Vitamin B12 plays an important role in neurocognitive development and function( Reference Black 1 ), and its deficiency has been associated with a number of poor health outcomes including anaemia, congenital malformations, neurological deficits and hyperhomocysteinaemia( Reference Lindenbaum, Healton and Savage 2 – Reference Louwman, van Dusseldorp and van de Vijver 5 ). Animal foods, fortified foods and supplements are the main sources of vitamin B12. Consequently, vitamin B12 deficiency may be more prevalent in vegetarians, vegans, and people living in low-income communities where they may have limited purchasing power to acquire animal food sources or may not have access to fortified foods or supplements.
Some studies suggest that vitamin B12 deficiency and marginal deficiency are highly prevalent worldwide( Reference McLean, de Benoist and Allen 6 ). In the few countries where nationally representative data exist, the prevalence of vitamin B12 deficiency (serum vitamin B12<130 to <150 pmol/l) ranges from 11 % to 15 % in women from Germany, the UK( Reference McLean, de Benoist and Allen 6 ) and Vietnam( Reference Laillou, Pham and Tran 7 ), to 31 % among elderly from the UK. Regional surveys found vitamin B12 deficiency in 49 % of adults in India, 40 % of children in Kenya, 28 % of pregnant women in Nepal and 20 % of women in New Zealand( Reference McLean, de Benoist and Allen 6 ). Vitamin B12 deficiency is also of public health concern in Latin America. In nationally representative studies conducted in Argentina( Reference Duran, Mangialavori and Biglieri 8 ), Chile( Reference Castillo-Lancellotti, Margozzini and Valdivia 9 ), Costa Rica( Reference Blanco, Rodriguez and Cunningham 10 ), Venezuela( Reference Garcia-Casal, Osorio and Landaeta 11 ) and Mexico( Reference Villalpando, Montalvo-Velarde and Zambrano 12 , Reference Allen 13 ), the prevalence of vitamin B12 deficiency was, respectively, 3·4–4·7 %, 8·5 %, 5·3 %, 11·4 % and 25–30 %. High prevalences of deficiency, ranging from 13 % to 82 %, were reported in non-nationally representative studies conducted in Cuba( Reference Arnaud, Fleites-Mestre and Chassagne 14 ) and Guatemala( Reference Casterline, Allen and Ruel 15 , Reference Jones, Ramirez-Zea and Zuleta 16 ).
Only one study to date has investigated the prevalence of vitamin B12 deficiency in Colombia, a country lacking a vitamin B12 fortification policy where disparities in food purchasing power remain wide. Among schoolchildren from low- and middle-income families in Bogotá, the prevalence of vitamin B12 deficiency (serum vitamin B12<148 pmol/l) and marginal deficiency (serum vitamin B12≥148 and <222 pmol/l) was 1·6 % and 15·0 %, respectively( Reference Villamor, Mora-Plazas and Forero 17 ). In this population, low vitamin B12 concentrations were associated with poverty and low intake of animal food sources. While results from this investigation suggest that vitamin B12 deficiency is a non-negligible public health problem in this setting, its results cannot be generalized to the whole country. A nationally representative sample would be needed to evaluate the vitamin B12 status of Colombians. Furthermore, it would allow the examination of vitamin B12 status in particularly vulnerable population groups including pregnant women and women of reproductive age. Our study aimed to estimate the prevalence of vitamin B12 deficiency and marginal deficiency using serum concentrations of vitamin B12 in a nationally representative sample of Colombian women and children. In addition, we investigated the sociodemographic correlates of serum vitamin B12 concentrations in these groups.
Methods
Study population
The Colombian National Nutrition Survey (ENSIN) was conducted in 2010 by the Colombian Institute of Family Welfare (Instituto Colombiano de Bienestar Familiar). Details of the survey have been published elsewhere( 18 ). In brief, participants were selected to represent 99 % of the country’s population using a multistage stratified sampling scheme. All municipalities from the thirty-two departments in the country were grouped into strata based on similar geographic and sociodemographic characteristics. One municipality was randomly chosen from each stratum, with probability proportional to the population size. Clusters of about ten households each were then randomly chosen from within these strata and household members were invited to participate. The survey included 50 670 households, representing 4987 clusters from 258 strata.
Trained personnel administered questionnaires to the head of the household to obtain information on demographic characteristics, measures of food insecurity and wealth. Anthropometric measurements were obtained in all household members with the use of standardized techniques and calibrated instruments. Height was measured with the use of a stadiometer (Diseños Flores S.R. Ltda, Bogotá, Colombia), to the nearest millimetre. Weight was measured on SECA 872 scales to the nearest 100 g. In a random sub-sample of participants, blood was collected by venepuncture and serum was separated into aliquots. All samples were stored in liquid nitrogen until processing at the National Institute of Health of Colombia. Serum vitamin B12 was quantified in these samples using the method of direct chemiluminescence (ADVIA Centaur equipment, Siemens). This method offers high sensitivity and is less costly, easier to implement and safer than microbiological, chromatographic or spectrophotometric assays( Reference Kumar, Chouhan and Thakur 19 ).
The study was conducted according to the guidelines laid down in the Declaration of Helsinki. Consent for participation in the survey was obtained by the Colombian Institute of Family Welfare prior to enrolment( 18 ). The Health Sciences and Behavioral Sciences Institutional Review Board at the University of Michigan determined that analyses of these anonymized data were exempt from review.
Data sources
The survey included 188 599 people. Serum vitamin B12 was quantified in a random re-weighted sub-sample (n 9523) of children 1–17 years of age and women 18–49 years of age. They constituted the final analytical sample. There were 7243 boys and non-pregnant girls aged <18 years, 1781 pregnant women and 499 non-pregnant women aged ≥18 years.
The main outcomes of interest were mean serum vitamin B12 concentrations (pmol/l) and the prevalence of vitamin B12 deficiency (serum concentration <148 pmol/l) and marginal deficiency (serum concentration ≥148 and <222 pmol/l). Adequate concentration of serum vitamin B12 was defined as ≥222 pmol/l.
We examined vitamin B12 status according to sociodemographic and anthropometric characteristics. Food security status was measured using a modified version of the Community Childhood Hunger Identification Project( Reference Wehler, Scott and Anderson 20 ), which has been previously adapted for and validated in a Colombian population( Reference Alvarez, Estrada and Montoya 21 ). There are twelve questions, addressed to the head of the household, regarding food insecurity experienced within the past 30 d due to lack of money for food. Only seven questions are asked to households without children. Response options for each question (‘no’, ‘seldom’, ‘sometimes’ or ‘always’) were assigned codes 0, 1, 2 or 3, respectively. Codes were added through all responses and the sum was categorized into a four-level variable: food secure (sum=0), mild food insecurity (sum=1–7 or 1–12 in households without or with children, respectively), moderate food insecurity (sum=8–14 or 13–24) or severe food insecurity (sum≥15 or ≥25). Wealth was measured using a survey designed for the international Demographic and Health Surveys( Reference Rutstein and Johnson 22 ). A continuous index representing household wealth was quantified by principal component analysis of various socio-economic indicators including household assets, type of flooring, number of bedrooms per person and mode of transportation. This variable was categorized into quintiles. In adults, BMI was estimated from measured weight and height as kg/m2. In children (age<18 years), we estimated height- and BMI-for-age Z-scores with the use of the WHO growth standard( Reference de Onis, Martorell and Garza 23 ) or reference( Reference de Onis, Onyango and Borghi 24 ) for children and adolescents if 60 months of age or older.
Data analysis
All analyses were conducted with the use of the complex survey design routines of the Stata statistical software package version 12. We estimated weighted prevalences of vitamin B12 deficiency and marginal deficiency for boys and non-pregnant girls <18 years, pregnant women and non-pregnant women ≥18 years. Next, we estimated mean vitamin B12 serum concentrations and their standard errors by levels of predictors including age, sex, socio-economic status indicators, urbanicity and region of origin. Linear regression models were used to estimate adjusted mean differences between groups and 95 % confidence intervals, with vitamin B12 serum concentration (pmol/l) as the dependent variable and sociodemographic variables as the predictors. In addition, we conducted tests for linear trend by introducing a variable representing categories of ordinal correlates as a continuous predictor into the models, accounting for the complex survey design. All analyses were conducted separately for boys and non-pregnant girls <18 years and for pregnant women. Analyses of vitamin B12 correlates could not be conducted among non-pregnant women ≥18 years because of small sample size.
Results
Mean serum vitamin B12 concentration was 302 (se 4) pmol/l; the prevalence of vitamin B12 deficiency and marginal deficiency was, respectively, 6·6 % (95 % CI 5·2 %, 8·3 %) and 22·5 % (95 % CI 21·1 %, 23·9 %). Among children (age <18 years), pregnant women and adult non-pregnant women, prevalence of vitamin B12 deficiency was 2·8 % (95 % CI 2·3 %, 3·3 %), 18·9 % (95 % CI 16·6 %, 21·5 %) and 18·5 % (95 % % CI 4·4 %, 53·1 %), respectively (Fig. 1). Prevalence of marginal vitamin B12 deficiency in the same groups was 18·2 % (95 % CI 17·0 %, 19·5 %), 41·0 % (95 % CI 38·0 %, 44·1 %) and 18·8 % (95 % CI 7·7 %, 39·2 %).
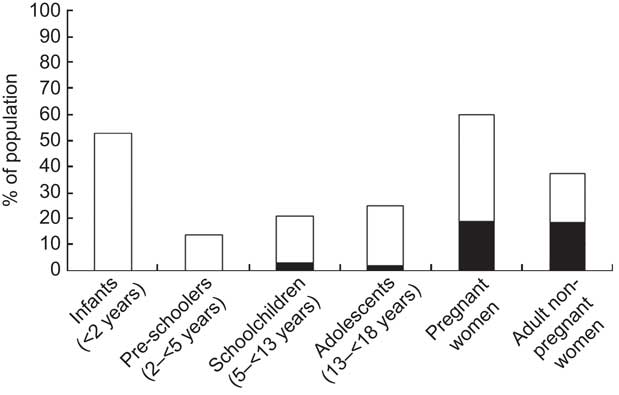
Fig. 1 Prevalence of vitamin B12 deficiency (serum concentration<148 pmol/l; ![]() ) and marginal deficiency (serum concentration≥148 and<222 pmol/l;
) and marginal deficiency (serum concentration≥148 and<222 pmol/l; ![]() ) in Colombian children and women, National Nutrition Survey 2010. Infants, n 16; pre-schoolers, n 84; schoolchildren, n 6908; adolescents, n 235; pregnant women, n 1781; adult non-pregnant women, n 499
) in Colombian children and women, National Nutrition Survey 2010. Infants, n 16; pre-schoolers, n 84; schoolchildren, n 6908; adolescents, n 235; pregnant women, n 1781; adult non-pregnant women, n 499
Next, we examined correlates of serum vitamin B12 in children (Table 1). In bivariate analyses, mean serum vitamin B12 was significantly positively related to female sex, education of the household head, wealth, food security, and living in urban areas or in Bogotá and the Central or Atlantic regions. After multivariable adjustment, serum vitamin B12 was 12 pmol/l higher in girls than boys (P=0·004). Children whose parents had at least secondary education had mean serum vitamin B12 concentration 12 pmol/l (95 % CI 3, 21 pmol/l; P=0·009) higher than those whose parents had only primary education or less. There was a monotonic positive relationship of serum vitamin B12 with food security; compared with children from households without food insecurity, serum vitamin B12 was on average 21 pmol/l lower (P=0·003) in children from households with severe food insecurity. The associations with wealth index and geographic area were attenuated in multivariable analysis and became non-statistically significant. Mean serum vitamin B12 was not associated with child’s age or height-for-age Z-score. There was an inverse, non-statistically significant relationship between BMI-for-age Z-score and mean serum vitamin B12 concentration.
Table 1 Serum vitamin B12 concentrations (pmol/l) in Colombian childrenFootnote * (age<18 years, non-pregnant) according to sociodemographic characteristics

* National Nutrition Survey 2010; n 7243.
† Totals may be less than 7243 due to missing values.
‡ Test for linear trend for ordinal predictors. For sex, urbanicity and country region, P is from ANOVA. All tests incorporated the complex sampling survey design.
§ From linear regression models with serum vitamin B12 as a continuous outcome and indicator variables for all characteristics presented in the table as predictors. Estimates for education were from a model excluding wealth index and food security, which could be on the causal pathway. Estimates for wealth index excluded food security.
|| Adjusted test for linear trend or ANOVA for ordinal or categorical correlates, respectively.
¶ According to the WHO( Reference de Onis, Martorell and Garza 23 , Reference de Onis, Onyango and Borghi 24 ).
** The wealth index is a composite measure of a household’s cumulative living standard. The wealth index is calculated using easy-to-collect data on a household’s ownership of selected assets such as televisions and bicycles, materials used for housing construction, type of water supply and sanitation facilities.
In pregnant women, serum concentrations of vitamin B12 were significantly positively associated with education of the household head (Table 2). After adjustment, compared with having less than primary education, university education of the household head was related to on average 24 pmol/l higher serum vitamin B12 levels in pregnant women (P=0·03). Living in the National territories, the Eastern or the Pacific regions was associated with significantly lower serum vitamin B12 levels than living in other regions. Serum vitamin B12 was not significantly associated with age, height, wealth, food insecurity or urbanicity among pregnant women.
Table 2 Serum vitamin B12 concentrations (pmol/l) in Colombian pregnant womenFootnote * according to sociodemographic characteristics

* National Nutrition Survey 2010; n 1781.
† Totals may be less than 1781 due to missing values.
‡ Test for linear trend for ordinal predictors. For urbanicity and region, P is from ANOVA. All tests incorporated the complex sampling survey design.
§ From linear regression models with serum vitamin B12 as a continuous outcome and indicator variables for all characteristics presented in the table as predictors. Estimates for education were from a model excluding wealth index and food security, which could be on the causal pathway. Estimates for wealth index excluded food security.
|| Adjusted test for linear trend or ANOVA for ordinal or categorical correlates, respectively.
¶ The wealth index is a composite measure of a household’s cumulative living standard. The wealth index is calculated using easy-to-collect data on a household’s ownership of selected assets such as televisions and bicycles, materials used for housing construction, and type of water supply and sanitation facilities.
Discussion
We estimated the prevalence of vitamin B12 marginal deficiency and deficiency in children, pregnant women and adult non-pregnant women in a representative sample of the Colombian population in 2010 using serum concentrations of vitamin B12. We found a substantial combined prevalence of vitamin B12 deficiency and marginal deficiency of 28·7 % in these groups. Prevalence of deficiency was close to 20 % among women of childbearing age. Because this figure is above 5 %, vitamin B12 deficiency can be characterized as a country-wide public health problem in this subgroup( Reference McLean, de Benoist and Allen 6 ).
Our findings are similar to those in other Latin American countries, where high prevalences of vitamin B12 deficiency and marginal deficiency were common public health problems in the 1990s( Reference McLean, de Benoist and Allen 6 , Reference Allen 25 ). A national survey in Mexico (1999) showed that the combined prevalence of deficiency and marginal deficiency in children and women reached approximately 40 %( Reference Allen 13 ). However, a more recent nationally representative survey in Argentina (2007) demonstrated that the prevalence of deficiency in women was 3·4 %, much lower than the figure reported here( Reference Duran, Mangialavori and Biglieri 8 ). Only one previous study had examined vitamin B12 status in Colombia. Among low- and middle-income children from Bogotá, the combined prevalence of deficiency and marginal deficiency reached 17 % in 2006( Reference Villamor, Mora-Plazas and Forero 17 ), similar to the figure found for this age group and city in the present survey.
The potential consequences of vitamin B12 deficiency are substantial at the individual and population levels. Deficiency of vitamin B12 may result in anaemia, congenital malformations, neurological deficits and hyperhomocysteinaemia( Reference Lindenbaum, Healton and Savage 2 – Reference Louwman, van Dusseldorp and van de Vijver 5 ). Fortification of food vehicles with vitamin B12 is a useful public health nutrition intervention to curb vitamin B12 deficiency at the population level. In Colombia, although a policy of wheat flour fortification with micronutrients has been in effect since 1996, vitamin B12 is not included. The results of the present study support the notion that wheat flour fortification with vitamin B12 should be considered at the national level.
We had the opportunity to examine the associations of several sociodemographic and anthropometric indicators with vitamin B12 serostatus. While age was not related to vitamin B12 status in children, boys had lower serum vitamin B12 concentrations compared with girls. The sex difference in vitamin B12 status could be due to increased requirements for boys than girls at times of accelerated growth( Reference Villamor, Mora-Plazas and Forero 17 ). It is also possible that boys have lower intake of vitamin B12 from animal food sources, but lack of systematically collected dietary information in the survey prevented us from examining this potential mechanism. Indicators of socio-economic status were positively associated with serum vitamin B12 concentrations in children, consistent with results from a previous study of schoolchildren from Bogotá( Reference Villamor, Mora-Plazas and Forero 17 ). In addition, children living in urban areas and in geographic regions with greater economic and structural development in general had higher serum concentrations than those from rural and poorer areas. This socio-economic-related variation in vitamin B12 status in children is likely due to access to animal food sources, which tend to be the most expensive items of the family shopping basket. Adherence to an ‘animal protein’ dietary pattern was strongly positively related to vitamin B12 serostatus in children from this setting( Reference Villamor, Mora-Plazas and Forero 17 ).
We found an inverse trend between children’s BMI-for-age Z-score and serum vitamin B12 concentrations (P=0·08), consistent with previous studies of adults and children( Reference Baltaci, Kutlucan and Ozturk 26 – Reference Pinhas-Hamiel, Doron-Panush and Reichman 29 ). In a study of Israeli children, obesity was associated with a fourfold higher risk of low serum vitamin B12 concentrations (<181·5 pmol/l)( Reference Pinhas-Hamiel, Doron-Panush and Reichman 29 ). The inverse association of serum vitamin B12 concentrations with BMI could be a result of increased consumption of carbohydrates and fats at the expense of animal-source protein among obese children. They may also have increased vitamin B12 requirements than non-obese children due to their larger body size. Whether vitamin B12 deficiency could lead to obesity cannot be determined by cross-sectional surveys. Future examination of this question is critical because vitamin B12 is a cofactor in methylation reactions and DNA hypomethylation has been related to faster weight gain in children( Reference Perng, Rozek and Mora-Plazas 30 ). In addition, vitamin B12 is also a cofactor in the synthesis of methionine from homocysteine; thus, vitamin B12 deficiency or marginal deficiency may lead to hyperhomocysteinaemia, which has been associated with the development of insulin resistance in prepubertal children( Reference Martos, Valle and Morales 31 ) and also predicts the development of morbidity and mortality from CVD( Reference Clarke, Collins and Lewington 32 ).
Women had a higher prevalence of vitamin B12 deficiency or marginal deficiency than children. This was possibly due to their greater nutritional requirements that may not be met through dietary sources. The recommended daily intake of vitamin B12 in women above the age of 14 years is 2·4 μg/d, compared with 1·8 μg/d for children aged 9–13 years and 1·2 μg/d for children aged 4–8 years( 33 ). Furthermore, malabsorbtion of food-bound vitamin B12 increases with age and may account in part for a higher prevalence of deficiency in adults. Pregnant women had the highest prevalence of vitamin B12 deficiency and marginal deficiency of the groups studied, and they also have the highest requirements; their recommended daily intake is 2·6 μg/d( 33 ). In pregnant women, like in children, education of the household head and living in regions with greater economic and structural development were positively associated with serum vitamin B12 concentrations. However, other socio-economic indicators including the wealth index and food security were not related to vitamin B12 serostatus among pregnant women. These null associations could be due to lack of variability in vitamin B12 concentrations given the high prevalence of deficiency and marginal deficiency in this group. It is also possible that the impact of socio-economic status on vitamin B12 varies within households. For example, mothers may prioritize providing animal food sources to their children when they are insufficient for the whole family( Reference Dammann and Smith 34 ). As a result, variables like household food insecurity may not fully capture the situation of women in the survey.
We found substantial geographic and urban/rural differences in vitamin B12 status of children and pregnant women. The lowest concentrations were found in rural areas and in country regions with traditionally poorer living conditions, as measured with the Human Development Index( Reference McLean, de Benoist and Allen 6 ). The mechanisms to explain a positive association of the Human Development Index with vitamin B12 serostatus may be related to food security, purchasing power to obtain animal food sources and prevalence of intestinal parasites that may interfere with vitamin B12 absorption.
There are several strengths to our study. It is the first description of vitamin B12 status in Colombia using a nationally representative sample. Second, serum vitamin B12 is a valid biomarker of intake( Reference Villamor, Mora-Plazas and Forero 17 ). Third, we had an opportunity to identify specific population subgroups where vitamin B12 deficiency is a particularly serious problem. The study also has some limitations. First, it is based on a prevalence survey, which prevents us from establishing temporal relationships between vitamin B12 serostatus and the sociodemographic correlates examined. Second, folate status indicators were not available in the survey. The effects of vitamin B12 deficiency may mask those of inadequate folic acid status and it is important to consider both nutrients simultaneously( Reference Herbert and Bigaouette 35 ). Serum folate concentrations in children were high in the Bogota School Children Cohort( Reference Villamor, Mora-Plazas and Forero 17 ), and high folate levels combined with low vitamin B12 concentrations were associated with anaemia in the same study( Reference Arsenault, Mora-Plazas and Forero 36 ) and with anaemia and cognitive impairment in studies among the elderly( Reference Selhub, Morris and Jacques 37 ). These findings cannot be generalized to other areas of the country where folate deficiency may be more prevalent. In addition, the policy to fortify wheat flour with micronutrients including folic acid has not been formally evaluated. Thus, there are strong justifications for measuring folate in the next national nutrition survey in Colombia. Third, the survey did not measure vitamin B12 serum concentrations in men >18 years or in the elderly. Due in part to decreased gastric acidity and food-bound malabsorption of vitamin B12, the elderly are at an increased risk for vitamin B12 deficiency( 33 ). It would be critical to include this high-risk subgroup in future surveys. Fourth, due to sample size limitations, we could not examine the associations of vitamin B12 with sociodemographic characteristics in non-pregnant adult women.
Measuring serum vitamin B12 concentrations at the population level is practical and relatively inexpensive, and has been implemented in a number of nationally representative surveys. Nevertheless, the use of serum vitamin B12 as a proxy of vitamin B12 status requires consideration. This measurement represents both vitamin B12 intake and stores and not necessarily the vitamin available to tissues; thus, low values may not capture developing deficiencies. Instead, low serum vitamin B12 concentrations may be indicative of long-term low intake or absorption abnormalities( 33 ), whereas concentrations of vitamin B12 metabolites such as total homocysteine, methylmalonic acid (MMA) or holotranscobalamin may be more sensitive indicators of deficiency status. Furthermore, the sensitivity and specificity of serum vitamin B12 as an indicator of vitamin B12 status vary according to age, concentrations of the binding protein and other factors( Reference Pennypacker, Allen and Kelly 38 , Reference Lindenbaum, Rosenberg and Wilson 39 ). In the elderly, for example, a high proportion of people with normal serum concentrations of vitamin B12 may present with neurological abnormalities caused by vitamin B12 deficiency( Reference Lindenbaum, Rosenberg and Wilson 39 ). Low serum concentrations of vitamin B12 also lack sensitivity to identify persons who underutilize the vitamin due to genetic defects on MMA-CoA mutase, transcobalamin II, or enzymes in the pathway of cobalamin adenosylation( 33 , Reference Kano, Sakamoto and Miura 40 , Reference Jansen, Kalousek and Fenton 41 ).
The use of specific cut-off points of serum vitamin B12 to define deficiency and marginal deficiency also deserves comment. We used the conventionally accepted cut-off points of <148 pmol/l (deficiency) and 148–222 pmol/l (marginal deficiency). While these cut-off points may be considered somewhat arbitrary, there is a pathophysiological rationale to their use. MMA and homocysteine concentrations associated with anaemia, megaloblastosis and neuropathy decrease substantially when serum vitamin B12 concentrations are greater than 148 pmol/l( Reference Selhub, Morris and Jacques 37 , Reference de Benoist 42 ), even in the absence of clinical manifestations. Because pregnant women, the elderly and young children may have elevated MMA levels in the presence of vitamin B12 concentrations >148 pmol/l( Reference Morris, Jacques and Rosenberg 43 , Reference Vanderjagt, Ujah and Patel 44 ), a second cut-off point is necessary to capture probable (or marginal) deficiency. Infants and young children with serum vitamin B12 concentrations<222 pmol/l have lower Hb concentrations and higher MMA levels than children with higher vitamin B12 values( Reference Jones, Ramirez-Zea and Zuleta 16 , Reference Bailey, Carmel and Green 45 , Reference Schneede, Dagnelie and van Staveren 46 ). Thus, a range of 148–222 pmol/l is conventionally used to indicate marginal deficiency that may later lead to severe deficiency. Although there is some variability in the choice of serum vitamin B12 cut-off points to define deficiency, those used in the present study allow for cross-comparisons with other nationally representative surveys and previous reports in this population.
Conclusion
In conclusion, vitamin B12 deficiency and marginal deficiency are non-negligible public health problems in Colombian children and women. The prevalence of vitamin B12 deficiency is particularly high in pregnant women. Socio-economic status of the family, urbanicity and geographic region are strong predictors of vitamin B12 serostatus.
Acknowledgements
Financial support: This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. Conflicts of interest: None. Authorship: O.F.H. and E.V. designed the research and performed the statistical analyses. O.F.H. obtained the data. J.B.W., O.F.H. and E.V. wrote the paper. O.F.H. and E.V. had primary responsibility for the final content. All authors have read and approved the final version of the manuscript. Ethics of human subject participation: The research was conducted in accordance with guidelines laid down by the Declaration of Helsinki. The Health Sciences and Behavioral Sciences Institutional Review Board at the University of Michigan exempted from review all research on these anonymized data sets.





