The World Health Organization recommends that all infants should be exclusively breastfed for the initial 6 months of life. 1 Infants with congenital heart disease (CHD) frequently face feeding difficulties and challenges regarding nutrition, development, and growth. Reference Argent, Balachandran, Vaidyanathan, Khan and Kumar2 These infants are trapped in a vicious cycle of undernutrition, infectious complications, and delay in surgery which in turn leads to more undernutrition. It has been seen that children with heart diseases are prone to high rates of early weaning. Reference Tandberg, Ystrom, Vollrath and Holmstrom3 Previous studies have shown that the nutritional status of children with CHD at the time of surgery is associated with surgical outcomes. Reference Radman, Mack and Barnoya4–Reference Xu, Lin, Zhang, Zheng and Lin7 Growth failure in infancy can also have a long-term impact and is associated with cognitive impairment. Reference Xu, Lin, Zhang, Zheng and Lin7,Reference Dykman, Casey, Ackerman and McPherson8 Therefore, it is imperative to prevent growth failure in children with heart disease so that both short- and long-term negative consequences of malnutrition may be avoided. To prevent growth failure, alternate modes, formulae, and pathways for feeding infants with heart diseases have been developed. Reference El-Koofy, Mahmoud and Fattouh9–Reference Centeno-Malfaz, Moráis-López and Caro-Barri13 However, because of a lack of robust scientific data on this subject, there is wide variability in practices of feeding an infant with complex CHD. Reference Elgersma, McKechnie, Gallagher, Trebilcock, Pridham and Spatz14 Several studies have also documented improved weight gain in children with heart disease using a predefined feeding protocol. Reference Xu, Lin, Zhang, Zheng and Lin7,Reference El-Koofy, Mahmoud and Fattouh9,Reference Marino, Johnson and Davies15–Reference Zhang, Zhou, Chen, Cao and Chen17 Most of these studies have used supplemental formulas to make energy-dense feeds, which may not be a viable option for parents in low- and middle-income countries because of issues related to cost. There is also a higher risk of contracting infections if bottle feeding is done unhygienically. To prevent growth failure, infants with heart disease are sometimes advised to give expressed breast milk by spoon to decrease the energy expenditure while sucking and to provide all the nutritive values of breast milk. This method appears logical and can easily be implemented without any additional financial burden to the family, but its feasibility and effectiveness have not been tested objectively.
This study was done to assess the effect of supplemental feeding of expressed breast milk on weight gain in infants with un-operated acyanotic CHD and increased pulmonary blood flow.
Material and methods
This was a prospective open-label, pilot, randomised control trial conducted in a tertiary care teaching hospital from 1st January 2021 to 31st May 2022. The study was approved by the Institutional Ethics Committee. Patients attending the outpatient clinic of the Division of Paediatric Cardiology were assessed for enrollment. Eligible infants fulfilling the inclusion criteria, and not having any exclusion criteria, were enrolled after taking informed consent from the parents.
Inclusion criteria
-
Acyanotic CHD with post tricuspid shunt and age < 5 months at enrollment
-
Modified Ross score > 6
-
On full breastfeeding
Exclusion criteria
-
Mother not available for breastfeeding
-
Infants with obstructive cardiac left to right shunt and complex cardiac conditions.
-
Infants who have undergone any cardiac surgery.
-
Prematurity < 37 weeks.
-
Known non-cardiac chronic conditions that are likely to affect growth including
-
“Malignancy, neuromuscular, respiratory, renal, gastrointestinal, immunodeficiency and metabolic, genetic, or other congenital anomalies”.
Definition of terms
Exclusive breastfeeding
Exclusive breastfeeding means giving a baby no other food or drink, including no water, in addition to breastfeeding (except medicines and vitamin or mineral drops; expressed breastmilk is also permitted).
Predominant breastfeeding
Breastfeeding a baby, but also giving small amounts of water or water-based drinks – such as tea, fruit juice, oral rehydration salt solution; drop and syrup forms of vitamins, minerals, and medicines, and ritual fluids (in limited quantities). Except for fruit juice and sugar water, no food-based fluid is allowed under this definition.
Full breastfeeding
Includes exclusive or predominant breastfeeding.
Direct breastfeeding
Feeding the infant directly from the mother’s breast. Giving expressed breast milk via spoon or a bottle is not considered direct breastfeeding.
Assessment of modified Ross score
Ross score is a clinical score commonly used to assess the severity of heart failure in children. The Ross score was assessed preferably while the infant was asleep at least 1 hour after the last feed. The Laer-modified Ross score was used in this study. Reference Ross18 Those with a score of >6 were eligible for enrollment.
Sample size calculation
Assuming a mean difference of 100 g in weight gain between the two groups as clinically significant, with an effect size of 0.67, alpha error of 0.05, and power of 80% the calculated sample size was 35 patients per group. However, due to the circumstances related to the COVID pandemic, only 25 patients per group could be recruited during the period of the study. After enrollment, each infant was randomised between the intervention group and control group by allocation of random numbers by computer software.
Control group
The mothers were counselled to give the baby at least 8 feeds per 24 hours by direct breastfeeding. Giving expressed breast milk was not allowed.
Intervention group
The mothers were counselled to give at least 8 feeds per 24 hours. A minimum pre-determined volume of expressed breast milk was targeted to be given by spoon during the day. The total volume of feed required in the intervention group was calculated with a target of 100 kcal/kg/day. For this, the breast milk was assumed to contain 67 Kcal/100 ml. Mothers were counselled to provide 30–50% of the total target calories by expressed breast milk with spoon- feeding. A range of volume was calculated for each infant (which was to be given by spoon-feed) according to the weight. Mothers were advised to give the amount of milk by spoon over 24 hours. Ad-lib breastfeeding over and above the minimum target was allowed to be given.
In both groups, the demographic data, history, and physical examination (weight, length, and occipitofrontal circumference were recorded). Weight was measured before a scheduled feed using the same digital weighing machine and recorded to the nearest second decimal. Weight was measured with the child wearing a dry diaper. The weight of the diaper was subtracted from the measured weight giving the infant’s actual weight. Echocardiographic parameters were recorded in a standard proforma.
After enrollment, infants were called for a follow-up at 2 weeks and 4 weeks, at which the anthropometric measures were recorded. Data regarding any illness in the intervening period were recorded and compliance with the protocol was noted. Counselling regarding the technique of breastfeeding was done both at the time of enrollment and at each follow-up visit. Weight gain (in grams) at 15 and 30 days as well as weight gain velocity (in g/kg/day) were compared between the two groups.
Data analysis
The presentation of the categorical variables was done in the form of numbers and percentages (%). On the other hand, the quantitative data were presented as the means ± SD.
The data normality was checked by using the Kolmogorov–Smirnov test. In cases where the data were abnormal, we used nonparametric tests. The following statistical tests were applied for the results: The comparison of the variables which were quantitative and not normally distributed in nature were analysed using the Mann–Whitney test (for two groups), and the Independent t-test was used for the comparison of normally distributed data between two groups. The comparison of the qualitative variables was analysed using the Chi-square test. If any cell had an expected value of less than 5, then Fisher’s exact test was used.
The data entry was done in the Microsoft Excel spreadsheet, and the final analysis was done with the use of Statistical Package for Social Sciences (SPSS) software, IBM manufacturer, Chicago, USA, ver 25.0. For statistical significance, a p-value of less than 0.05 was considered statistically significant.
Results and observations
In total, 50 infants with CHD and post-tricuspid left to right shunt were included and randomised into two groups. Flow chart of patient enrollment is shown in Figure 1. Eight patients (4 each in both groups) had moderate to large Patent ductus arteriosus and the rest of the patients had ventricular septal defects.
Salient baseline demographic, clinical, and echocardiography parameters of the infants enrolled in the study are shown in Table 1. Both groups were comparable in terms of clinical and echo parameters at the time of enrollment with no significant difference between the two groups.
Table 1. Salient baseline demographic, clinical, and echocardiography parameters
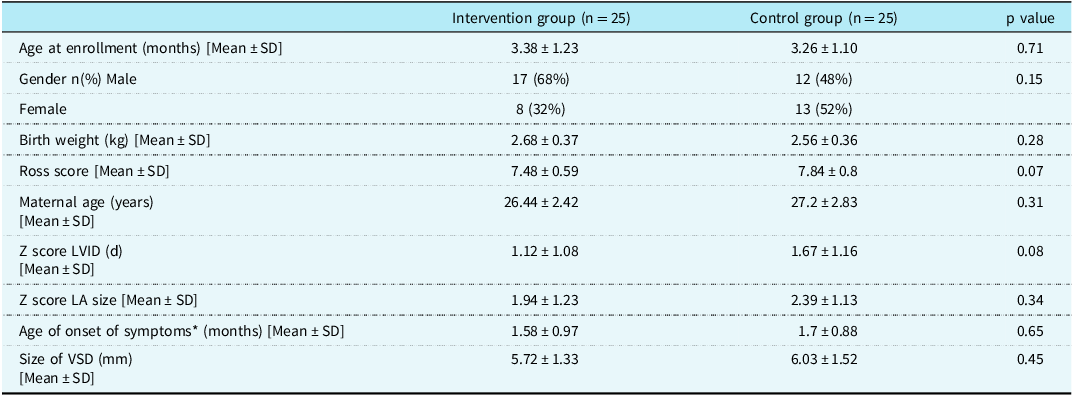
LVID (d) = Left ventricular internal dimension in diastole; LA = left atrium; VSD = ventricular septal defect.
*Age of onset of symptoms,is the age of the infant when symptoms related to heart disease were first noted by the parents.
The data regarding weight gain and follow-up details are given in Table 2.
Table 2. Showing weight gain and follow-up data
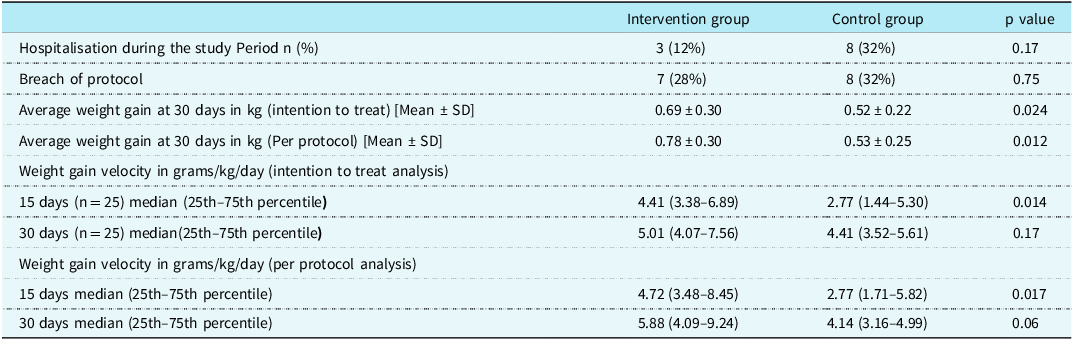
Average weight gain at 30 days was significantly higher in the intervention group (supplemental spoon-feeding along with breastfeeding) as compared to the controls (direct breastfeeding alone) as shown in Figure 2. Weight gain velocity in the two groups (g/kg/day) was significantly greater in the intervention group compared to controls at 15 days. At 30 days, although there was a higher velocity of weight gain in the intervention group compared to controls, the difference was not statistically significant.
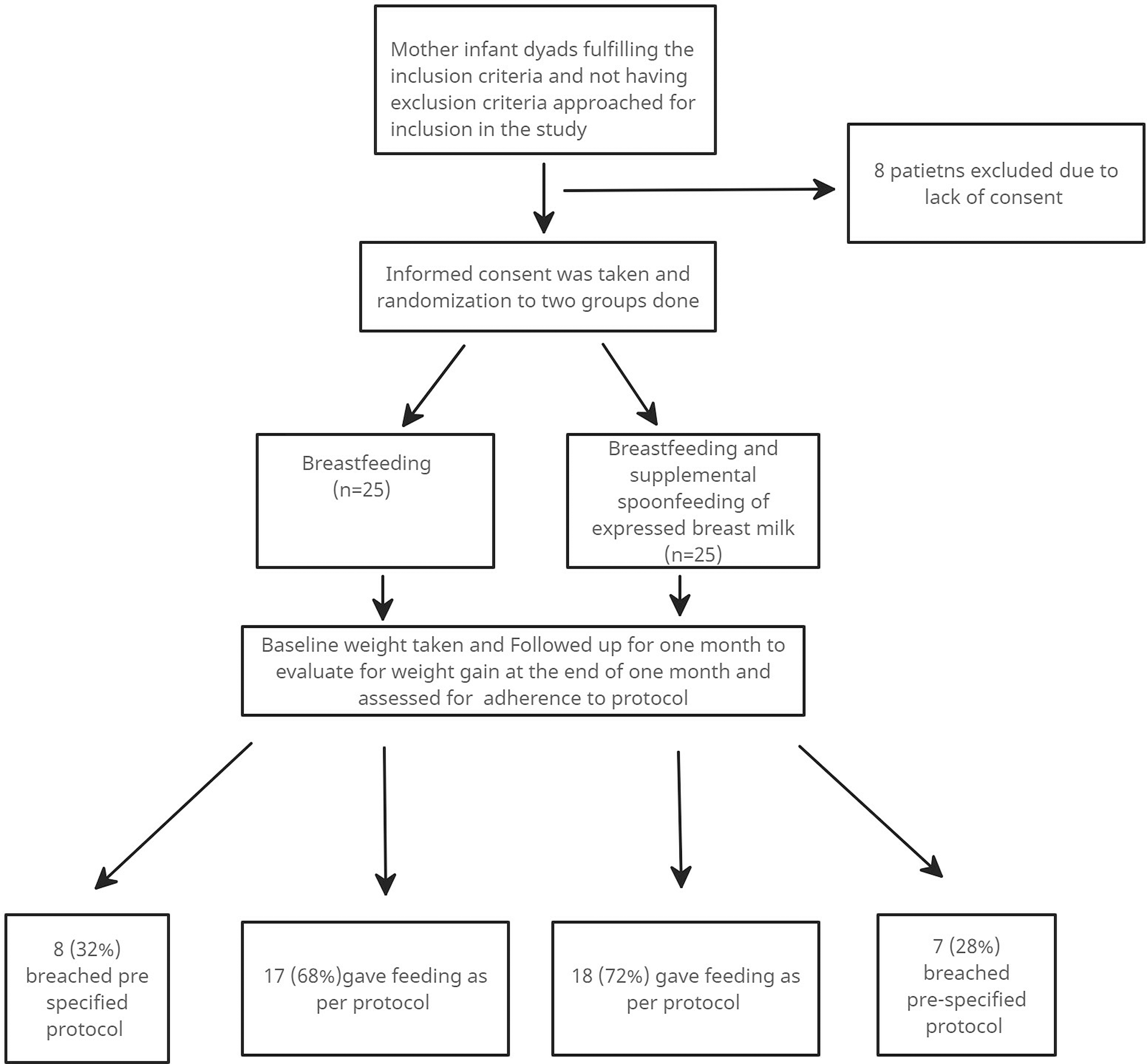
Figure 1. Flow chart of enrollment of the patients in the study.

Figure 2. A comparison of weight gain at 1 month (kg) between the intervention group.
In the “spoon feeding” group, there were seven infants who breached the pre-defined protocol. Four out of seven breaches of protocol were related to vomiting and maternal fear related to aspiration. In the “breastfeeding only” group, there were eight infants who had breached protocol. Three out of these eight infants were given formula milk by spoon due to maternal perception of the inadequacy of breast milk output.
Three infants in the “spoon-feeding” group and five infants in the “breastfeeding only” group had a breach of protocol when they were hospitalised due to intercurrent illnesses and were given formula feed.
Parents were advised to adhere to their respective feeding protocols during hospitalisation; however, the final decision regarding this was taken by the physicians who were not related to the study. None of the infants could adhere to their feeding protocol during hospitalisation.
Discussion
In this study, it was found that the difference in weight gain at one-month follow-up was significantly greater in infants with ventricular septal defect who were given expressed breast milk with a spoon along with breastfeeding compared to those who were given breastfeeding alone.
Several studies have shown breastfeeding to be well tolerated by infants with CHD. Reference Marino, O’Brien and LoRe19–Reference Torowicz, Seelhorst, Froh and Spatz22 However, there is limited scientific data regarding the adequacy of direct breastfeeding for promoting growth in unoperated children with CHD and left-to-right shunts. The resting daily energy expenditure in infants with CHD is increased compared to normal infants. Reference Barton, Hindmarsh, Scrimgeour, Rennie and Preece23 Therefore, some healthcare providers suggest spoon-feeding expressed breast milk to infants with unoperated CHD with the assumption that breastfeeding will require more energy than spoon-feeding. Though we did not measure the energy expenditure in this study, we assessed the effectiveness of supplemental spoon-feeding of expressed breast milk in improving weight gain and found that it could be a useful way of increasing weight in infants with left to right shunts.
Previous publications have focused on the use of energy-dense formula feed given by bottle or nasogastric tube for improving weight gain in children with unoperated CHD. Reference Xu, Lin, Zhang, Zheng and Lin7,Reference El-Koofy, Mahmoud and Fattouh9,Reference Lin, Lin, Lin, Zhang, Chen and Zheng11,Reference Zhang, Lin, Lin, Cao and Chen12,Reference Marino, Johnson and Davies15-Reference Zhang, Zhou, Chen, Cao and Chen17 This might be difficult to implement in low- and middle-income countries because of the cost of formula feeding. There is also an attendant risk of transmitting infection by bottle feeding if it is not done in hygienic conditions. This is especially relevant for resource-limited settings where due to unhygienic conditions, the milk as well as the bottle may be contaminated by bacterial pathogens. Reference Suthienkul, Siripanichgon, Promachot, Echeverria, Lexsomboon and Rakue24–Reference Marege, Regassa, Seid, Tadesse, Siraj and Manilal26
Although some previous studies have suggested that spoon-feeding may have a useful role in feeding infants who are unable to breastfeed, these studies have been done exclusively in premature infants. Reference Hoover27–Reference Aytekin and Albayrak30 The method of feeding expressed breast milk by spoon can be easily implemented even in resource-limited settings and therefore potentially useful for infants with left to right shunts who are prone to suffer from failure to thrive and poor weight gain.
Infants in the intervention arm had higher average weight gain at the end of 30 days compared to the control arm of the study both in the intention to treat as well as per protocol analysis. The weight gain at one month, seen in the intervention group of this study (780 gm) was slightly lower than that seen in a recent study in Chinese infants with unrestrictive ventricular septal defects (900 gm) by Lin et al. The authors had given bottle feeding of breast milk supplemented with human milk fortifier in their intervention arm.
The weight gain velocity (in g/kg/day) was also higher in the intervention group compared to the control group at 15 days. At 30 days, the weight gain velocity continued to be higher in the intervention group; however, the difference from the control groups was statistically insignificant.
The lack of statistically significant weight gain velocity seen at 30 days could be due to the small sample size of this study. We could not enroll an adequate number of patients during the time frame of the study due to the COVID-19 pandemic. Another reason for this could be due to difficulty in adhering to protocols, especially during intercurrent illnesses. The p-value for the difference in weight gain velocity in the intention to treat analysis was 0.06. It is possible that with better adherence, a higher number of patients, and a longer follow-up, the weight gain velocity might have also been significantly different between the two groups.
There are many difficulties faced by a caregiver while feeding children with CHD. Of these, intercurrent illnesses are the most prominent. 28% of infants in the intervention group and 32% of controls could not adhere to their predefined feeding protocols. Most often, the deviation from protocols was during hospitalisation due to intercurrent illnesses. Hospitalisation for intercurrent illness during the study period was reported in 22% of patients (12% for those receiving supplemental spoon-feeding v/s 32% for those receiving breastfeeding alone) which was largely on account of lower respiratory tract infection and congestive heart failure.
Apart from intercurrent illnesses, there were some instances where maternal fears and perceptions related to feeding led to a breach of protocol. 4 out of 7 breaches of protocol in the spoon-feeding group were related to vomiting during spoon-feeding and maternal fear regarding aspiration. 3 out of 8 breaches of protocol in the breastfeeding group alone were due to maternal perception of the inadequacy of breast milk.
Lactational counselling in this study was provided by a paediatric postgraduate student both at the start of the study and after two weeks. It is known that lactation counselling by professional counsellors improves breastfeeding initiation rates in mothers. Reference Patel and Patel31 It is possible that counselling by a professional lactation counsellor could have improved adherence to protocol in this study. However, professional lactation counselling is usually not available in our setting and primary care paediatricians usually assume this role in day-to-day practice, as was done in this study.
There are several limitations of this study. First, this was a small study with an inadequate sample size and a short follow-up. Larger multicentric studies with adequate sample sizes and longer follow-ups need to be done before concluding that supplemental spoon-feeding of expressed breast milk is effective in increasing weight gain in children with CHD. Second, adherence to protocol was not directly assessed by the investigators but was judged based on interviews of mothers at 2 and 4 weeks.
Finally, an arbitrary value of 100 kcal/day was used for calculating the total spoon-feeding volume as it was impossible to achieve higher calories without substantially increasing the volume of feeds or making feeds energy dense with supplements.
Conclusion
The findings from our study suggest that in infants with CHD and post tricuspid left to right shunts, supplemental feeding of expressed breast milk by spoon along with breastfeeding leads to better weight gain compared to breastfeeding alone.
There is a need to perform larger, adequately powered studies with longer follow-ups to define the role of spoon-feeding of expressed breast milk in improving weight gain in infants with CHD.
Acknowledgements
None.
Financial support
None.
Competing interests
None.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national guidelines on human experimentation (please name) and with the Helsinki Declaration of 1975, as revised in 2008, and has been approved by the institutional committee of RML Hospital on 24/12/2020 (F.No. TP md/MD) (77/2020/IEC/ABVIMS/RMLH/343) and registered in the clinical trial registry of India (CTRI/2021/01/030713).







