Non-technical Summary
Tooth marks preserved on bones can yield a wealth of information regarding the behavior and ecology of the carnivores that produced them. For this reason, scientists studying both modern and fossil vertebrates have inspected and interpreted these features for decades. Although previous studies have provided important insights, they have also described tooth marks using sets of terms that have been incompletely defined, have incorporated behavioral hypotheses in definitions, and/or have been inconsistently applied. To address these problems, we introduce the category-modifier (CM) system, the first system to both sort tooth marks into clearly defined main categories and use descriptive modifiers to characterize their appearance more precisely. The CM system is designed to apply to a wide range of vertebrates, to enable comparisons across disciplines and studies, and to help researchers keep their investigations into behavioral hypotheses free of circular reasoning.
Introduction
Teeth are specialized structures used for defense, for combat, and for obtaining and processing food. The teeth of a carnivorous animal may make varying levels of contact with bone in the course of either predation or scavenging, and this type of contact may also occur during antagonistic or defensive biting. When one or more teeth damage, and potentially penetrate, the bone of a living or recently deceased vertebrate, evidence of this tooth-on-bone interaction is preserved in the form of a visible mark on the bone surface (Binford Reference Binford1981), known as a “tooth mark” or “bite mark” (Erickson and Olson Reference Erickson and Olson1996; Jacobsen Reference Jacobsen1998; D'Amore and Blumenschine Reference D'Amore and Blumenschine2009; Baquedano et al. Reference Baquedano, Domínguez-Rodrigo and Musiba2012; Drumheller and Brochu Reference Drumheller and Brochu2014). Such marks can yield a wealth of important behavioral and ecological information, which in the case of extinct vertebrates is especially difficult to obtain by other means. Numerous papers have assessed tooth marks on bones from both modern and fossil assemblages and have attributed them to a range of likely producers, including fish (e.g., Lebedev et al. Reference Lebedev, Mark-Kurik, Karatajūtė-Talimaa, Lukševičs and Ivanov2009; Collareta et al. Reference Collareta, Lambert, Landini, Di Celma, Malinverno, Varas-Malca, Urbina and Bianucci2017; Cortés et al. Reference Cortés, De Gracia, Carrillo-Briceño, Aguirre-Fernández, Jaramillo, Benites-Palomino and Atencio-Araúz2019; Randle and Sansom Reference Randle and Sansom2019), mammals (e.g., Haynes Reference Haynes1980, Reference Haynes1983; Longrich and Ryan Reference Longrich and Ryan2010; Parkinson et al. Reference Parkinson, Plummer and Hartstone-Rose2015; Arriaza et al. Reference Arriaza, Aramendi, Maté-González, Yravedra and Stratford2021), crocodilians (Njau and Blumenschine Reference Njau and Blumenschine2006; Noto et al. Reference Noto, Main and Drumheller2012; Boyd et al. Reference Boyd, Drumheller and Gates2013; Drumheller and Brochu Reference Drumheller and Brochu2014, Reference Drumheller and Brochu2016; Njau and Gilbert Reference Njau and Gilbert2016), varanids (D'Amore and Blumenschine Reference D'Amore and Blumenschine2009, Reference D'Amore and Blumenschine2012), mosasaurs (Everhart Reference Everhart2008; Einarsson et al. Reference Einarsson, Lindgren, Kear and Siverson2010), and theropod dinosaurs (Currie and Jacobsen Reference Currie and Jacobsen1995; Erickson and Olson Reference Erickson and Olson1996; Jacobsen Reference Jacobsen1998; Hone and Rauhut Reference Hone and Rauhut2009; Bell and Currie Reference Bell and Currie2010; Hone and Watabe Reference Hone and Watabe2010; Hone et al. Reference Hone, Tanke and Brown2018; Drumheller et al. Reference Drumheller, McHugh, Kane, Riedel and D'Amore2020; Brown et al. Reference Brown, Tanke and Hone2021, Reference Brown, Currie and Therrien2022).
Unfortunately, comparisons of findings across studies, sometimes even within a given discipline or on a given taxonomic group, are often hampered by complications arising from irreconcilable terminological frameworks used by different authors. For example, two such terminological frameworks may confusingly apply some terms in ways that overlap but are not identical (Fig. 1), even when one framework was introduced as a modified version of the other or both represent independently modified versions of a “common ancestor” (Fig. 2). Although some authors have reframed and/or clarified the definitions put forth in previous studies (e.g., Selvaggio [Reference Selvaggio1994] and Andrews and Fernández-Jalvo [Reference Andrews and Fernández-Jalvo1997] redefined Binford's [Reference Binford1981] main categories), there are several of these modified frameworks in existence, and there is no guarantee that any given framework will be universally adopted even within a single discipline (Fig. 2). In some frameworks, the definitions of different mark categories are ambiguous and/or incorporate interpretations regarding mark formation, introducing an element of circularity into attempts to test hypotheses of feeding behavior (D'Amore and Blumenschine Reference D'Amore and Blumenschine2009; Hone and Watabe Reference Hone and Watabe2010; Njau and Gilbert Reference Njau and Gilbert2016; Table 1, Fig. 2). Others include categories of damage that are defined by co-occurrence of multiple types of tooth mark on a single bone and therefore apply to individual elements rather than individual marks. For example, Tanke and Currie (2006) defined Type 1 tooth markings as circular to oval punctures; Type 2 as transverse gouges, scores, or tooth drags; and Type 3 as small-scale parallel groupings of shallow furrows or striations. Type 4, however, was defined as co-occurrence of two or more of the preceding types on one bone, ruling out the possibility of assigning an individual tooth mark to Type 4.

Figure 1. Images from the literature, to illustrate the application of terms used in previous publications to tooth marks produced by various vertebrates. A, C, E, and G are the unaltered figures, and B, D, F, and H show the same figures with colored overlays indicating how the marks were characterized by the authors who originally described them, using blue to indicate marks characterized as essentially non-elongate (“pits” or “Type 1 marks”) and pink to indicate marks characterized as essentially elongate (“scores,” “Type 2 marks,” “drag marks,” or “bite-and-drag marks”). A and B show one Type 1 and two Type 2 marks on a right dentary of the theropod Sinraptor, thought to have been inflicted by another Sinraptor (modified from original images of IVPP 10600 used in Tanke and Currie [2000]). C and D show the medial surface of the deltopectoral crest of a Saurolophus left humerus bearing drag (black arrows) and bite-and-drag (gray arrows) marks thought to have been inflicted by Tarbosaurus (modified from original images of MPC-D100/764 used in Hone and Watabe [2010]). E and F show pits and scores on a goat femur, inflicted by Komodo monitors. G and H show the lateral surface of the distal part of a cow tibia, bearing pits (bisected pits are indicated by white arrows) and scores inflicted by Nile crocodiles (modified from Njau and Blumenschine [2006]). Scale bars, 10 mm. Terminologies used by previous studies are presented in Tables 1 and 2. Details of tooth-marked elements are provided in Supplementary Table 1. Numbering of marks corresponds to that used in Supplementary Tables 4 and 5.
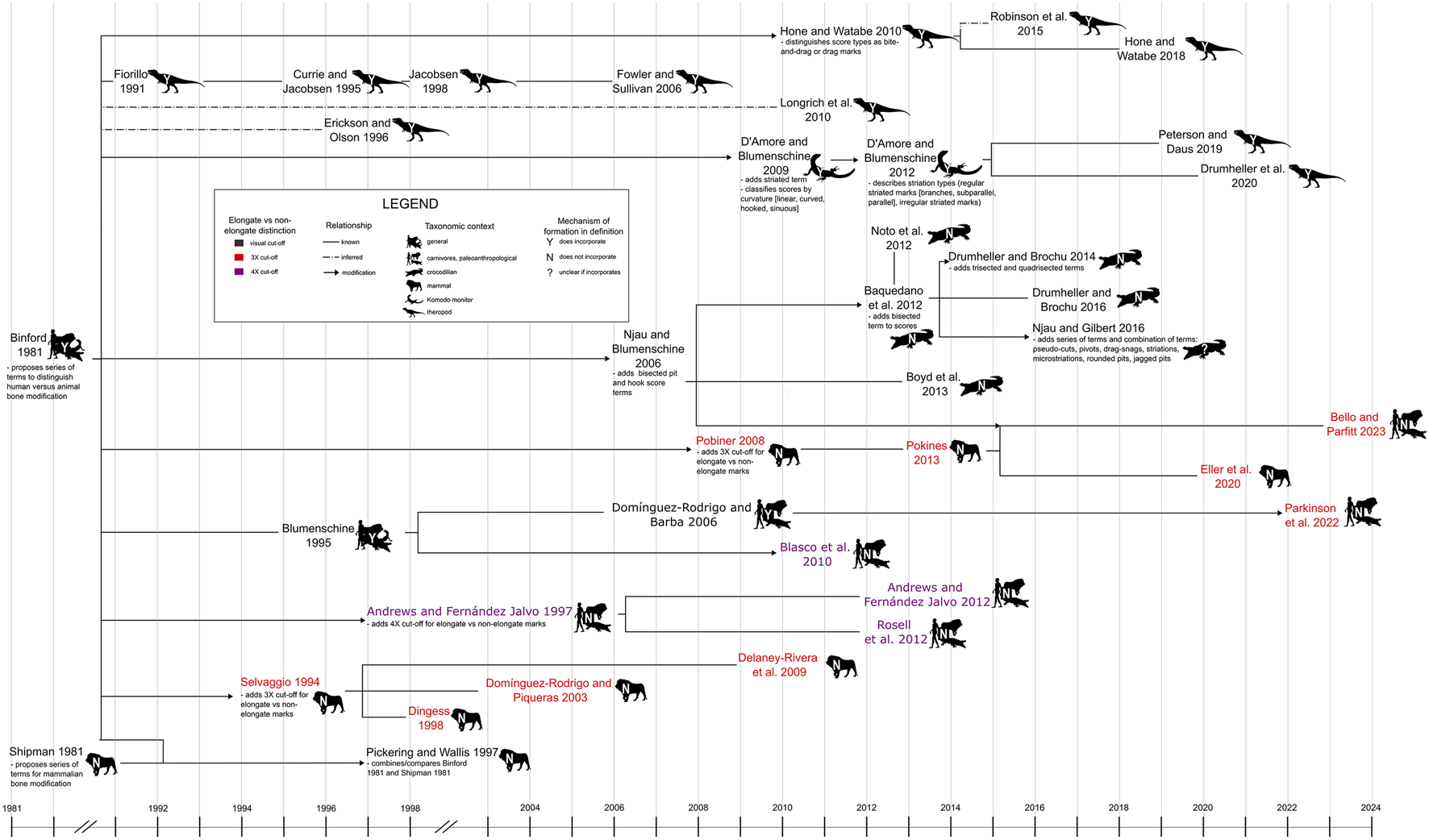
Figure 2. Relationships among terminological frameworks used in previous tooth mark studies, highlighting how different frameworks have differentiated between elongate and non-elongate types of mark and whether or not they have incorporated inferred mechanisms of tooth mark formation into the definitions of the terms used. References cited in figure: Binford Reference Binford1981; Shipman Reference Shipman1981; Fiorillo Reference Fiorillo1991; Selvaggio Reference Selvaggio1994; Blumenschine Reference Blumenschine1995; Currie and Jacobsen Reference Currie and Jacobsen1995; Dingess Reference Dingess1998; Erickson and Olson Reference Erickson and Olson1996; Jacobsen Reference Jacobsen1998; Andrews and Fernández Jalvo Reference Andrews and Fernández-Jalvo1997, Reference Andrews, Fernández-Jalvo and Bell2012; Pickering and Wallis Reference Pickering and Wallis1997; Dominguez-Rodrigo and Piqueras Reference Domı́nguez-Rodrigo and Piqueras2003; Domingues-Rodrigo and Barba Reference Domínguez-Rodrigo and Barba2006; Njau and Blumenschine Reference Njau and Blumenschine2006; Fowler and Sullivan Reference Domínguez-Rodrigo and Barba2006; Pobiner Reference Pobiner2008; D'Amore and Blumenschine Reference D'Amore and Blumenschine2009, Reference D'Amore and Blumenschine2012; Delaney-Rivera et al. Reference Delaney-Rivera, Plummer, Hodgson, Forrest, Hertel and Oliver2009; Blasco et al. Reference Blasco, Rosell, Arsuaga, Bermúdez de Castro and Carbonell2010; Hone and Watabe Reference Hone and Watabe2010, Reference Hone, Tanke and Brown2018; Longrich et al. Reference Longrich and Ryan2010; Baquendano et al. Reference Baquedano, Domínguez-Rodrigo and Musiba2012; Noto et al. Reference Noto, Main and Drumheller2012; Rosell et al. Reference Rosell, Cáceres, Blasco, Bennàsar, Bravo, Campeny and Esteban-Nadal2012; Boyd et al. Reference Boyd, Drumheller and Gates2013; Pokines Reference Pokines2013; Drumheller and Brochu Reference Drumheller and Brochu2014; Robinson et al. Reference Robinson, Jasinski, Sullivan, Sullivan and Lucas2015; Drumheller and Brochu Reference Drumheller and Brochu2016; Njau and Gilbert Reference Njau and Gilbert2016; Peterson and Daus Reference Peterson and Daus2019; Drumheller et al. Reference Drumheller, McHugh, Kane, Riedel and D'Amore2020; Eller at el. Reference Eller, Pobiner, Friend, Austin, Hofman and Sholts2020; Parkinson Reference Parkinson, Plummer, Oliver and Bishop2022; Bello and Parfitt Reference Bello and Parfitt2023. Original image credits for silhouettes: M. Michaud (lion), S. Traver (crocodilian, Komodo monitor), T. M. Keesey (human), and T. Dixon (theropod).
Table 1. Basic types of tooth mark identified in previous studies. Definitions enclosed in square brackets have been inferred based on descriptions and photographs.

Table 2. Additional descriptive terms and names for features used in selected tooth mark classification systems. For the basic categories used in the various systems, see Table 1. Definitions enclosed in square brackets have been inferred based on descriptions and photographs.
We aim to address these issues by presenting the first tooth mark classification system with clearly defined, hierarchically nested categories that can be combined with different modifiers to describe the curvature and texture of each mark. If widely adopted, this system will lead to greater consistency among studies. The category-modifier (CM) system presented here is also more flexible than the terminological frameworks currently used, because the former is able to accommodate varying levels of detail depending on the amount of information that researchers have at their disposal (e.g., photographs vs. three-dimensional data). The system can potentially be expanded to incorporate new terms, without altering the definitions of previously established ones. The CM system is designed to facilitate an improved understanding of the relationships among tooth morphology, behavior, and tooth-mark characteristics, and should be applicable across a range of disciplines.
Terminology Used
Here, we use the term “tooth mark,” rather than “bite mark,” to refer to any feature observed on a bone that is thought to have been produced by contact with a tooth. Although use of the word “mark” has been controversial in the past, Zonneveld et al. (Reference Zonneveld, Fiorillo, Hasiotis and Gingras2022) have recently argued that this is appropriate so long as a descriptive modifier (e.g., “tooth”) is applied. We consider “tooth mark” preferable to “bite mark,” because the former has looser implications regarding the origin of the feature in question. “Bite mark” implies that the feature was formed during biting, whereas “tooth mark” leaves open the possibility of formation during types of tooth–bone contact that are only arguably describable as biting, such as scraping and scratching (Brown et al. Reference Brown, Currie and Therrien2022).
Previous Terminological Frameworks
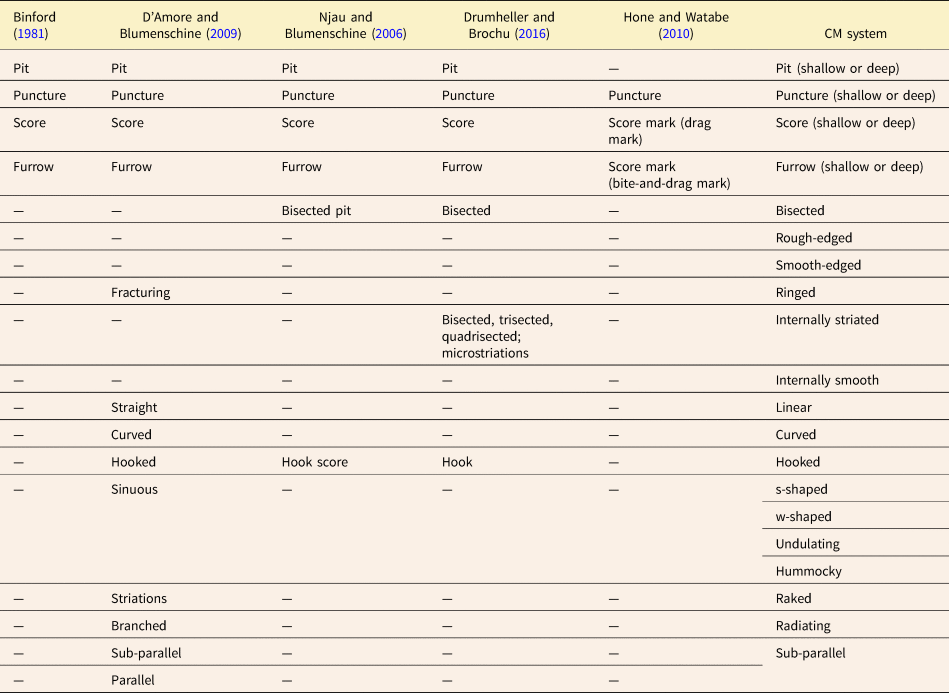
Binford's (Reference Binford1981) seminal paper distinguishing between carnivore-caused and human-caused patterns of bone modification introduced four main tooth mark categories (scores, pits, furrows, punctures) based on whether or not the tooth mark fully penetrated the cortical bone layer, and whether or not the mark appeared elongate. Since then, many tooth mark workers have used these same four fundamental categories in one form or another, often with clearer definitions and sometimes with different names. Some studies (e.g., Selvaggio Reference Selvaggio1994; Andrews and Fernández-Jalvo Reference Andrews and Fernández-Jalvo1997) clarified Binford's original categories by using some threshold value of the length-to-width ratio to quantitatively differentiate elongate marks (scores/furrows) from non-elongate marks (pits/punctures) (Table 1, Fig. 2). Others (e.g., Hone and Watabe Reference Hone and Watabe2010) used Binford's (Reference Binford1981) original definitions but also introduced subcategories to describe variations in mark appearance thought to reflect different mechanisms of formation. Others, such as D'Amore and Blumenschine (Reference D'Amore and Blumenschine2009) and Njau and Blumenschine (Reference Njau and Blumenschine2006), dealt with intracategory variation in a different way, by introducing modifiers that could be added to the original category names to create such combinations as “bisected pits” and “hook scores” (Table 2, Fig. 2).
Several sets of terms used by various authors to explicitly or implicitly classify tooth marks into main types are summarized in Table 1. Some authors have also included names for tooth mark subtypes, names for features associated with tooth marks, and/or additional descriptive terms (e.g., “hooked”) that can be applied to certain tooth marks (Table 2). For terms that were introduced by some authors (e.g., Binford Reference Binford1981) without being defined explicitly, we formulated the definitions appearing in Tables 1 and 2, based on whatever inferences about the intended meaning of each term could be drawn from the text and figures provided.
In some cases, authors appear to have used particular terms without offering explicit definitions because such definitions were not necessary to achieve their specific research goals. For instance, Binford (Reference Binford1981) aimed to distinguish between human and animal modification of bones, rather than to conduct an in-depth analysis of the types of tooth marks observed. Other studies have avoided classification systems altogether and instead taken a purely descriptive approach to documenting tooth marks, presenting qualitative observations without applying terms that could be interpreted as referring to distinct tooth mark types (Bell and Currie Reference Bell and Currie2010; DePalma et al. Reference DePalma, Burnham, Martin, Rothschild and Larson2013; Hone and Tanke Reference Hone and Tanke2015; Brown et al. Reference Brown, Tanke and Hone2021). Finally, some workers (Andrews and Fernández-Jalvo Reference Andrews and Fernández-Jalvo1997; Selvaggio and Wilder Reference Selvaggio and Wilder2001; D'Amore and Blumenschine Reference D'Amore and Blumenschine2009) have both classified tooth marks into types and supplemented this qualitative approach with measurements of mark dimensions such as length and width. However, some definitions used in previous studies incorporate hypotheses of mark formation and/or do not specify clear boundaries between certain mark types (Tables 1, 2, Fig. 2).
Proposed Classification Scheme
Tooth-Marked Specimens Used for CM System Construction
To provide an empirical basis for development of the CM system, we observed firsthand the morphology and appearance of 1570 tooth marks on 351 dinosaur elements (1551 tooth marks on 346 elements from subadult to adult individuals of large-bodied ornithischian taxa, 19 tooth marks on 5 elements from subadult to adult theropods). These specimens were collected from the Upper Cretaceous Aguja, Bearpaw, Dinosaur Park, Foremost, Frenchman, Hell Creek, Horseshoe Canyon, Judith River, Kirtland, Lance, Milk River, Oldman, Scollard, St. Mary River, Two Medicine, and Wapiti Formations of North America. The tooth marks were cast as silicone impressions, using Smooth-On Dragon Skin 10 Very Fast platinum-cure silicone, and the impressions are reposited at the University of Alberta and Royal Tyrrell Museum of Palaeontology. The CM system was developed first and foremost with reference to the variation we observed among the silicone impressions, but can also be applied to observations made directly from tooth marks on bones. To increase the applicability of the CM system across a wider range of vertebrate taxa, we also examined photographs and descriptions in the literature of tooth marks produced by mammals (Binford Reference Binford1981; Delaney-Rivera et al. Reference Delaney-Rivera, Plummer, Hodgson, Forrest, Hertel and Oliver2009; Longrich and Ryan Reference Longrich and Ryan2010; Young et al. Reference Young, Stillman, Smith and Korstjens2015; Koungoulos et al. Reference Koungoulos, Faulkner and Asmussen2018; Parkinson et al. Reference Parkinson, Plummer, Oliver and Bishop2022), crocodilians (Njau and Blumenschine Reference Njau and Blumenschine2006; Baquedano et al. Reference Baquedano, Domínguez-Rodrigo and Musiba2012; Noto et al. Reference Noto, Main and Drumheller2012; Boyd et al. Reference Boyd, Drumheller and Gates2013; Drumheller and Brochu Reference Drumheller and Brochu2014), mosasaurs (Everhart Reference Everhart2008; Einarsson et al. Reference Einarsson, Lindgren, Kear and Siverson2010), and sharks (Schwimmer et al. Reference Schwimmer, Stewart and Williams1997; Shimada and Hooks Reference Shimada and Hooks2004; Collareta et al. Reference Collareta, Lambert, Landini, Di Celma, Malinverno, Varas-Malca, Urbina and Bianucci2017; Shimada and Hanks Reference Shimada and Hanks2020). Some 1014 Komodo monitor tooth marks produced across 81 goat postcranial elements in a series of feeding trials (D'Amore and Blumenschine Reference D'Amore and Blumenschine2009) were also assessed through characterization of the tooth marks on the original bones using the CM system.
Measurements
The CM system is partially based on a set of measurements that may be taken directly from a tooth-marked specimen or from photos or silicone peels of a tooth mark (Fig. 3). As is also typical in more descriptively oriented studies (Bell and Currie Reference Bell and Currie2010; DePalma et al. Reference DePalma, Burnham, Martin, Rothschild and Larson2013; Hone and Tanke Reference Hone and Tanke2015; Brown et al. Reference Brown, Tanke and Hone2021), use of the CM system entails recording the length, width, and if possible, depth of each tooth mark. Rather than only giving these measurements for descriptive purposes, however, the CM system uses ratios among mark dimensions to assign tooth marks to categories (Table 3), as has been done in a handful of previous studies (Selvaggio Reference Selvaggio1994; Andrews and Fernández-Jalvo Reference Andrews and Fernández-Jalvo1997; Domı́nguez-Rodrigo and Pinqueras Reference Domı́nguez-Rodrigo and Piqueras2003; Parkinson et al. Reference Parkinson, Plummer, Oliver and Bishop2022). In the case of elongate tooth marks, defined as those having a length-to-width ratio of at least 3.0, orientation and curvature are also measured. All measurements except depth can easily be obtained from photos, but depth can only be measured from silicone peels or directly from specimens.

Figure 3. Measurements used in the category-modifier (CM) system to characterize tooth marks comprising a single arc (A–C) or comprising more than one arc (D–F). A, D, Length (l), width (w), and angle (a) of a tooth mark, all measured parallel to the bone surface. Tooth-mark angle is taken relative to the bone long axis (denoted by gray dotted line). B, E, Cross-sectional views denoted by the red plane in A and D, respectively, showing measurement of tooth mark depth perpendicular to the bone surface. C, Tooth mark curvature (c), determined from three points taken along the length of the mark: the two end points and the point of greatest inflection along the mark's length. F, Tooth mark curvatures (c1, c2), determined by dividing the mark into single curves using visual estimation.
Table 3. Classification of tooth marks into categories in the category-modifier (CM) system, based on tooth mark dimensions and on whether a mark fully penetrates the cortical bone. Bold text denotes primary categories, plain text denotes secondary categories, and italic text denotes tertiary categories. Measurements used in defining categories are provided in Fig. 3.

The tooth-mark measurements used in the CM system are defined as follows.
Length: greatest linear dimension, measured parallel to the bone surface;
Width: greatest value of the dimension parallel to the bone surface but perpendicular to length;
Depth: greatest value of the dimension perpendicular to the bone surface;
Horizontal orientation: angle between the line segment that defines length, and an anatomical reference axis such as the long axis of a tooth-marked limb bone; and
Curvature: angle defined by three points situated along one edge: the two ends of the edge, and the point of greatest curvature along the edge's length. When a mark is approximately uniformly curved along its length, the third point is placed at the midpoint of the mark. For marks comprising two or more curves with different orientations, the transition points between successive curves are determined by visual estimation.
Workflow and Application
Step 1: Assignment to Main Categories
Each mark can be assigned to a primary category based on the length-to-width ratio (Table 3, Fig. 4A, Fig. 5A). Following Selvaggio (Reference Selvaggio1994), a mark with a length-to-width ratio of at least 3.0 is considered “elongate,” while one with a ratio of less than 3.0 is considered “non-elongate.” Others, such as Andrews and Fernández-Jalvo (Reference Andrews and Fernández-Jalvo1997), have also used the length-to-width ratios of tooth marks to assign marks to elongate and non-elongate categories, but with a threshold of 4.0 (Blasco et al. Reference Blasco, Rosell, Arsuaga, Bermúdez de Castro and Carbonell2010; Andrews and Fernández-Jalvo Reference Andrews, Fernández-Jalvo and Bell2012; Rosell et al. Reference Rosell, Cáceres, Blasco, Bennàsar, Bravo, Campeny and Esteban-Nadal2012) (Fig. 2). We have adopted a threshold of 3.0 because this appears to be the most common choice in the literature (Selvaggio Reference Selvaggio1994; Dingess Reference Dingess1998; Domı́nguez-Rodrigo and Pinqueras Reference Domı́nguez-Rodrigo and Piqueras2003; Delaney-Rivera et al. Reference Delaney-Rivera, Plummer, Hodgson, Forrest, Hertel and Oliver2009; Eller et al. Reference Eller, Pobiner, Friend, Austin, Hofman and Sholts2020; Parkinson et al. Reference Parkinson, Plummer, Oliver and Bishop2022; Bello and Parfitt Reference Bello and Parfitt2023). However, it should be noted that 4.0 or any other threshold value can be used in the framework of the CM system, provided the threshold value is indicated and the lengths and widths of the marks under study are reported.

Figure 4. Proposed method of tooth-mark characterization via the category-modifier (CM) system. A, Stepwise progression from primary categories based on length and width, through secondary categories based on presence or absence of full cortical bone penetration, to tertiary categories based on depth. B, Modifiers used to describe external and internal tooth mark appearance, showing real examples observed in fossil specimens, with blue coloration indicating the texture described by the modifier: rough-edged (TMP 2014.015.0078), bisected (material used in Drumheller and Brochu [2014]), smooth-edged (UALVP 53140; also shows smooth interior), raked (TMP 1985.012.0026), ringed (TMP 1966.010.0106), internally chattered (UALVP 57561; also shows rough edge), internally striated (UALVP 57561), internally smooth (UALVP 49014; also shows smooth edge), mixed internal appearance showing both spongy and chattered textures (UALVP 60000; also shows rough edge), internally spongy (MOR 799; also shows smooth edge), and occupied (MOR 549; fibula in cross-section). C, Descriptive modifiers applied to elongate tooth marks based on number of arcs and arc curvature. Abbreviation: c = point of maximum curvature. Scale bar, 1 cm. Specimen information provided in Supplementary Table 1.

Figure 5. Worked example showing application of the category-modifier (CM) system to tooth marks on a single element (proximal end of the lateral surface of a hadrosaurid fibula; UALVP 57561). A, Assignment of tooth marks to primary categories based on length-to-width ratio (first part of “Step 1” described in text). B, Assignment to secondary categories based on whether or not the cortical bone layer has been fully penetrated (middle part of “Step 1” described in text). C, Assignment to tertiary categories based on width-to-depth ratio (last part of “Step 1” described in text). D, Application of external textural modifiers to identified tooth marks (“Step 2” described in text). E, Application of internal textural modifiers to identified tooth marks (“Step 2” described in text). F, Assignment of curvature modifiers to elongate tooth marks (“Step 3” described in text). Raw measurements and CM system categories are available in Supplementary Tables 2 and 3 for all numbered tooth marks shown. Abbreviation: NE = non-elongate.
As in previous studies, we consider tooth marks that penetrate through the cortical bone layer into the underlying cancellous bone to differ fundamentally from those that do not. Accordingly, tooth marks are assigned to secondary categories depending on whether they are “penetrating” or “non-penetrating.” Elongate tooth marks are termed “scores” if they are non-penetrating and “furrows” if they are penetrating, whereas non-elongate marks are termed “pits” if non-penetrating and “punctures” if penetrating. If depth measurements are available, then tooth marks can be further assigned to “deep” or “shallow” tertiary categories, a deep mark being defined as having a depth-to-width ratio of 2.0 or more (Table 3, Figs. 4, 5B). The 2.0 threshold used here is inevitably somewhat arbitrary, but considering depth independently of cortical bone penetration is advantageous, because cortical bone may vary widely in thickness among skeletal elements, among parts of a single skeletal element, and across taxa. A tooth mark made in thick cortical bone may be deep, but still classifiable as a score or pit (Table 3, Figs. 4, 5C). Conversely, a tooth mark penetrating a thin layer of cortical bone may be shallow, but classifiable as a furrow or puncture. However, a meaningful depth measurement cannot be obtained for a penetrating tooth mark if the tooth broke through the cortical bone into an internal cavity, rather than into cancellous bone. In such cases, a tertiary category assignment is not possible.
Although rare, bones containing embedded teeth have been reported in the paleontological literature (Currie and Jacobsen Reference Currie and Jacobsen1995; Farlow and Holtz Reference Farlow and Holtz2002; Buffetaut et al. Reference Buffetaut, Martill and Escuillié2004; Bell and Currie Reference Bell and Currie2010; Xing et al. Reference Xing, Bell, Currie, Shibata, Tseng and Dong2012), and presumably occur in modern settings as well. The indentation containing an embedded tooth will likely conform to the tooth's dimensions, and will then represent a non-elongate mark (a pit or puncture, depending on whether the cortex is penetrated) unless the tooth has a very high length-to-width ratio.
Step 2: Descriptive Modifiers Based on Mark Appearance
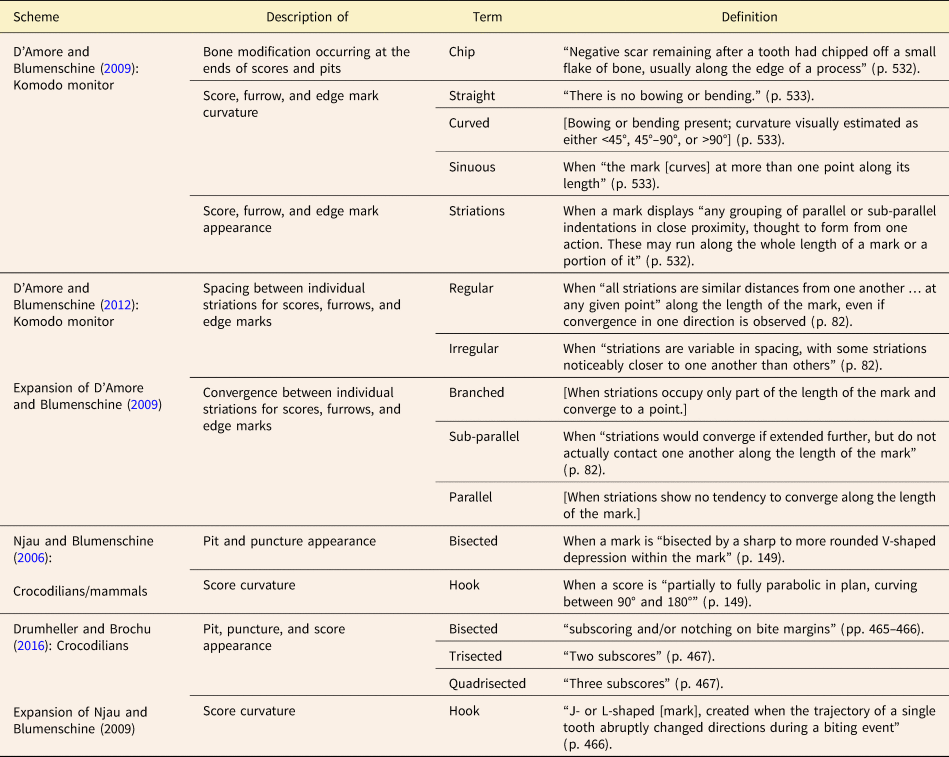
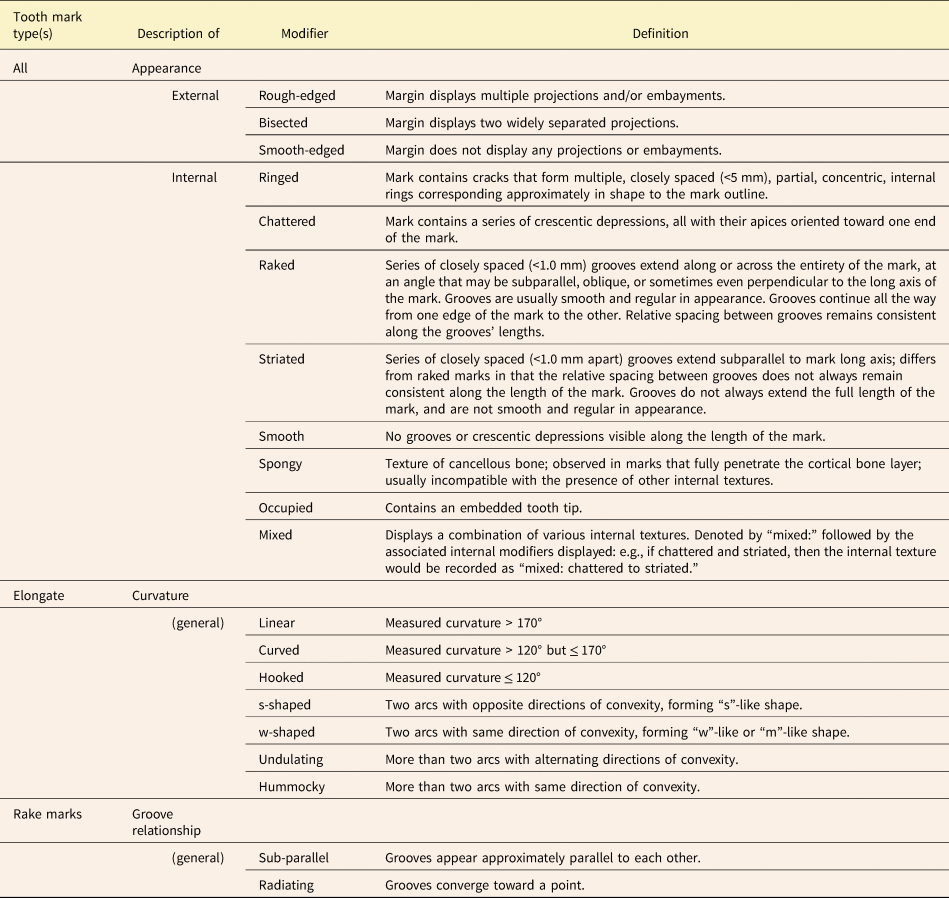
Once a mark has been assigned to primary, secondary, and if applicable, tertiary categories, descriptive modifiers can be applied to capture the physical appearance of the tooth mark in greater detail (Table 4, Figs. 4B and 5D, E). An elongate or non-elongate mark with jagged edges is described as “rough-edged,” while one with smooth edges is described as “smooth-edged.” A mark that has two notches projecting outward from its margins is described as “bisected.” Crocodilian tooth marks are often bisected, the notches being made by the tooth carinae (Njau and Blumenschine Reference Njau and Blumenschine2006; Drumheller and Brochu Reference Drumheller and Brochu2016; Njau and Gilbert Reference Njau and Gilbert2016; Drumheller et al. Reference Drumheller, D'Amore, Njau, Woodward and Farlow2023). Other descriptive modifiers pertain to the appearance of the interior of the tooth mark. If the carnivore's tooth tip has broken off inside the tooth mark and is therefore obscuring any internal texture, the mark is “occupied.” If the interior of a tooth mark is visible but completely lacks any patterning or other distinguishing characteristics, the mark is described as “smooth.” If a tooth mark contains a series of crescentic depressions, each oriented approximately transverse to the long axis of the tooth mark, it is described as “chattered.” A tooth mark that contains a series of partial, concentric, internal rings similar in shape to the tooth mark's overall outline is described as “ringed.” A “spongy” tooth mark is one floored by cancellous bone, so penetrating tooth marks are normally spongy unless the tooth broke through to an internal cavity. Tooth marks containing a series of closely spaced grooves (<1 mm spacing) that do not extend along the mark's entire length and/or do not collectively occupy the mark's entire width can be described as “striated.” However, a mark fully occupied by closely spaced grooves and lacking well-defined margins is described as having a “raked” morphology. The mean orientation of these grooves can be parallel, oblique, or perpendicular to the long axis of the overall mark. The grooving in a raked mark can be described as either “sub-parallel” or “radiating,” depending on whether the grooves appear approximately parallel to each other or diverge (Table 4). In some instances, tooth marks display different internal textures in different places, sometimes due to variations in depth along the tooth mark. Such instances are assigned the general modifier “mixed:” followed by the modifiers corresponding to the specific textures observed. For example, if a tooth mark is striated at one end and transitions to a smooth internal texture at the other, this would be recorded as “mixed: striated to smooth.”
Table 4. Descriptive modifiers that can be applied to individual tooth marks in the category-modifier (CM) system to describe the appearance of the margin, the appearance of the interior, and for elongate marks, the mark's curvature.
Step 3: Descriptive Modifiers Based on Curvature
Following D'Amore and Blumenschine (Reference D'Amore and Blumenschine2009), elongate marks with different degrees and types of curvature are distinguished. However, our curvature parameter is defined differently (see “Measurements”), in that it does not rely on establishing a “vertical” long axis for each mark for reference but rather on measuring angles based on end points and inflection points (Table 4, Figs. 4C, 5F). The CM system also distinguishes between elongate marks with multiple inflection points (“s-shaped,” “w-shaped,” or “undulating”) and those with only one inflection point (“straight,” “curved,” or “hooked”). Marks with a single inflection point are described as “straight” if the measured curvature is more than 170°, as “curved” if the measured curvature is between 170° and 120°, and as “hooked” if the measured curvature is less than 120°. Marks with two inflection points are described as “w-shaped” if the inflections are both in the same direction, and as “s-shaped” if the inflections are in opposite directions. Marks with more than two inflection points are described as “hummocky” if the inflections are all in the same direction, and as “undulating” if the inflection directions alternate.
Discussion
The main categories used in the CM system correspond conceptually to those in other classification systems, but are redefined using clear, consistent boundaries. For example, the CM system follows previous ones in using length-to-width ratios as a basis for assigning tooth marks to different categories (Fig. 2, Tables 1, 5). Like several other studies (Selvaggio Reference Selvaggio1994; Domı́nguez-Rodrigo and Pinqueras Reference Domı́nguez-Rodrigo and Piqueras2003; Parkinson et al. Reference Parkinson, Plummer, Oliver and Bishop2022; Bello and Parfitt Reference Bello and Parfitt2023), the present system uses an explicit threshold value of this ratio (3.0) to categorize tooth marks as either elongate or non-elongate. Once a tooth mark has been assigned to one of these primary categories, it can then be assigned to a secondary category based on whether the tooth penetrated all the way through the cortical bone, and to a tertiary category based on width-to-depth ratio (assuming a depth measurement is available). Descriptive modifiers can then be applied to the tooth mark. In previous systems (e.g., Bell and Currie Reference Bell and Currie2010; Hone and Watabe Reference Hone and Watabe2010), marks were assigned to one of a set of mutually exclusive categories, whereas the stepwise approach presented here includes three levels of categories (primary, secondary, and tertiary) plus a series of additional modifiers. The advantages of this proposed stepwise approach are summarized below.
Table 5. Comparison of previous terminologies used in the literature with that used in the category-modifier (CM) system.
Advantages of the CM System
Simple Category Definitions
An important feature of the CM system is the use of clear and simple criteria for defining tooth mark categories. Although a few previous studies have applied explicit categorization systems to tooth marks (Selvaggio Reference Selvaggio1994; Andrews and Fernández-Jalvo Reference Andrews and Fernández-Jalvo1997; Domı́nguez-Rodrigo and Pinqueras Reference Domı́nguez-Rodrigo and Piqueras2003; Parkinson et al. Reference Parkinson, Plummer, Oliver and Bishop2022), they have sometimes incorporated inferred modes of mark formation into the definition of terms used (Binford Reference Binford1981; Tanke and Currie Reference Tanke and Currie2000; D'Amore and Blumenschine Reference D'Amore and Blumenschine2009; Hone and Watabe Reference Hone and Watabe2010; Table 1, Fig. 2). These previous studies provided the terminological framework upon which the CM system was built (Table 5, Fig. 6), but CM system definitions are purely descriptive and are not based on inferred modes of tooth mark formation. With such definitions, different behavioral hypotheses can be tested without any danger of slipping into circular reasoning.

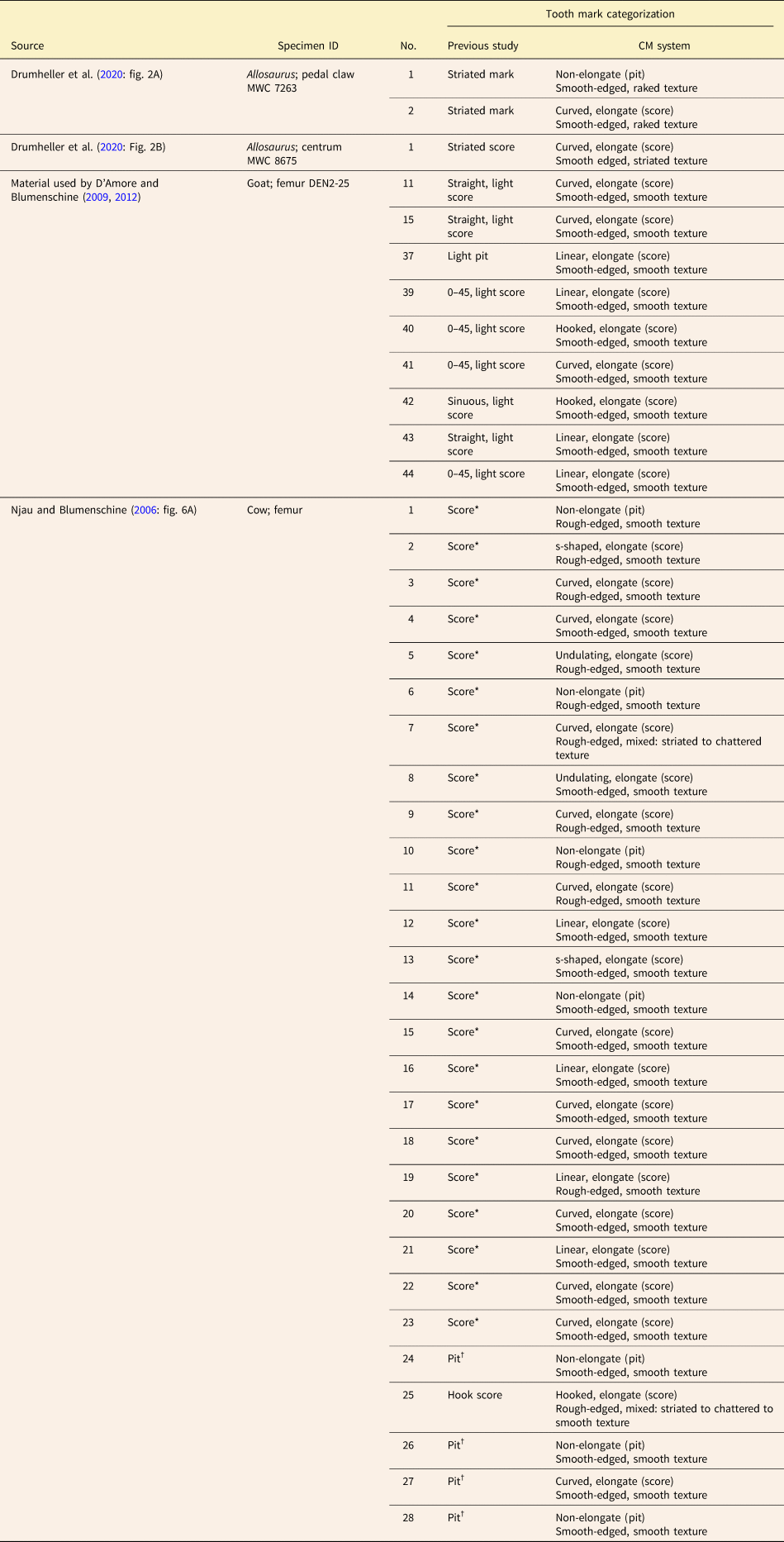
Figure 6. Drawings of tooth marks made by various vertebrates and illustrated in previous publications, comparing the categorizations given in the original sources (A, C, E, G) with those that would be assigned to the same marks under the category-modifier (CM) system (B, D, F, H). A, B, Striated tooth marks on an Allosaurus pedal claw (MWC 7263), thought to have been inflicted by another Allosaurus (modified from Drumheller et al. Reference Drumheller, McHugh, Kane, Riedel and D'Amore2020). C, D, Striated score on an Allosaurus centrum (MWC 8675), thought to have been inflicted by another Allosaurus (modified from Drumheller et al. Reference Drumheller, McHugh, Kane, Riedel and D'Amore2020). E, F, Pits and scores on a goat femur inflicted by Komodo monitors (specimen DEN2-25 used in D'Amore and Blumenschine [2009]). G, H, Pits, scores, hook score, and scores with internal striations on the medial surface of the shaft of a subadult cow femur inflicted by Nile crocodiles (modified from Njau and Blumenschine Reference Njau and Blumenschine2006). Scale bars, 10 mm. Information on tooth-marked elements provided in Supplementary Tables 4 and 5. Terminologies used by previous studies are presented in Tables 1 and 2. Specific categorizations are available in Table 6 for all numbered tooth marks shown.
Table 6. Original categorizations from the literature of the individual tooth marks shown in Fig. 6, compared with how the same tooth marks would be categorized under the category-modifier (CM) system. *Identification inferred from clear categorization of other, similar tooth marks in caption of figure cited; note that some of the pits listed could be considered bisected. †Description of mark type given in caption of cited figure or in accompanying text. Terminological frameworks used in previous studies are presented in Tables 1 and 2. Provenance information for tooth-marked elements is provided in Supplementary Tables 4 and 5.
Linnaean ichnotaxonomy represents a possible alternative to the CM system and other non-taxonomic methods of tooth mark categorization, and ichnotaxonomic names have occasionally been applied to tooth marks in the paleontological and neontological literature (e.g., Mikuláš et al. Reference Mikuláš, Kadlecová, Fejfar and Dvořák2006; LaBarge and Njau Reference LaBarge and Njau2024). However, we consider the ichnotaxonomic approach, at least in its current state of development, less suitable for classifying tooth marks, which present a pattern of morphological variation that in many respects is a multidimensional continuum. Erecting ichnotaxa to capture this variation would largely represent an exercise in drawing boundaries on this continuum, as in the CM system. However, the boundaries would be implied by diagnoses of individual ichnotaxa rather than laid out explicitly as part of a comprehensive framework. Furthermore, the ichnotaxa would have to be either broadly defined or numerous in order to encompass the same range of tooth mark morphology as the CM system. Finally, the use of ichnotaxonomic names would introduce a layer of unnecessary complexity to tooth mark research, as variation in tooth mark features is readily described using vernacular terms. This said, application of the CM system in conjunction with a redefined ichnotaxonomic framework (e.g., LaBarge and Njau Reference LaBarge and Njau2024) would be worth future exploration.
If the CM system is widely adopted, its explicitly defined tooth mark categories will also facilitate greater consistency across studies and even across disciplines, as there will be little room for subjective biases to influence the process of assigning marks to categories (Tables 5, 6). Without such definitions, for example, a researcher particularly interested in puncture-type marks might be inclined to assign an unwarranted number of marks to this category, and marks with moderate length-to-width ratios (e.g., 2.5–4.5) might be “eyeballed” as elongate by some researchers and non-elongate by others.
Measurements and Databases
The availability of an established system that uses simple measurements to define tooth mark categories may encourage researchers to publish the dimensions of any tooth marks they study, ensuring that the data are available to others in the future. Tooth mark measurements could then be amassed from the literature for use in surveys across time and space, as is often done for measurements of vertebrate skeletons. Large datasets of the kind that would result from this approach are needed to draw more comprehensive behavioral inferences than can be obtained from a handful of case studies and to reveal changes in behavior at the greatest temporal scales. Carnivorous theropods, for instance, likely fed in a different manner to modern mammalian carnivores, if only because of morphological differences in the teeth, jaws, and associated musculature.
Flexibility and Versatility of the CM System
The CM system is hierarchical, in that tooth marks are first assigned to a primary category (elongate or non-elongate) based on the ratio of length to width, then to a secondary category (penetrating or non-penetrating) based on the bone layer impacted, and finally to a tertiary category (deep or shallow) based on the ratio of width to depth. This stepwise approach allows data from two-dimensionally measured tooth marks (taken from photographs) and three-dimensionally measured tooth marks (taken directly from specimens, or from high-fidelity casts or peels) to be combined and compared at the primary classification level, and potentially also at the secondary level if images of sufficient quality are available. Thus, researchers who only have access to photographs of tooth marks that interest them will still be able to make partial comparisons to other marks that have been measured in three dimensions.
Incorporation of New Information
Finally, the hierarchical design of the CM system allows new categories and/or descriptors to be easily incorporated as needed. If a previously unknown type of tooth mark were to be discovered, for example, the system could be adapted to accommodate whatever category or descriptor was required to characterize the new tooth marks, without necessarily affecting the classification of previously known ones. Furthermore, the use of simple ratios between tooth mark dimensions to define primary and tertiary categories makes the system flexible and easy to revise. If boundaries between categories are adjusted in the future for expediency, it will be possible to straightforwardly and unambiguously reclassify previously described marks based on nothing more than their dimensions, which in many cases should be available in the literature. We advocate routine publication of tooth mark measurements, even in papers that do not use the CM system, so that the measurements will be available to researchers who may wish to either add the tooth marks to datasets that are based on the CM system or make use of tooth mark dimensions in some other way.
Beyond the CM System
The scope of the CM system is limited to categorization and formalized description of individual tooth marks based on their morphology. However, the ichnological study of tooth marks on bones potentially encompasses further interpretive steps, including recognition of tooth mark sets, identification of behaviors that potentially produced observed tooth marks, and identification of possible tooth mark makers. Part of the purpose of the CM system is to facilitate such interpretive work by capturing morphological information about tooth marks in a standardized form that is simple, intuitive, and easy to understand.
Descriptions of tooth-marked bones (e.g., Rogers et al. Reference Rogers, Krause and Curry Rogers2003; Hone and Watabe Reference Hone and Watabe2010; Longrich and Ryan Reference Longrich and Ryan2010; Paik et al. Reference Paik, Kim, Lim, Huh and Lee2011) have often reported sets of two or more approximately parallel, closely spaced marks, with the implication that the marks belonging to a given set may have been formed by multiple teeth during a single jaw movement. Recognition of such tooth mark sets or “serial bite marks” (sensu Njau and Blumenschine Reference Njau and Blumenschine2006) opens up the possibility of using the characteristics of a tooth mark set, considered as a whole, to aid in tooth mark maker identification. Rogers et al. (Reference Rogers, Krause and Curry Rogers2003), for example, found that a set of four parallel marks on a haemal arch of the theropod Majungasaurus crenatissimus were spaced at intervals comparable to those between the teeth of a conspecific skull from the same locality, suggesting cannibalistic behavior in M. crenatissimus. Brown et al. (Reference Brown, Tanke and Hone2021) used orientation and relative positioning of the tooth marks on a hadrosaurid ungual, which were arranged in multiple semi-parallel rows, to infer possible theropod gnawing behavior. Although Brown et al. (Reference Brown, Tanke and Hone2021) did not use a quantitative method to assign the tooth marks to different sets, their study illustrates the properties that should be present in marks that are considered to form a series: similar orientation, relatively consistent spacing, and proximity. In principle, it might be desirable to establish explicit criteria for assigning tooth marks to sets, perhaps involving limits on the linear distance and angular deviation among tooth marks belonging to a putative set. If, however, tooth mark sets are understood as groups of tooth marks likely to have been made in a single action, then the ultimate criterion for testing the validity of a putative set is whether one can postulate a motion by which a plausible tooth mark maker could readily have produced all the marks in the set. Addressing this issue for any given specimen will require some kind of analysis based on the dental anatomy of candidate tooth mark makers and how their dentitions would have contacted the surface of the tooth-marked bone in different kinematic scenarios, taking into account the complications posed by the possibility of missing teeth. If one or more tooth positions were vacant when tooth–bone contact took place, the resulting gap(s) in the tooth mark set may make it appear that the marks were produced by a series of similarly directed jaw motions, when in reality they were produced by a single such motion. For all these reasons, the CM system does not attempt to introduce general guidelines for recognizing tooth mark sets, and workers will presumably continue to identify sets largely on an informal, qualitative basis, as in past studies (e.g., Fowler and Sullivan Reference Fowler, Sullivan, Lucas and Sullivan2006; D'Amore and Blumenschine Reference D'Amore and Blumenschine2009; Hone and Watabe Reference Hone and Watabe2010; Brown et al. Reference Brown, Tanke and Hone2021). However, the CM system may nevertheless play a role in tooth mark set analyses, by providing a framework for consistent descriptions of individual marks that can be compared within and across putative sets.
Identification of sets of tooth marks likely to have been produced in a single motion is a special case of the more general problem of determining how tooth marks were generated by the actions of a carnivore. Tooth mark proportions can be informative in this respect, in that a very high length-to-width ratio implies extensive movement of the tooth parallel to the surface of the bone, whereas a very low ratio implies the opposite. Tooth mark curvature and observable features of a tooth mark's margins and interior may also prove helpful in inferring the pattern of craniomandibular kinematics that produced a given tooth mark. Depth may also indicate the approximate level of force that the tooth mark maker must have exerted (Erickson et al. Reference Erickson, Kirk, Su, Levenston, Caler and Carter1996; Gignac et al. Reference Gignac, Makovicky, Erickson and Walsh2010). Both of these relationships need to be more fully explored. Once again, such inferences are too dependent on particulars of both the tooth-marked bone and possible tooth mark makers for formulation of an explicit set of general guidelines to be feasible, but the CM system should facilitate description of tooth marks in a clear, consistent way that will provide a foundation for subsequent kinematic and dynamic interpretations. The fact that the CM system classifies and describes tooth marks based purely on their morphological features, rather than their inferred mode of formation, permits inferences about the latter issue to be made with little risk of circular reasoning.
The consistency and relative thoroughness with which tooth marks are described in the framework of the CM system should also lay useful groundwork for identification of possible tooth mark makers. Previous work on mammalian tooth marks has had mixed success in differentiating between different tooth mark makers (sometimes called “actors”) using mark length and width (Selvaggio and Wilder Reference Selvaggio and Wilder2001; Forrest Reference Forrest2003; Domı́nguez-Rodrigo and Piqueras Reference Domı́nguez-Rodrigo and Piqueras2003; Coard Reference Coard2007; Delaney-Rivera et al. Reference Delaney-Rivera, Plummer, Hodgson, Forrest, Hertel and Oliver2009; Young et al. Reference Young, Stillman, Smith and Korstjens2015; Koungoulos et al. Reference Koungoulos, Faulkner and Asmussen2018). However, it should be noted that even studies that were unable to attribute marks to a specific tooth mark maker were still successful in differentiating among marks made by small- and medium/large-bodied carnivores. Studies that have had the most success in identifying potential mammalian tooth mark makers have considered a number of different factors, including the linear dimensions of the tooth marks in question, their qualitative morphological features, and their distribution across the tooth-marked element(s) (Young et al. Reference Young, Stillman, Smith and Korstjens2015; Pante et al. Reference Pante, Muttart, Keevil, Blumenschine, Njau and Merritt2017; Dominguez-Rodrigo and Baquedano Reference Domínguez-Rodrigo and Baquedano2018; Koungoulos et al. Reference Koungoulos, Faulkner and Asmussen2018; Arriaza et al. Reference Arriaza, Aramendi, Maté-González, Yravedra, Baquedano, González-Aguilera and Domínguez-Rodrigo2019; Pobiner et al. Reference Pobiner, Dumouchel and Parkinson2020; Abellán et al. Reference Abellán, Jiménez-García, Aznarte, Baquedano and Domínguez-Rodrigo2021; De La Garza et al. Reference De La Garza, Madsen, Eriksson and Lindgren2021). A similarly broad-based approach appears to work well for identifying non-mammalian tooth mark makers such as crocodyliforms (Forrest Reference Forrest2003; Noto et al. Reference Noto, Main and Drumheller2012; Scheyer et al. Reference Scheyer, Delfino, Klein, Bunbury, Fleischer-Dogley and Hansen2018; De La Garza et al. Reference De La Garza, Madsen, Eriksson and Lindgren2021). For theropods, identification appears to be more straightforward, as previous studies have used tooth crown heights, fore-aft basal lengths, and basal widths to narrow down tooth mark maker identity (Erickson and Olson Reference Erickson and Olson1996; Peterson et al. Reference Peterson, Henderson, Scherer and Vittore2009; Gignac et al. Reference Gignac, Makovicky, Erickson and Walsh2010; DePalma et al. Reference DePalma, Burnham, Martin, Rothschild and Larson2013).
One reason that theropodan tooth mark makers are seemingly simpler to identify than mammalian ones may be the nature of the tooth marks themselves. Most of the marks considered in mammalian studies have been pits, features whose shape captures that of the tooth tip rather than that of the tooth as a whole. In most theropod studies, the marks considered have predominantly been punctures, which would more completely reflect the proportions of the tooth crown, provided the penetrating tooth mark did not shatter the cortical bone and create a puncture wider than the crown (Delaney-Rivera et al. Reference Delaney-Rivera, Plummer, Hodgson, Forrest, Hertel and Oliver2009). In the absence of shattering, the width of a puncture should be no greater than that of the crown, although the puncture may be narrower than the crown if only part of the latter penetrated the bone. A parallel argument applies to maximum crown height, as compared to tooth mark depth. In principle, ascribing tooth marks to particular makers may be possible if their dimensions are initially used to establish the minimum size of the crowns that produced them, and the evidence provided by their distribution and textures is subsequently taken into account. Narrowing down a list of potential tooth mark makers is often difficult in practice (Selvaggio and Wilder Reference Selvaggio and Wilder2001; Forrest Reference Forrest2003; Domı́nguez-Rodrigo and Piqueras Reference Domı́nguez-Rodrigo and Piqueras2003; Coard Reference Coard2007; Delaney-Rivera et al. Reference Delaney-Rivera, Plummer, Hodgson, Forrest, Hertel and Oliver2009), as carnivores of a range of body sizes are known to show overlap in their tooth mark dimensions, but this problem can potentially be mitigated by large sample sizes and attention to multiple lines of evidence. Recent studies that have employed multivariate tooth mark datasets and considered the distribution of tooth marks across one or more elements, combined with 3D morphology from surface scans or a combination of linear measurements and notes on internal morphology, have shown some potential in differentiating among possible tooth mark makers (Pante et al. Reference Pante, Muttart, Keevil, Blumenschine, Njau and Merritt2017; Dominguez-Rodrigo and Baquedano Reference Domínguez-Rodrigo and Baquedano2018; Arriaza et al. Reference Arriaza, Aramendi, Maté-González, Yravedra, Baquedano, González-Aguilera and Domínguez-Rodrigo2019; Pobiner et al. Reference Pobiner, Dumouchel and Parkinson2020; Abellán et al. Reference Abellán, Jiménez-García, Aznarte, Baquedano and Domínguez-Rodrigo2021; De La Garza et al. Reference De La Garza, Madsen, Eriksson and Lindgren2021).
Conclusions
The CM system proposed in this paper aims to clarify and refine the terminology used to describe and categorize tooth marks. The use of categories based on tooth mark dimensions and cortical bone penetration, in combination with clearly defined descriptive modifiers, should allow the CM system to be applied in a consistent manner across studies of tooth marks made by a wide diversity of vertebrate carnivores. This will encourage broad collaboration and facilitate comparisons among different studies. Because primary category assignments are based entirely on measurements in the plane of the tooth-marked surface, tooth marks can be assigned to primary categories based on photographs alone, and some modifiers can likewise be applied even to marks that are unavailable for examination in three dimensions. Finally, the CM system is more flexible than previously proposed tooth mark classification frameworks, allowing for future expansion to accommodate new information as needed. Given this potential for further development, we hope that the system will not only prove useful to other researchers, but also provide a foundation for them to build upon as tooth marks continue to be surveyed across vertebrate taxa.
Acknowledgments
Financial support was provided by a Dinosaur Research Institute Student Research Grant awarded to T.C.A.W.-H., and by NSERC Discovery Grants RGPIN-2017-06246 and RGPIN-2023-04916. We would like to thank the curators and collections managers at the Canadian Museum of Nature (Ottawa, Ontario, Canada), Museum of the Rockies (Bozeman, Montana, USA), Royal Tyrrell Museum of Palaeontology (Drumheller, Alberta, Canada) and University of Alberta Laboratory for Vertebrate Palaeontology (Edmonton, Alberta, Canada). A special thank you to D. D'Amore for helpful discussions and feedback on early versions of the article and for lending us the goat elements bearing the Komodo monitor tooth marks that were used in D'Amore and Blumenschine (Reference D'Amore and Blumenschine2009, Reference D'Amore and Blumenschine2012), together with data on these specimens. Special thanks also to S. Drumheller for sharing helpful insights regarding crocodilian tooth marks and providing raw images of bisected tooth marks studied by Drumheller and Brochu (Reference Drumheller and Brochu2014). Our gratitude goes out to J. Milligan for discussions on ichnotaxonomy. We would like to extend our thanks to D. Hone for granting permission to re-use images from Hone and Watabe (Reference Hone and Watabe2010) and providing copies of the original figures from that publication, and to D. Tanke for granting permission to re-use images from Tanke and Currie (Reference Tanke and Currie2000). Thanks also go out to N. Fröbisch for editorial assistance and reviewer S. Drumheller for her thoughtful comments that greatly improved this article.
Competing Interest
The authors declare no competing interests.
Data Availability Statement
Silicone peels examined in this study and used in the construction of the CM system are accessioned in the University of Alberta Laboratory for Vertebrate Palaeontology (UALVP), Edmonton, Alberta, Canada and Royal Tyrrell Museum of Palaeontology (TMP), Drumheller, Alberta, Canada. Descriptions of all tooth marks examined for construction of the CM system will be archived at the UALVP and published in subsequent articles. Supplementary Tables 1–5, containing specimen information, measurements, and modifiers for specimens listed in Table 6 and illustrated in Figures 1, 3, 5, and 6, are available from https://doi.org/10.5061/dryad.jq2bvq8jf.















