There is increasing evidence that maternal nutrition before and during pregnancy influences the course of pregnancy, foetal development and the child's health in adult lifeReference Barker1–Reference Harding5. Thus, it is important to determine maternal exposure to healthy and toxic substances in food, and large cohort studies are carried out to investigate the effects of diet on pregnant women and their children. However, dietary intake is notoriously difficult to measure because of recall errors, misreporting and the difficulty of assessing portion sizesReference Cade, Thompson, Burley and Warm6. A food-frequency questionnaire (FFQ) is the preferred method in most population studies because administration is easy, intake can be assessed over a long time period and the cost is lowReference Willett and Willett7. Every FFQ has to be validated to get an expression of the degree to which it is an accurate measure in the target population before the outcome variables can be checked against dietary intake. A validation study should preferably include biological markers (biomarkers) of dietary intake in addition to a dietary reference method as no true external reference measurement of dietary intake existsReference Cade, Thompson, Burley and Warm6, Reference Nelson, Margetts and Nelson8. With two independent reference methods in addition to the FFQ, the triangular method is applicableReference Ocke and Kaaks9.
Since no FFQ had been designed or validated for pregnant women in Norway, a new questionnaire was specially developed to be used for assessing diet during the first four months of pregnancy, when the foetus is most vulnerableReference Godfrey and Barker3, Reference Harding5.
Flavonoids are found in fruit and vegetables, and their presence in urine reflects short-term intakeReference Nielsen, Haren, Magnussen, Dragsted and Rasmussen10, Reference Rasmussen and Breinholt11. The urinary flavonoid concentration has only been used as a measure in a few studies, but these have shown a clear dose–response relationship with fruit and vegetable intakeReference Krogholm, Haraldsdottir, Knuthsen and Rasmussen12, Reference Brevik, Rasmussen, Drevon and Andersen13. The plasma level of several carotenoids rises with increasing intake of carotenoid-rich foodsReference Hunter and Willett14–Reference van het Hof, Brouwer, West, Haddeman, Steegers-Theunissen and van Dusseldorp17, but no studies have yet been done on this relationship in pregnant women. Carotenoids are generally found in the same food groups as flavonoids, but have a different distribution, and reflect intake over a longer time span than urinary flavonoids doReference Block, Norkus, Hudes, Mandel and Helzlsouer18, Reference El Sohemy, Baylin, Spiegelman, Ascherio and Campos19. Correlations between estimated intake and biomarkers are generally weaker than correlations between two dietary methods because biomarkers will be influenced by factors unrelated to intake, such as individual differences in absorption, metabolism and distributionReference Willett, Lenart and Willett20.
In the present study, the method of triads was used to calculate validity coefficients (VCs) using FFQ estimates for fruit and vegetable intakes and two independent biomarkers, 24-hour urine flavonoid excretion and plasma carotenoid concentration. This approach calculates the correlation between the FFQ measure and the ‘true’ but not known intake. To our knowledge, no previous studies have applied two independent biomarkers for calculation of triangular VCs for fruit and vegetable intake.
The aim of the present study was to validate the ability of the FFQ to measure the intake of fruit, vegetables and tea reported by pregnant women using biomarkers and a food diary (FD) as reference methods.
Materials and methods
The Norwegian Mother and Child Cohort study
This study is a subproject in MoBa, the Norwegian Mother and Child Cohort StudyReference Magnus, Irgens, Haug, Nystad, Skjaerven and Stoltenberg21. In brief, MoBa is a pregnancy cohort that in the period 1999–2005 has included >60 000 pregnancies, and that aims to include 100 000 by the end of 2007. Pregnant women are recruited to the study by postal invitation after they have signed up for the routine ultrasound examination in their local hospital. Participants are asked to provide biological samples and to complete questionnaires covering a wide range of information up to the age of 7 years for the child. The cohort database is linked to the Medical Birth Registry of Norway and other national health registries.
Food-frequency questionnaire
The MoBa FFQ22 was mailed to all participants at gestational weeks 12–15. It is a semi-quantitative questionnaire designed to capture dietary habits and intake of dietary supplements during the first four months of pregnancy. The FFQ included 33 questions related to the use of vegetables, three related to intake of potatoes and two related to intake of legumes. The FFQ included 20 questions related to fruit, three questions related to juice and three questions related to the intake of tea. For each food item, the frequency of use was reported by selecting one out of eight to 10 frequencies ranging from never to several times monthly, weekly or daily. Global questions are asked regarding hot meals, fruit and vegetables, and were used for scaling to adjust estimation. When portion sizes were not given in the questionnaire, consumption frequencies were converted into food amounts by the use of standard Norwegian portion sizes for women. The questionnaires were optically read and, for nutrient calculations, FoodCalc23 and the Norwegian food composition tableReference Rimestad, Borgejordet, Vesterhus, Sggnestveit, Løken and Trygg24 were used.
Validation study subjects and design
Eligible, healthy pregnant women in MoBa assigned to Bærum Hospital (Norway) were invited to participate in a validation study when they came for routine ultrasound examination at 17–18 weeks of gestation. Exclusion criteria were hyperemesis and anorexia. Before inclusion, subjects had to have completed the MoBa FFQ. The inclusion period lasted from 15 January 2003 to 1 February 2004.
The women participating in the validation study were asked to keep a 4-day weighed FD and to provide one 24-hour urine collection and a blood sample. They were given detailed information and the materials for data collection at a meeting with the project coordinator in groups of five to 10. Data on weight, height and age were provided in the FD. Length of education and pre-pregnancy smoking status in validation study subjects were computed from questionnaire no. 1 in MoBa.
Of 120 subjects included, one dropped out due to illness. The average time interval between completion of the FFQ and participation in the study was 24 days (standard deviation (SD) 12 days). The study protocol was approved by the regional ethics committee of Southern Norway, and informed written consent was obtained from all participants in the validation study.
Four-day weighed food diary
Participants were asked to weigh and record all food, beverages and food supplements consumed during three consecutive weekdays and one weekend day. Each participant was given an FD and a digital balance (Philips Essence HR 2389 with a precision of ± 1 g), and asked to continue with their normal food intake. Upon collection, each FD was checked for completeness of description by a nutritionist (A.L.B.).
Food supplements
Food supplement intake was recorded in both the FFQ and the FD. Because food supplements influence biomarker levels in blood and urine, participants using supplements containing carotenoids or flavonoids were identified. Food supplements were not included in the dietary intake estimates.
Twenty-four-hour urine collection
Each participant collected one 24-hour urine sample at the end of the FD period, although urine was not collected at weekends. Participants were provided with one 2.5 and two 1.0 l bottles and a funnel for urine collection. Aliquots of 50 and 20 ml (1 M) of HCl, respectively, had been added to the 2.5 and 1.0 l bottles. The participants were asked to add pre-weighed vials of 1000 and 400 mg ascorbic acid to the 2.5 and 1.0 l bottles, respectively, before use, and instructed to keep the bottles in a cool place. All urine bottles from one participant were pooled and the samples stored within 8 h of the 24-hour collection. Urine samples for flavonoid analysis were adjusted to pH < 4 and stored at − 70°C until analysis.
Determination of enterolactone and flavonoids in urine
Urine concentrations of enterolactone, two isoflavonoids and nine flavonoids were determined by liquid chromatography–mass spectrometry at the Department of Toxicology and Risk Assessment, Danish Institute for Food and Veterinary Research, according to established methodsReference Krogholm, Haraldsdottir, Knuthsen and Rasmussen12.
Blood sampling
Blood samples were taken at the time of recruitment. Plasma was obtained by centrifugation of heparinised blood within 2 h of venepuncture and stored at − 70°C until analysis.
Determination of carotenoids in plasma
Plasma concentrations of carotenoids were determined by high-performance liquid chromatography at Vitas AS, Norway according to the method described by Brevik et al. except that the column dimensions were 2.1 mm × 150 mm, the particle size 3 μm, the mobile phase flow was 0.3 ml min− 1 and the injection volume was 25 μlReference Brevik, Andersen, Karlsen, Trygg, Blomhoff and Drevon25.
Statistical analysis
While plasma carotenoids were normally distributed, intake of most foods and food groups was not normally distributed, nor was urinary excretion of flavonoids. Dietary intake is presented as medians, 5th and 95th percentiles. Spearman correlations were calculated for biomarkers and intake of various food groups, urine and plasma biomarkers, and the two dietary methods. The Mann–Whitney U-test was used to test for possible differences between biomarker-supplement and non-biomarker-supplement users, smokers and non-smokers, different education levels and participants with and without nausea.
Correlations with biomarkers were calculated for all participants (n = 119) and after exclusion of participants taking food supplements containing flavonoids (n = 20) or carotenoids (n = 13). The ability of the FFQ and FD to classify pregnant women correctly into quintiles according to intake of fruit, vegetables and tea was checked against the selected biomarkers, and Fisher's exact test was used to compare the proportion of subjects classified correctly by the two dietary methods.
The VCs for intake assessed by the FFQ in relation to the ‘true’ but not known intake (VCFFQ-T) were calculated according to the method of triads advocated by Ocké and KaaksReference Ocke and Kaaks9. The equation 
To obtain 95% confidence intervals (CIs) for the VCs, we used the percentile bootstrap methodReference Ocke and Kaaks9. This method is carried out by resampling the observed data with replacement. From resampled data, we then calculate bootstrap VCs. The resampling is repeated 1000 times and the 95% CI is then given as the interval from the 2.5 percentile to the 97.5 percentile of the bootstrap VCs.
A problem with the triad formula is that VCs may become larger than 1, often referred to as the Heywood caseReference Ocke and Kaaks9. Since VCs are estimates of the correlation between the true values and measurements (intake), this does not make sense. A second problem is that the VCs cannot be calculated when a correlation between measurements is negative (due to the square root in the triad formula). Such problems did not occur for the actual estimates in our data. However, it did occur for several bootstrap samples. The same problem has been reported in many studiesReference Kabagambe, Baylin, Allan, Siles, Spiegelman and Campos26–Reference McNaughton, Marks, Gaffney, Williams and Green28 and is due to the fairly crude estimation method. Generally, one would prefer maximum likelihood estimation (MLE)Reference Mardia, Kent and Bibby29 for factor analysis models, which is the basis for the triad estimatesReference Ocke and Kaaks9. MLE always give VCs that can be interpreted as correlations, taking values between − 1 and +1. Furthermore, negative correlations between measurements do not pose a problem. Also, when the method of triads gives estimates that can all be interpreted as correlations, these are equal to the MLEs. To obtain MLEs, use of statistical software is required, but this is also necessary for bootstrapping. Our bootstrap CIs were subsequently based on MLE. We also calculated VCs and corresponding bootstrap CIs from log-transformed measurements rather than on ranks. Differences between the methods were negligible.
We used the statistical program R30 for the MLE and bootstrap procedure. All other analyses were performed with SPSS, version 12 (SPSS Inc.).
Results
Subjects
The 119 participants in the validation study varied with regard to age (mean: 31 years, range 23–44 years), weight (at time of FD: mean 70 kg, range 52–111 kg), weight prior to pregnancy (mean 65 kg, range 47–116 kg) and height (mean 1.68 m, range 150–185 m). Ninety-one subjects (76%) reported having experienced nausea during the present pregnancy, and 18 subjects (15%) reported nausea still at the time of answering the FFQ. Eleven subjects (9%) reported daily smoking and 13 subjects (11%) reported occasional smoking prior to becoming pregnant. Ninety-eight subjects (84%) had completed >12 years of education.
Food intake
The values for intake of protein, carbohydrates, folate, vitamin C, magnesium and fibre were significantly greater estimated by the FFQ than by the FD, but those for total energy, added sugar and fat were not significantly different (Table 1). The intake of vegetable and most fruit groups was significantly greater with the FFQ apart from apples, where the intake was significantly greater with the FD (Table 2).
Table 1 Daily intake (crude and energy-adjusted) and Spearman correlations of various nutrients (diet only) estimated by food-frequency questionnaire (FFQ) and food diary (FD)

P5 – 5th percentile; P95 – 95th percentile.
* Correlation P < 0.05.
** Correlation P < 0.01.
† Difference between FFQ and FD, P < 0.05.
†† Difference between FFQ and FD, P < 0.01.
‡ n = 42 (excluding participants with weight change >1 kg during the study period).
Table 2 Daily intake (crude and energy-adjusted) and Spearman correlations of various food groups recorded by food-frequency questionnaire (FFQ) and food diary (FD)

P5 – 5th percentile; P95 – 95th percentile; SumFJV1 – sum of fruits, fruit juices and all vegetables except potatoes; SumFJV2 – sum of fruits, fruit juices and all vegetables including potatoes.
* P < 0.05.
** P < 0.01.
† Difference between FFQ and FD, P < 0.05.
†† Difference between FFQ and FD, P < 0.01.
‡ Raw vegetables – leafy green vegetables, tomatoes, cucumber, peppers.
§ Cooked vegetables – excluding cruciferous vegetables, roots and potatoes.
Total intake of fruit, juice and vegetables was significantly greater in non-smoking women (P = 0.015) than in smokers, and in women with >12 years of education (P = 0.010) than in women with ≤ 12 years of education. The differences between groups were found with both the FFQ and the FD.
Biomarkers
The median (5th and 95th percentile) excretion of the sum of nine flavonoids in 24-hour urine was 1281 μg (158, 10 343) in participants taking no flavonoid-containing supplement (n = 99) and 2336 μg (364, 36 611) in flavonoid-containing supplement users (n = 20). The quantitatively largest amounts of flavonoids excreted in urine were the citrus flavonoids hesperetin, naringenin and eriodictyol, followed by phloretin, a flavonoid found in apples. The flavonoids found in vegetables, tea and wine (quercetin, kaempferol, isorhamnetin, tamarixetin and apigenin) were excreted in much smaller amounts. The isoflavonoids genistein and daidzein were identified along with enterolactone, a lignan metabolite produced by the intestinal microflora from indigestible plant substances. The median excretion of isoflavonoids in 24-hour urine was 59 μg in non-flavonoid-containing supplement users and 118 μg in flavonoid-containing supplement users, and that of enterolactone was 389 and 455 μg, respectively.
The mean concentration of the sum of six carotenoids in plasma was 1.70 μmol l− 1 (SD 0.50 μmol l− 1) in participants taking no carotenoid-containing supplement (n = 106) and 2.10 μmol l− 1 (SD 0.55 μmol l− 1) in carotenoid-containing supplement users (n = 13). The largest amounts of carotenoids were lycopene, found in tomatoes, and β-carotene, α-carotene and lutein, found primarily in roots and green vegetables. Zeaxanthin and β-cryptoxanthin are colourful substances found in vegetables and fruits.
Statistically significant differences between non-biomarker-supplement users and biomarker-supplement users were found for urine excretion of apigenin, eriodictyol, naringenin and hesperetin, and for the plasma concentration of lutein and β-carotene (P < 0.05 for all). No differences in fruit and vegetable intake were found between biomarker-supplement users and non-biomarker-supplement users.
Correlations between biomarkers and food intake
The correlations were generally stronger between biomarkers and FD results than between biomarkers and FFQ results (Table 3). However, there were no significant differences (Fisher's exact test) between the proportion of subjects classified into the same or adjacent quintiles by biomarkers and those classified by either of the dietary assessment methods (Table 4). Significantly increased biomarker levels were found across increasing quintiles of intake of the corresponding fruit and vegetable groups.
Table 3 Correlations between intake of fruit and vegetables determined by dietary methods (FFQ and FD) and biomarkers (BM)

FFQ – food frequency questionnaire; FD – food diary; SumFJV1 – sum fruits, fruit juices and vegetables except potatoes; sum citrus flavonoids – sum of eriodictyol, naringenin and hesperetin; sum isoflavonoids – sum of genistein and daidzein; sum flavonoids – all flavonoids measured except isoflavonoids; SumIT – sum of isorhamnetin and tamarixetin.
Correlations are reported for non-biomarker-supplement users (n = 99 for flavonoids and n = 106 for carotenoids) and for all participants (n = 119).
* P < 0.05.
** P < 0.01.
Table 4 Cross-classification of subjects by quintiles based on food-frequency questionnaire (FFQ), biomarkers (BM) and food diary (FD)
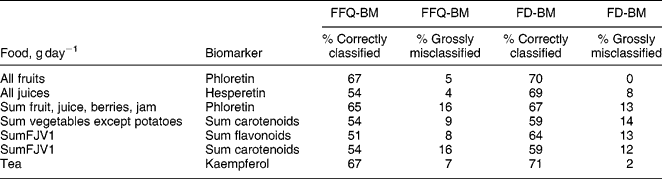
SumFJV1 – sum of fruits, juices and all vegetables except potatoes.
Correctly classified if classified into the same or adjacent quintiles, grossly misclassified if classified into opposing quintiles.
Fruit and juice intake was more strongly correlated with flavonoid concentration, and vegetable intake with carotenoid concentration. The strongest correlations were found between citrus fruits/juices and citrus flavonoids, and between all fruits and phloretin. The strongest correlations between carotenoids and food intake were found between α-carotene, carrots and cooked vegetables.
Tea intake was correlated with kaempferol as expected, but unexpectedly significant correlations were found between coffee consumption and β-carotene with both the FFQ and FD (Table 3).
Statistically significant correlations between urine and plasma biomarkers were found for urine naringenin and plasma β-cryptoxanthin (r = 0.40, P < 0.001), for urine hesperetin and plasma zeaxanthin (r = 0.27, P = 0.003), and for urine eriodictyol and β-cryptoxanthin (r = 0.32, P < 0.001). Significant correlations were also found for urine isorhamnetin and plasma α-carotene (r = 0.32, P < 0.001) and β-carotene (r = 0.28, P = 0.002). Furthermore, urine phloretin correlated with plasma α-carotene (r = 0.23, P = 0.011) and urine enterolactone with plasma α-carotene (r = 0.20, P = 0.028).
Triangular VCs were calculated for FFQ and biomarkers and for FD and biomarkers, respectively. VCs were also calculated for FFQ, FD and one biomarker (Table 5). The highest VC for the FFQ was found for intake of citrus fruit/juice.
Table 5 Validity coefficients (VCs) and 95% confidence intervals for fruit and vegetable intake

CitrusFJ–sum of all citrus fruits and juice; Cooked vegetables–excluding cruciferous vegetables and potatoes.VCs are presented for calculations based on one dietary estimate (food-frequency questionnaire (FFQ) or food diary (FD)) and two independent biomarkers (BM1 and BM2) and for calculations based on two dietary estimates (FFQ and FD) and one biomarker (BM). Total n = 119.
Discussion
In this study, the validity of a new FFQ designed for pregnant women was tested for intake of fruit, vegetables and tea. Biomarkers in urine and plasma and a 4-day weighed FD were used as reference methods. The study demonstrated an association of fruit/vegetable intake with biomarkers in both urine and plasma. Lower correlation coefficients were found with the FFQ than with the FD, but this may be due to the time lapse between the assessment methods and the superior detail of an FD compared with an FFQ. The VCs estimated for the FFQ were high, suggesting that the FFQ is a valid tool for assessment of fruit and vegetable intake in pregnant women in MoBa.
The amounts of excreted flavonoids in the pregnant women were in line with those previously reported for urinary excretion of flavonoids in non-pregnant populations in Denmark and FinlandReference Nielsen, Freese, Cornett and Dragsted31, Reference Nielsen, Freese, Kleemola and Mutanen32, and the plasma levels of carotenoids corresponded well with levels found in female participants in other European populationsReference Al Delaimy, van Kappel, Ferrari, Slimani, Steghens and Bingham33.
The group of women in the present study was not a representative sample of pregnant Norwegian women. However, the differences in fruit and vegetable intake in relation to education and smoking showed that our sample was sufficiently heterogeneous to reflect food patterns found in non-pregnant womenReference Billson, Pryer and Nichols34, Reference Pollard, Greenwood, Kirk and Cade35.
As in most validation studies, the estimated daily intake of nutrients and foods was greater when measured by the FFQ than by the FDReference Willett, Lenart and Willett20. The FFQ covers a longer time span and may thus be a better reflection of habitual intake than the FD, since the use of the questionnaire is based on the idea that the average diet over time is a better measure of exposure than intake over a few specific daysReference Willett and Willett7. Hence the two methods are not really comparable, and this is why we chose biomarkers as the main reference method.
Most pregnant women use some kind of dietary supplement in pregnancy, and many supplements contain carotenoids or flavonoids that could obscure the correlation of biomarkers in blood and urine with fruit and vegetable intake. In this study, flavonoid- and carotenoid-containing supplements were found to increase the excretion of certain flavonoids and also the plasma concentration of certain carotenoids. However, they had little impact on the correlations found between these biomarkers and fruit and vegetable intake because the majority of the participants were not taking such supplements (Table 3). Biomarker-supplement and non-biomarker-supplement users did not differ with regard to intake of fruit, juice and vegetables or related micronutrients, and this may indicate that dietary supplements are not taken as a substitute for eating fruit and vegetables in pregnancy, but in addition.
The present study indicates that specific biomarkers (phloretin for apples and α-carotene for carrots) can also serve as indicators for larger food groups. Phloretin was found to be a marker of total fruit intake, and α-carotene to be a general marker for total vegetable intake, showing that those eating fruit and vegetables also usually eat apples and carrots.
In a Finnish study of non-pregnant adults, urinary flavonoids were more closely correlated with total fruit and vegetable intake than were plasma carotenoidsReference Nielsen, Freese, Kleemola and Mutanen32. The same was observed in the present study, where flavonoids tended to be a better biomarker for total fruit and vegetable intake than carotenoids: r FD-BM = 0.31 and 0.20, respectively (Table 3). However, flavonoids were found to be a better biomarker for fruit intake alone and carotenoids for vegetable intake alone. Phloretin was found to be a highly significant marker for total fruit intake (r FD = 0.44, r FFQ = 0.33), and, as expected, the citrus flavonoids (hesperetin, naringenin and eriodictyol) specifically reflected the intake of citrus fruits and juices (r FD = 0.66, r FFQ = 0.41). The flavonol kaempferol was found to be a significant marker for tea intake (r FD = 0.50, r FFQ = 0.41).
The carotenoid that was most highly correlated with food intake was α-carotene; it was found to be significantly correlated with intake of both carrots and cooked vegetables (r FD = 0.48, r FFQ = 0.33 and r FD = 0.39, r FFQ = 0.37, respectively). β-Cryptoxanthin was found to have a higher correlation with total fruit and vegetable intake than any of the other carotenoids measured. This is in accordance with two other studies that also found β-cryptoxanthin to be more highly correlated with fruit and vegetable intake than other carotenesReference Block, Norkus, Hudes, Mandel and Helzlsouer18, Reference Michaud, Giovannucci, Ascherio, Rimm, Forman and Sampson36.
Overall, the association between the FD and biomarkers was closer than that between the FFQ and biomarkers, both in the bivariate (Table 3) and in the triangular comparison (Table 5). However, in light of associations between the FD and the urine biomarkers being the biologically best attainable, the correlations found between this FFQ and biomarkers were sound for several groups of fruit and vegetables. It is important to remember that the time frame covered by the different assessment methods in this study varied from days (FD, urine flavonoids) and weeks (carotenoids) to months (FFQ). Furthermore, the correlations between the different methods used for the validation of the FFQ have probably been weakened because the participants were pregnant women undergoing considerable metabolic changes and because the FFQ covered a time when most of the participants had experienced nausea. However, our finding of statistically significant correlations between the FFQ, FD and biomarkers indicates that the FFQ provides a useful estimate of dietary fruit and vegetable intake in pregnant women.
Using the method of triads resulted in validity coefficients for FFQ that were higher than the correlation coefficients between FFQ and each of the biomarkers. Despite the fact that the triangular method states the need for independent measurements, all other published studies have used two dietary methods and one biomarker in the triangular equationReference Ocke and Kaaks9Reference Kabagambe, Baylin, Allan, Siles, Spiegelman and Campos26–Reference McNaughton, Marks, Gaffney, Williams and Green28. To our knowledge, this is the first study that has applied three totally independent measurements. Triangular VCs calculated using the FFQ, FD and one biomarker, as used in other studies, resulted in comparable VCs (Table 5), but a positive correlation of the random measurement errors between the FFQ and FD is likely to occur. Like others who have used the method of triads, we found wide CIsReference Andersen, Veierød, Johansson, Sakhi, Solvoll and Drevon27. However, the range between the lower confidence limit and the calculated VC, i.e. 0.40–0.65 for citrus fruits and juice, indicates satisfactory validity of the questionnaire.
Our bootstrap CIs were based on MLE of validity coefficients, rather than triad estimates. Strictly, the MLEs require normal distributions for the measurements. This may be somewhat in contrast to the use of Spearman correlations between the measurements which are based on the ranks rather than the actual measurements.
We thus investigated by means of stochastic simulation how the use of ranks instead of actual normally distributed measurements affected VCs in a setting resembling our data. This simulation showed no practical difference either on estimates of VCs or on bootstrap CIs.
The correlations found between the two dietary methods were within the range observed in other validation studies in pregnant womenReference Erkkola, Karppinen, Javanainen, Rasanen, Knip and Virtanen37, Reference Robinson, Godfrey, Osmond, Cox and Barker38, and lower than those reported in non-pregnant populationsReference Byers39–Reference Subar, Thompson, Kipnis, Midthune, Hurwitz and McNutt41. For logistical reasons, the time between the administration of the FFQ and the reference methods was on average 24 days, which meant that 65% of the participants had had weight changes of >1 kg. The correlation between the total energy intake estimated with the FFQ and that with the FD reflects this, since it rose from 0.27 to 0.52 when participants with a weight change of >1 kg were excluded. However, this left only 42 subjects, which is too small a sample for calculating the other correlationsReference Willett, Lenart and Willett20. Furthermore, the significant correlations found between dietary measures for the FFQ and the FD (Tables 1 and 2) and biomarkers (Table 3) also confirm that although energy intake changes during pregnancy, the composition of the diet is more or less maintained.
The highest correlations between the FFQ and FD were found for tea and coffee (r = 0.53, r = 0.80). Tea and coffee intake varies less and is easier to recall than intake of most other food groups. The correlation between intake of coffee and β-carotene was surprising, as coffee does not contain carotenoids. This could be a random finding, but occurred with both the FFQ and the FD. However, Svilaas et al. found that coffee was the main contributor to the total intake of antioxidants, and that this total was significantly correlated with plasma carotenoids, which supports the hypothesis that unknown antioxidants may interact with and somehow ‘salvage’ known antioxidants such as carotenesReference Svilaas, Sakhi, Andersen, Svilaas, Strom and Jacobs42.
When the FFQ and the FD were compared in terms of biomarkers, the questionnaire was found to be less accurate than the FD at ranking individuals according to fruit, juice and vegetable intake. This is not surprising in view of the greater detail and shorter time frame of the FD.
Conclusions
The validation of dietary intake in pregnant women is complicated because of the accompanying nausea and weight gain and the considerable metabolic changes. However, we were able to demonstrate statistically significant correlations between the dietary intake assessed with the new FFQ and biomarkers. Our study indicates that the new FFQ provides valid estimates of the fruit, juice, vegetable and tea intake of Norwegian women during the first four months of pregnancy, and that it can be used to rank individuals within the distribution.
Acknowledgements
Sources of funding: The work was financially supported by research grants from the Research Council of Norway and the European Commission, 6th Framework Programme, Priority 5 on Food Quality and Safety (FOOD Contract No. 016320 Integrated Project), ‘Newborns and Genotoxic exposure risk: development and application of biomarkers of dietary exposure to genotoxic chemicals and of biomarkers of early effects, using mother–child birth cohorts and biobanks (NewGeneris)’. Flavonoid analyses were supported by the Research Centre for Environmental Health (ISMF) and the Danish Research Agency (FELFO).
Conflict of interest declaration: None of the authors had a conflict of interest.
Authorship responsibilities: A.L.B., M.H., J.A. and H.M.M. contributed to the study design and planning. A.L.B. conducted the study, S.E.R. performed the flavonoid analyses and S.O.R. assisted with the bootstrap procedure. All authors took part in the interpretation of the data and the preparation of the manuscript.
Acknowledgements: The authors would like to thank all the women who participated in the validation study.







