference Libby31). An important marker of inflammation is an elevation in CRP, an acute-phase reactant secreted by hepatocytes in response to pro-inflammatory cytokines such as IL-6(Reference Libby, Willerson and Braunwald32). In several large epidemiological studies, CRP has been shown to be a strong, independent predictor of CVD risk in both men and women(Reference Ridker, Cushman and Stampfer33, Reference Ridker, Hennekens and Buring34). The mechanism by which inflammation increases CVD risk is not known, but during periods of acute inflammation lipid metabolism is altered, giving a a proatherogenic profile(Reference Khovidhunkit, Kim and Memon35). This is what we observed in our HF-HF and C-HF female offspring, which had elevated CRP levels. Interestingly, we did not find such increases in the male offspring, suggesting that the effect of HF exposure on CRP levels is sex specific. The increase in CRP levels found only in female offspring on the HF diet may be a consequence of increased sensitivity to the HF diet brought about by circulating female sex hormones(Reference Ridker, Hennekens and Buring34, Reference Ridker, Hennekens and Rifai36) or due to the fact that the HF diet itself may result in high CRP in females. This observation is supported by human studies where CRP levels tend to be higher in women than in men(Reference Ridker, Cushman and Stampfer33, Reference Ridker, Hennekens and Buring34, Reference Visser, Bouter and McQuillan37–Reference Ridker, Hennekens and Buring39).
Perspectives
The current findings indicate that long-term consumption of an HF diet by the mother predisposes her offspring to obesity, hypercholesterolaemia, hypertension and development of fatty liver in adult life. This adverse effect on the offspring is induced during development and is not necessarily or completely reversed by either consumption of a postnatal C diet or indeed an HF diet. The results from the present study provide an experimental basis for investigating consequences of dietary transitions relevant to humans, where the woman's diet both before and during pregnancy and lactation may be a contributing factor to the development of metabolic and cardiovascular disease in her children.
Acknowledgements
We appreciate the technical assistance provided by Ronald Lee (Histochemistry Research Unit, University of Southampton) in performing the liver histology. Authors contributions are as follows: M. M. E. conducted the animal experiments and analysed the data; F. R. C. helped design the study and analysed the data; F. W. A. and D. M. assisted with laboratory analysis; S. K. O. helped design the study and M. A. H. designed the study and assisted with analysis. All authors were involved in writing the paper.
The present study was supported by the British Heart Foundation. M. M. E. is supported by a BUPA/HOPE research fellowship.
There are no conflicts of interest to declare.





