Epidemiological studies have pointed to diets rich in whole-grain (WG) cereal foods reducing the risk of many diet-related diseases, including CVD(Reference McKeown, Meigs and Liu1, Reference Mellen, Walsh and Herrington2), diabetes(Reference De Munter, Hu and Spiegelman3), obesity, the metabolic syndrome(Reference Sahyoun, Jacques and Zhang4) and some cancers(Reference Larsson, Giovannucci and Bergkvist5), with similar results being found across many populations. The mechanisms that have been proposed to underlie the reduction in disease risk revolve around the fibre and phytochemical content of WG compared with their refined counterparts(Reference Slavin, Jacobs and Marquart6, Reference Fardet7). In the published literature, a number of interventions based on WG cereal diets have found that WG lower plasma total and LDL-cholesterol(Reference Leinonen, Poutanen and Mykkanen8–Reference Giacco, Clemente and Cipriano11), blood pressure(Reference Behall, Scholfield and Hallfrisch12–Reference Tighe, Duthie and Vaughan14), improve insulin parameters(Reference Leinonen, Liukkonen and Poutanen15–Reference Pereira, Jacobs and Pins17) and improve markers of bowel function(Reference McIntosh, Noakes and Royle18, Reference Costabile, Klinder and Fava19). In most cases, these studies have been carried out on people already at risk of disease (e.g. overweight/obesity, hypercholesterolaemia, hyperinsulinaemia). Often these intervention studies have used specially prepared foods based on a single cereal grain (e.g. rye or barley), rather than WG foods that are readily available for consumers, and endpoints generally only span standard blood clinical chemistry measurements. Additionally, human intervention studies have not always found clear results from feeding WG cereals on markers of CVD risk(Reference Melanson, Angelopoulos and Nguyen20–Reference Brownlee, Moore and Chatfield22), which has led to some discussion about the acute impact of a WG diet(Reference McKeown and Jacobs23).
In addition to variable results from intervention studies, mechanisms of how a WG diet may improve health have also been unclear. While dietary fibre may explain many potential benefits of WG(Reference Slavin24), epidemiological research has often found benefits for CVD even when correcting for fibre intake, suggesting that other components in WG may also play a role(Reference Jacobs and Gallaher25). Of recent interest is the role of glycine betaine (betaine), a methyl donor found in high concentrations in WG wheat- and rye-based foods(Reference Waggle, Lambert and Miller26, Reference Bruce, Guy and Rezzi27). Supply of a higher amount of betaine may be an additional mechanism for the reduction in CVD risk due to WG intake, primarily via its role in the conversion of the cardiovascular risk factor homocysteine to methionine(Reference Vos28, Reference Konstantinova, Tell and Vollset29).
One of the goals of dietary recommendations for WG cereals is to get healthy people to replace servings of refined grains (RG; cereal grains with the bran and germ removed by milling) with WG products in order to reduce the risk of CVD(30), though few intervention studies have specifically compared the effect of matched WG foods with RG foods in healthy subjects(Reference Tighe, Duthie and Vaughan14, Reference Andersson, Tengblad and Karlstrom21). In order to better understand some of the possible mechanisms behind the potential benefits of replacing RG with WG foods, we designed a controlled, exploratory study to compare the short-term impact of a diet rich in commercially available WG cereal products with a diet rich in RG cereal products on parameters related to cardiovascular and gastrointestinal health in healthy subjects.
Materials and methods
Study design
The study was designed as a cross-over trial to compare the effect of a diet rich in WG with a diet rich in RG. After a 1-week lead-in where subjects were asked not to consume any WG foods (controlled using diaries and plasma alkylresorcinols(Reference Ross, Pineau and Kochhar31)) in order to have a similar baseline with regard to WG intake, subjects were randomly assigned to either a WG or RG diet for 2 weeks. This diet was controlled with all foods being supplied by the study centre. After the 2-week intervention period, subjects were asked not to consume any WG foods (as for the lead-in) for 2 weeks in order to monitor how long any changes due to the intervention diet lasted (post-intervention period). After a washout period of 5–7 weeks between the end of the post-intervention period and the start of the next lead-in, the subjects repeated the same protocol, but ate the other diet during the intervention period (Fig. 1). At the start of the study and at 1-week intervals during the following 5 weeks of the study, blood and 24 h urine samples were collected (total of six time points). Additionally, spot faecal collections (first stool passed during the day) were carried out at baseline and after the 2-week intervention diet period. Because changes due to the diets may not be evident in fasting plasma samples, a postprandial substudy was carried out. On day 8 of both intervention diets, subjects consumed either a WG breakfast cereal (shredded wheat) or a RG breakfast cereal (cornflakes), appropriate to the diet they were currently following. Subjects gave blood samples at 0, 60, 120, 180 and 240 min after the breakfast meal (Fig. 1). The present study was conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human subjects were approved by the Ethical Committee of the Lausanne Region, Vaud Canton, Switzerland. Written informed consent was obtained from all subjects. The study was carried out from February 2008 to December 2008.

Fig. 1 Study design. During the first intervention diet period, subjects ate either a diet rich in whole-grain (WG) cereals or a diet rich in refined-grain (RG) cereals. During the second intervention diet period, they ate the diet they had not eaten during the previous period. ‘PI diet’ is the 2-week post-intervention diet when subjects were asked not to consume any whole grains. ▲, Sampling points; ‘P’, when the postprandial sampling took place on day 8 of each 14 d dietary intervention.
Subjects
A total of twenty-two subjects were recruited to the study. Written informed consent was obtained from each subject, and selection took place following a medical questionnaire and confirmation of adherence to inclusion/exclusion criteria (healthy (normal blood lipid panel, BMI 19–28 kg/m2, no chronic or recent illness, no recent use of antibiotics or medication and non-smokers), between 20 and 50 years old and low habitual WG intake ( < 30 g/d, as determined by an FFQ(Reference Ross, Pineau and Kochhar31))).
Study foods
Foods used in the study were all commercially available from Nestlé SA worldwide or purchased from supermarkets (Lausanne, Switzerland and Huskvarna, Sweden). WG, energy, protein, fat, carbohydrate and fibre content were determined from the declared ingredient and nutrient compositions. The WG component of foods was assumed to have all three major anatomical fractions of the cereal grain (bran, germ and endosperm) in the same proportions found naturally. Cereal foods were also analysed for alkylresorcinol(Reference Ross and Kochhar32) and betaine(Reference Bruce, Guy and Rezzi27) content.
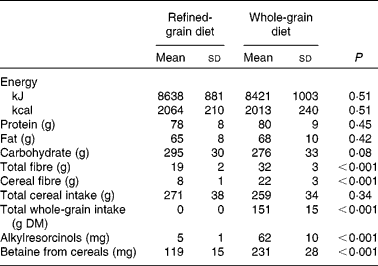
WG and RG menus were designed to supply a similar total energy intake, using cereal-based foods as the main source of carbohydrates (Table 1). Target daily energy intake was 8·5 MJ (2032 kcal) for the RG diet and 8·8 MJ (2107 kcal) for the WG diet. Bread (WG or RG wheat depending on the diet) was supplied with lunch and dinner, and was used as a discretionary food to make it easier for subjects to eat more or less depending on their individual energy requirements. Seven different daily menus were devised for each of the WG and RG diets, with each menu being repeated twice over the 2-week intervention diet period. A wide range of different WG and RG foods were used in the study to help avoid subject fatigue with the diet. It was not possible to obtain exact equivalent WG v. RG cereal foods as RG equivalents were not readily available (e.g. WG oats v. RG oats), but efforts were made to match equivalent cereals where possible. Overall, the WG diet contained 64 % WG wheat, 13 % WG oats and 9 % brown rice, with the rest from barley and rye, while the RG diet contained 66 % refined wheat, 27 % white rice and 8 % refined maize. The average prescribed daily WG intake during the WG diet was 150 g WG/d on a dry weight basis. The average prescribed total cereal intake was 277 g for the RG diet and 275 g for the WG diet (WG and RG ingredients combined, also dry weight).
Table 1 Sample menu*

* Cereal-based products at each occasion were either based on whole-grain or refined-grain cereal ingredients. Frozen meals contained either pasta or rice as their carbohydrate component. The 7 d menu was fed twice for each intervention arm.
† Foods that were available as either whole grain or refined grain.
During the intervention phases, meals were either eaten in a dining room at the metabolic unit of the Nestlé Research Centre, or provided pre-packaged for preparation at home. Compliance to the lead-in and post-intervention WG-free diets, and the intervention diet was monitored using food diaries and monitoring whether plasma alkylresorcinol concentrations remained within the expected range for a WG-free diet. Subjects were asked to note down any possible WG foods they ate during these periods. During the intervention phases, subjects were also asked to note the amount of all non-water beverages consumed, as well as any extra foods eaten and physical activity. Any food, including bread, remaining from the intervention diet was weighed and recorded to determine exact food intake. Beverages, extra food and remaining study foods were used for the quantification of overall nutrient intake during the study.
Nutrient intake during the intervention diet periods was estimated using a nutrient composition database program (Genesis; ESHA Research, Salem, OR, USA) using data from the Swiss nutrient composition tables.
Analytical methods
Plasma
Plasma was separated from blood by centrifugation at 1000 g for 10 min at 4°C. Plasma total, HDL- and LDL-cholesterol, TAG, glucose and C-reactive protein (CRP) were analysed using enzymatic kits in an XPAND autoanalyser (kits and instrument from Dade Behring, Düdningen, Switzerland). Homocysteine was analysed by HPLC using a kit (Bio-Rad AG, Reinach, Switzerland)(Reference Barbé, Abdelmouttaleb and Chango33). Betaine was analysed by isotope dilution liquid chromatography–MS/MS(Reference Bruce, Guy and Rezzi27). Alkylresorcinols were extracted by liquid–liquid extraction, purified by mixed-mode anion-exchange solid-phase extraction and analysed by GC–MS according to the method of Landberg et al. (Reference Landberg, Åman and Kamal-Eldin34).
Faeces
Faecal samples were collected into Stomacher bags (Seward Limited, Worthing, West Sussex, UK), and kept at 4°C before collection by the study centre, where they were stored at − 40°C. Faecal dry weight was determined after freeze-drying for 48 h. Faecal water was produced according to the method of Klinder et al. (Reference Klinder, Karlsson and Clune35), where two times (w/v) of distilled water (with 3 mm-sodium azide to prevent microbial growth) was added to the sample in a Stomacher bag and mixed using a Stomacher mixer (Seward Limited) for 2 min. If homogenisation was not complete, this was performed manually, and the samples were remixed using the Stomacher mixer. A 20 ml aliquot was removed into an ultracentrifuge tube, and tubes were centrifuged at 35 000 g for 120 min at 4°C. The supernatant was divided into aliquots for further analysis. Faecal water pH was determined using a pH probe.
Wet faeces were divided into aliquots before processing for faecal water and frozen at − 80°C before microbial analysis by quantitative PCR. Details of this procedure and probes used are provided in the supplementary material available at http://www.journals.cambridge.org/bjn. Total bacteria, Bacteroides, Bifidobacterium, Clostridium coccoides, Clostridium leptum, Enterobacteria, Enterococcus and Lactobacillus groups were quantified. These groups represent the phyla that make up the majority of the gut microbiota. During the intervention diet periods, subjects were asked each day to note down how many times they had passed stools. These data over 14 d were used to determine stool frequency (stools/d) over each dietary period.
Statistical analyses
All outcomes were analysed using ANCOVA in a mixed model setting where diet effects were examined using diet, period, baseline and other covariates as fixed effects and subject as a random effect. No carry-over effect was assumed. For the analysis of faecal bacterial counts, those below the detection limit were set to the detection limit (106 colony-forming units/g) for the purpose of analysis. Bacterial counts were then log-transformed and analysed using ANCOVA as above. In addition, ANOVA was used with repeated measures in order to assess the effect of diets over time.
All analyses were conducted on the seventeen completed subjects only. Furthermore, no correction of significant level was employed to adjust for the multiple testing of data because of the exploratory nature of the hypotheses studied. Therefore, unadjusted P values are reported, and the results are considered significant if P < 0·05 (trends reported if P < 0·2).
Statistical analyses were performed using NCSS for Windows 2007 (Kaysville, UT, USA) and SAS (version 9.1; SAS Institute, Cary, NC, USA).
Results
Subjects
Of the twenty-two subjects recruited to the study, five subjects withdrew prematurely and seventeen subjects completed the study. One subject dropped out due to an inability to comply with the intervention diet (consumed < 50 % of their estimated daily energy requirement), one subject dropped out due to an unrelated illness and three other subjects dropped out due to unexpected travel commitments during the study. Baseline (after the lead-in period) plasma lipid, glucose, CRP, homocysteine, betaine and alkylresorcinol concentrations did not differ between the two different arms of the study (Table 2).
Table 2 Baseline characteristics of subjects who completed the study
(Mean values with their standard errors)

RG, refined grain; WG, whole grain; CRP, C-reactive protein.
Food intake and compliance
The average energy intake was 8·4 MJ/d (2013 kcal/d) for the WG diet and 8·6 MJ/d (2064 kcal/d) for the RG diet (Table 3). There was no difference in overall energy, protein, fat or carbohydrate intake between the two diets. However, total fibre, cereal fibre, alkylresorcinol and cereal betaine intake were higher during the WG diet (all P < 0·001). The average recorded WG intake during the WG diet was 151 g/d. No subjects reported eating WG foods during the lead-in or post-intervention periods, nor during the RG diet. Total plasma alkylresorcinols for all subjects were below 82 (median 24) nmol/l during the lead-in and post-intervention periods with the exception of one subject. This subject had an alkylresorcinol concentration of 238 nmol/l one week after the RG diet, suggesting that they had recently consumed WG wheat before the sampling. Overall, the alkylresorcinol concentrations during the lead-in and post-intervention diet periods were not significantly different from the RG diet. This indicates that all subjects successfully avoided WG wheat and rye as requested with the exception of one subject at one time point. The C17 : 0-to-C21 : 0 homologue ratio, which can distinguish between a wheat- and a rye-based diet(Reference Linko-Parvinen, Landberg and Tikkanen36), was consistent with the consumption of wheat-based products.
Table 3 Actual nutrient intake during the two intervention diet periods
(Mean values and standard deviations)
Plasma biomarkers
After 1 week, plasma TAG were higher on the WG diet compared with the RG diet (1·09 v. 0·89 nmol/l; P = 0·05). After 2 weeks on the WG diet, there was a trend for a decrease in total cholesterol by 3 % and LDL-cholesterol by 9 % compared with the RG diet (both P = 0·09), while there was no longer any difference between the two diets for plasma TAG. There was no change in plasma HDL-cholesterol, glucose, CRP or homocysteine at 1 or 2 weeks after starting the WG diet, compared with equivalent time points for the RG diet (Table 4). Plasma betaine was elevated after 1 week of the WG diet (P = 0·01), and concentrations had dropped by 2 weeks, with a less marked difference between the two diets (P = 0·08). Mean baseline concentration of plasma alkylresorcinols, biomarkers of WG wheat and rye intake, was 46 (median 34; range 6–144) nmol/l, which was expected as the subjects were recruited as low-WG consumers. Plasma alkylresorcinols were increased by an average of 14·5 times after 1 week of the WG diet, remaining at approximately the same concentration after 2 weeks (both P < 0·001). During the RG diet, plasma alkylresorcinols were moderately increased to 44–47 nmol/l (P < 0·05 compared with after the lead-in and post-intervention periods), due to the high amount of refined-wheat products contained in the diet. Plasma alkylresorcinol concentrations returned to baseline values after 1 week of the post-intervention period (P>0·6). The mean of total alkylresorcinol concentrations during the post-intervention period was 28 (median 24; range 7–82) nmol/l (excluding one outlier), while the mean of total alkylresorcinol concentrations during the RG diet was 45 (median 41; range 25–98) nmol/l. Alkylresorcinol concentrations returned to baseline values after 1 week of the WG-free, post-intervention diet (after lead-in, mean 23 (median 15; range 7–67) nmol/l; 1 week post-intervention, mean 38 (median 32; range 14–79) nmol/l; 2 weeks post-intervention, mean 30 (median 26; range 8–82) nmol/l (P < 0·01)), demonstrating that there is no clear carry-over effect on plasma alkylresorcinols 1 week after stopping eating WG.
Table 4 Plasma clinical chemistry measurements and the effect of the whole-grain (WG) diet compared with the refined-grain (RG) diet
(Mean values with their standard errors and 95 % confidence intervals)

* Corrected for baseline. Statistical analyses were corrected for treatment order, age, sex, BMI and baseline concentrations.
† P = 0·05.
‡ P = 0·01.
§ P < 0·0001.
∥ P < 0·1.
During the postprandial challenge, there were no differences between the two diets for TAG or glucose (Figs. 2 and 3, respectively). After the WG breakfast, plasma betaine was elevated compared with the RG breakfast at comparative time points (Fig. 4). Betaine was increased by 18 % from baseline at 120 min after the WG meal (P < 0·01). Betaine was also increased from baseline at 60 min after the RG meal (P < 0·05), even though the betaine concentration in maize is low(Reference Bruce, Guy and Rezzi27). Plasma alkylresorcinols increased moderately from fasting concentrations, and were always different from the same time points during the RG meal (Fig. 5). Although energy intake was similar during both diets, there was a trend for the WG diet to lead to 0·5 kg weight loss over the 2-week intervention period, compared with the RG diet (P = 0·19).

Fig. 2 Plasma TAG concentrations monitored for 240 min after a whole-grain breakfast (shredded wheat) or a refined-grain breakfast (cornflakes). The interventions were taken against a background of a diet rich in whole grains or refined grains. Values are means, with standard errors represented by vertical bars. ♦, Refined-grain diet; ![]() , whole-grain diet.
, whole-grain diet.

Fig. 3 Plasma glucose concentrations monitored for 240 min after a whole-grain breakfast (shredded wheat) or a refined-grain breakfast (cornflakes). The interventions were taken against a background of a diet rich in whole grains or refined-grains. Values are means, with standard errors represented by vertical bars. ♦, Refined-grain diet; ![]() , whole-grain diet.
, whole-grain diet.

Fig. 4 Plasma betaine concentrations monitored for 240 min after a whole-grain breakfast (shredded wheat) or a refined-grain breakfast (cornflakes). The interventions were taken against a background of a diet rich in whole grains or refined grains. Values are means, with standard errors represented by vertical bars. a,b,y,z Time points within the same diet with unlike letters were significantly different (P < 0·05). All time points between diets were significantly different (P < 0·05). ♦, Refined-grain diet; ![]() , whole-grain diet.
, whole-grain diet.

Fig. 5 Plasma alkylresorcinol concentrations monitored for 240 min after a whole-grain breakfast (shredded wheat) or a refined-grain breakfast (cornflakes). The interventions were taken against a background of a diet rich in whole grains or refined grains. Values are means, with standard errors represented by vertical bars. a,b,c Time points within the same diet with unlike letters were significantly different (P < 0·05). All time points between diets were significantly different (P < 0·001). ♦, Refined-grain diet; ![]() , whole-grain diet.
, whole-grain diet.
Faecal parameters
There was no overall change in the faecal microbiota population, with the exception of the C. leptum group, which was slightly increased after the WG diet (P = 0·02), and a trend for an increase in Enterococcus spp. (P = 0·06) (Table 5). Inter-subject variability was large for total bacteria and all bacterial species analysed. There was a trend for a decrease in faecal water pH on the WG cereal diet compared with the RG diet (7·14 v. 7·38; P = 0·063 for the difference in change from baseline), while stool frequency was increased (1·40 v. 0·96 stools/d; P < 0·0001). Two subjects noted that they had problems with constipation during the RG diet.
Table 5 Faecal microbiota population measurements during the two dietary interventions, and the effect of the whole-grain (WG) diet compared with the refined-grain (RG) diet*
(Mean values with their standard errors and 95 % confidence intervals; log10 colony-forming units/g fresh weight)
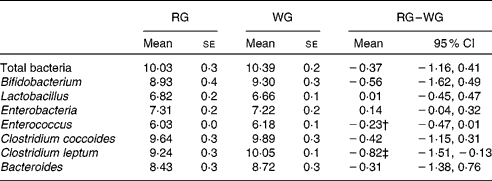
* Faecal samples were spot collections of the first bowel movement of the day. Statistical analyses performed on log-transformed data were corrected for baseline bacterial population counts, treatment order, age, sex and BMI.
† P = 0·056.
‡ P = 0·023.
Discussion
WG cereals are recommended as an important part of a healthy diet, and at least three servings (48 g)/d are specifically recommended in the Dietary Guidelines for Americans 2005, with up to 6 oz (170 g) recommended each day(30), while at least 75 g/10 MJ are recommended for adults in Denmark(37). In the present study, we designed a diet that used cereal grains as the main source of carbohydrates for each meal in order to see likely responses to a WG cereal diet compared with a RG diet over a relatively short time period (2 weeks). The two diets were well matched for overall energy content and macronutrient composition, and there was no difference between actual macronutrient intakes between the two diets, with the exception of fibre. The fibre content of the WG cereal diet, 34 g/d, is at the upper range of Swiss nutritional recommendations, while the RG diet (19 g/d) was the same as the normal intake among the French-speaking Swiss population (M. Vidal et al., unpublished results), so the RG diet cannot be defined as being especially low in fibre. The intake of other WG-related nutrients measured, betaine and alkylresorcinols, was at the upper range of estimated intakes from cereal foods(Reference Slow, Donaggio and Cressey38, Reference Ross, Becker and Chen39).
Some studies have found that a WG-rich diet does decrease plasma total and LDL-cholesterol(Reference Behall, Scholfield and Hallfrisch9, Reference Giacco, Clemente and Cipriano11, Reference Maki, Beiseigel and Jonnalagadda40), and in the context of the short duration of the intervention, a trend for a decrease in both total and LDL-cholesterol is remarkable, especially as all subjects were apparently healthy and normocholesterolaemic at baseline. The possible reason why some well-executed studies have not found an effect of WG on plasma cholesterol is the type of control used. For example, a WG diet has been compared with a low-WG diet(Reference Andersson, Tengblad and Karlstrom21) or subjects have added WG foods to their diet rather than replacing RG foods as recommended(Reference Brownlee, Moore and Chatfield22, Reference McKeown and Jacobs23). These results point to the use of a well-defined RG control diet being important for studying the effects of a WG diet. The trend for a reduction in plasma cholesterol in the present study lends further support to the repeated findings in epidemiological studies that diets high in WG cereals compared with those low in WG cereals are protective against CVD(Reference Mellen, Walsh and Herrington2).
No differences between diets were seen for plasma TAG, glucose, CRP or homocysteine. While some studies have found an effect of WG foods on these markers such as CRP(Reference Katcher, Legro and Kunselman41), it is possible that WG foods have a limited or no effect in subjects where the biomarkers are at normal concentrations in healthy subjects. In the case of glucose, specific challenge tests may be needed to observe the effects on glucose metabolism, as for a recent study which has found that a WG-based evening meal can improve glucose metabolism the morning afterwards(Reference Priebe, Wang and Weening42).
Some WG cereals, notably wheat and rye, are good sources of dietary betaine(Reference Bruce, Guy and Rezzi27), and it has been suggested to be one of the beneficial components of WG foods(Reference Vos28), yet apparently little research has been done on the effect of a WG cereal-rich diet on plasma betaine concentrations. Betaine is a methyl donor that is reported to have many metabolic functions(Reference Craig43), most notably as an osmolyte and as a donor of a methyl group in the conversion of homocysteine to methionine. Betaine has been found to lower homocysteine in humans(Reference Olthof, van and Boelsma44), though estimated dietary betaine intake was not associated with lower plasma homocysteine in women in an epidemiological setting(Reference Chiuve, Giovannucci and Hankinson45). Higher concentrations of plasma betaine were associated with lower non-HDL-cholesterol, TAG, BMI, body fat percentage, waist circumference(Reference Konstantinova, Tell and Vollset29) and markers of inflammation(Reference Detopoulou, Panagiotakos and Antonopoulou46), though betaine intake has not been associated with decreased CVD(Reference Dalmeijer, Olthof and Verhoef47). This highlights the importance of distinguishing between greater betaine intake and elevated plasma betaine, as only the latter has been associated with potential benefits. Elevated plasma homocysteine has been associated with increased CVD risk and increased markers of CVD risk (e.g. raised blood pressure and elevated serum lipids(Reference McNulty, Pentieva and Hoey48, Reference Refsum, Nurk and Smith49)), and association with reduced risk CVD has been found with WG cereal intake(Reference Mellen, Walsh and Herrington2, Reference Tighe, Duthie and Vaughan14). The elevation of plasma betaine after 1 week in the present study was relatively small, though none of the subjects had noticeably low betaine concentrations at baseline ( < 18 μmol/l)(Reference Allen, Stabler and Lindenbaum50), and the RG diet was not notably low in betaine or free choline (the direct metabolic precursor of betaine)(Reference Bruce, Guy and Rezzi27). Additionally, plasma betaine concentrations are regarded as being under strict metabolic control(Reference Ueland, Holm and Hustad51). Homocysteine concentrations were not affected during this intervention, though no subjects had notably elevated homocysteine concentrations (>15 μmol/l). During the postprandial challenge, plasma betaine, while being elevated compared with the RG diet at time 0, was further elevated after 120 min, suggesting a more important transient increase in plasma betaine, before plasma clearance. Betaine has previously been found to peak in plasma at 120–240 min post-dose after a high betaine meal(Reference Atkinson, Elmslie and Lever52), and at 40–80 min post-dose after feeding 1–6 g of pure betaine(Reference Schwab, Törrönen and Meririnne53). The estimated dose of betaine from the WG wheat breakfast cereal in the present study was 53 mg, about ten times less than that used in the test meal by Atkinson et al. (Reference Atkinson, Elmslie and Lever52) and 20–120 times less than the pure betaine used by Schwab et al. (Reference Schwab, Törrönen and Meririnne53), and more closely reflects the impact of a standard breakfast serving on plasma betaine. The differences found suggest that dose and matrix may have a small impact on pharmacokinetics. After 2 weeks, there was no difference between the two diets for plasma betaine. This may in part be explained by the fact that little of the body's betaine appears to be stored in plasma with rapid accumulation (up to twenty times that of plasma concentrations) in the liver and kidneys(Reference Ikeda, Miller and Sapienza54, Reference Slow, Lever and Chambers55). Previously, it has been reported that betaine may increase plasma LDL and TAG at the same time as decreasing plasma homocysteine(Reference Olthof, van Vliet and Verhoef56). This is suggested to be due to the increased supply of betaine, sparing choline for the production of VLDL particles, enabling better lipid transport from the liver and, consequently, a transient increase in plasma lipids(Reference Zeisel57). In the present study, the increase in betaine at 1 week was mirrored by an increase in TAG, with both dropping after 2 weeks. As there was no difference in lipid intake between the diets that would explain the TAG increase after 1 week, the possibility of a link with increased dietary betaine intake is intriguing.
Plasma alkylresorcinols have been proposed to be biomarkers of WG wheat and rye intake(Reference Ross, Kamal-Eldin and Åman58), with several studies now indicating a strong correlation between WG wheat and rye intake, and their presence in the plasma(Reference Ross, Pineau and Kochhar31, Reference Landberg, Kamal-Eldin and Andersson59). Not surprisingly in the present study, plasma alkylresorcinols were considerably elevated compared with the RG diet. As refined wheat still contains low concentrations of alkylresorcinols(Reference Ross and Kochhar32), low concentrations were still present in the plasma during the non-WG and RG diet periods. Presently, there are little data on the plasma concentrations of alkylresorcinols on a low/non-WG cereal diet, and results from the present study suggest that concentrations of total alkylresorcinols below 60 nmol/l probably indicate a diet not containing WG. Additionally, they confirm that plasma alkylresorcinols are responsive to WG wheat and rye in the diet, as indicated by the sharp increase in concentration on the WG intervention diet, though the amount of alkylresorcinols consumed in this diet is at the upper end of what would be consumed as part of a ‘normal’ diet(Reference Ross, Becker and Chen39).
During the postprandial study, there was a relatively small increase in plasma alkylresorcinols throughout the 240 min period measured. Only one study has looked at the pharmacokinetics of alkylresorcinols in human subjects, after a rye bran-based meal, and found that the T max occurs about 168 min(Reference Landberg, Linko and Kamal-Eldin60), whereas the concentration still appears to be increasing at 240 min in the present study. In a study on pigs habitually fed a rye bran-containing diet, plasma alkylresorcinols remained elevated between 120 and 360 min(Reference Linko, Ross and Kamal-Eldin61), and found that there was a marked difference in alkylresorcinol pharmacokinetics for habitual feeding and an acute meal. The data collected in the present study over 240 min would suggest that pharmacokinetics for alkylresorcinols in human subjects are also different if measured against the background of a habitual diet containing WG wheat or rye.
The trend for decreasing weight on the WG diet was unexpected, as most intervention studies addressing the question of weight loss or maintenance and WG diets have found no effect on total weight loss(Reference Melanson, Angelopoulos and Nguyen20, Reference Maki, Beiseigel and Jonnalagadda40, Reference Katcher, Legro and Kunselman41), though epidemiological evidence finds that WG intake is associated with lower BMI(Reference Harland and Garton62, Reference McKeown, Yoshida and Shea63), and one non-peer reviewed study has reported 0·9–1·4 kg weight loss on a WG diet compared with an energy-matched RG diet over 4–6 weeks(Reference Jacobs, Murtaugh and Pereira64). The subjects in the present study were all normal weight (BMI < 26 kg/m2) except for one subject who had a BMI of 29·6 kg/m2, making the trend for a 0·5 kg change in weight more remarkable.
Due to the inherently high fibre content of WG cereals compared with their refined counterparts, it has been hypothesised that WG cereals may play a role in modulating intestinal microbiota and alter intestinal transit. Some studies have found that WG wheat and/or rye diets alter faecal chemistry, increasing SCFA concentrations and decreasing faecal pH(Reference McIntosh, Noakes and Royle18). The increase in stool frequency was expected, and WG cereals have previously been demonstrated to be effective for improving stool frequency in constipated subjects(Reference Holma, Hongisto and Saxelin65). One study compared the effects of a WG wheat breakfast cereal with a wheat bran breakfast cereal over 3 weeks on faecal microbiota populations, and found that bifidobacteria and lactobacilli/enterococci populations increased with WG wheat(Reference Costabile, Klinder and Fava19), and a similar study has found that WG maize breakfast cereal increased faecal bifidobacteria populations after 3 weeks(Reference Carvalho-Wells, Helmolz and Nodet66). In the relatively short duration of the present study, there was only a weak effect (less than one log change) on the faecal microbiota, with a trend for an increase in Enterococcus, as for Costabile et al. (Reference Costabile, Klinder and Fava19) and a small increase in the C. leptum group. The C. leptum subpopulation is known to contain a number of bacterial species adapted for utilising fibre as a substrate and producing butyrate(Reference Shen, Zhang and Wei67). Abnous et al. (Reference Abnous, Brooks and Kwan68) found that C. leptum populations increased in rats fed either wheat or oat bran. Vitaglione et al. (Reference Vitaglione, Napolitano and Fogliano69) recently proposed that the phenolic compounds bound in the dietary fibre complex may also modify the intestinal environment which may play a role in the gut microbiota population, and clostridia are associated with the metabolism of phenolic compounds(Reference Selma, Espin and Tomas-Barberan70). The observed trend for a decrease in faecal water pH with the WG diet fits with the increase in butyrate-producing bacteria. The 2-week intervention period was probably too short to see a ‘full’ effect of the two diets, and most studies focusing on faecal microbiota last for at least 3 weeks(Reference Costabile, Klinder and Fava19). Nevertheless, the fact that small changes in faecal microbiota (and by implication, intestinal microbiota) were already evident after 2 weeks suggests that longer-term WG-based diets could have an effect on microbiota populations. Follow-up studies are required to investigate the long-term effects of a WG-rich diet on faecal microbiota populations.
The presence of many ‘trend’ results in the present study suggests that more significant results could have been obtained with a higher number of subjects. The daily amount of WG provided during the present study was high compared with usual intakes of about 16–25 g/d(Reference Cleveland, Moshfegh and Albertson71, Reference Maras, Newby and Bakun72) and minimum recommended intakes of 48–75 g(30, 37), but were well tolerated by the subjects, with no complaints noted about the WG component of the diets. This may be in part due to the fact that all WG foods were commercially available and so aimed at being palatable for the consumer rather than for use in a scientific study. The use of WG-based foods as the base for the carbohydrate component of a normal diet appears to be a good strategy for increasing WG intake, and appears to provide benefits for CVD risk markers, even in healthy subjects.
Most studies on the effects of WG cereals on biomarkers of disease have focused on specific types of grains (e.g. barley and brown rice(Reference Behall, Scholfield and Hallfrisch9, Reference Behall, Scholfield and Hallfrisch12, Reference Hallfrisch, Scholfield and Behall13), rye(Reference Leinonen, Poutanen and Mykkanen8, Reference Leinonen, Liukkonen and Poutanen15, Reference Juntunen, Laaksonen and Poutanen16, Reference Gråsten, Juntunen and Poutanen73), oats(Reference Maki, Beiseigel and Jonnalagadda40, Reference Pins, Geleva and Keenan74) and wheat(Reference Giacco, Clemente and Cipriano11)), while studies using a variety of grains have generally not found differences from the control(Reference Andersson, Tengblad and Karlstrom21, Reference Brownlee, Moore and Chatfield22). Those studies using a mixture of WG have attempted to replicate what could be possible with a ‘normal’ mixed diet, which has led to less control of WG intake during the control group/arm. In the present study, a mixed WG diet, containing mostly wheat-based products, found results on lowering LDL-cholesterol, a result that has been found previously for longer-term interventions based on barley and brown rice(Reference Behall, Scholfield and Hallfrisch9), oats(Reference Maki, Beiseigel and Jonnalagadda40, Reference Pins, Geleva and Keenan74) and wheat(Reference Giacco, Clemente and Cipriano11). It would appear then that the effect of WG cereals on lowering cholesterol does not necessarily need to be considered as associated specifically with those cereals high in soluble fibre (oats and barley), but could be ascribed to a diet that is generally high in WG cereals. While the reductionist approach naturally leads to the testing of individual cereals and cereal fractions to elucidate the key components responsible for observed effects, it is also important to conduct studies on mixed diets that are more likely to reflect what could be consumed on a regular basis and are likely to be easier for long-term consumption. The present study, while providing a high amount of WG/d, does demonstrate that a diet based on mixed commercially available WG products can lead to a reduction in LDL-cholesterol over a relatively short period of time.
Conclusions
The present study found a tendency for an effect of a WG diet on plasma cholesterol, further supporting the epidemiological evidence that WG cereals decrease the risk of CVD. The finding that plasma betaine is also elevated due to a WG diet presents new possibilities for mechanisms of how WG cereals may have protective effects(Reference Vos28), and adds weight to the hypothesis that the benefits of WG cereals are beyond being a source of fibre in the diet(Reference Fardet7). Future studies are needed to confirm that these results can be repeated with lower amounts of WG and over longer time periods.
Acknowledgements
A. B. R., D. G. and M. B. designed the study. A. B.-L., S. O.-A., M. B., A. B. and A. B. R. carried out the clinical part of the study. S. J. B., C. N.-M., M. V., A. B.-L., R. B., A.-C. P. and A. B. R. carried out chemical analyses; L.-B. F., S. K. and S. R. provided resources, and S. E.-A., D. G. and A. B. R. performed statistical analyses. All authors contributed to the final revision of the manuscript. The present study received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. The willing help of Fernand Beaud, colleagues from Nestlé worldwide and the Nestlé Research Center restaurant for supply of foods using the study, and participation of the subjects are gratefully acknowledged. All authors are employees of Nestlé SA, a part of a food company that produces a range of WG cereal products.












