Introduction
Chickpea (Cicer arietinum L.) is a diploid (2n = 2x = 16) self-pollinated cool season crop having genome size of 738 Mb (Varshney et al., Reference Varshney, Mohan, Gaur, Gangarao, Pandey, Bohra, Sawargaonkar, Chitikineni, Kimurto, Janila, Saxena, Fikre, Sharma, Rathore, Pratap, Tripathi, Datta, Chaturvedi, Mallikarjuna, Anuradha, Babbar, Choudhray, Mhase, Bhardwaj, Mannur, Harer, Guo, Liang, Nadrajan and Gowda2013; Kushwah et al., Reference Kushwah, Bhatia, Rani, Yadav, Singh, Bharadwaj and Singh2021a). It is second most consumed grain legume after dry bean, originated from south-eastern Turkey (Ladizinsky, Reference Ladizinsky1975) and cultivated largely in the semi-arid and arid regions of Asia and Africa (Gaur et al., Reference Gaur, Azam, Jeena, Khan, Choudhary, Jain, Yadav, Tyagi, Chattopadhyay and Bhatia2012). Chickpea seeds are rich in carbohydrates (around 60%), proteins (23%), nutrients, vitamins and essential amino acids in a readily digestible form (Jukanti et al., Reference Jukanti, Gaur, Gowda and Chibbar2012). It is free from anti-nutritional factors and also helps in improving soil fertility by fixation of atmospheric nitrogen (Kushwah et al., Reference Kushwah, Gupta, Bindra, Johal, Singh, Bharadwaj, Dixit, Gaur, Nayyar, Singh and Singh2020a).
Despite growing international demand, chickpea productivity is unstable and lower than the desired level (Millan et al., Reference Millan, Clarke, Siddique, Buhariwalla, Gaur, Kumar, Gil, Kahl and Winter2006) due to adverse effects of several biotic and abiotic stresses (Thudi et al., Reference Thudi, Upadhyaya, Rathore, Gaur, Krishnamurthy, Roorkiwal, Nayak, Chaturvedi, Basu, Gangarao, Fikre, Kimurto, Sharma, Sheshashayee, Tobita, Kashiwagi, Ito, Killian and Varshney2014), particularly drought (Krishnamurthy et al., Reference Krishnamurthy, Kashiwagi, Gaur, Upadhyaya and Vadez2010). About 90% of chickpea is sown under rainfed conditions with residual soil moisture (Kumar and Abbo, Reference Kumar and Abbo2001), which leads to 40–50% annual yield loss from drought stress alone (Sabaghpour et al., Reference Sabaghpour, Mahmoudi, Saeed, Kamel and Malhotra2006; Varshney et al., Reference Varshney, Thudi, May, Jackson and Janick2010). Drought stress reduces the conserved soil moisture and promotes evapotranspiration as the season progresses, thus reduces yields (Toker et al., Reference Toker, Lluch, Tejera, Serraj, Siddique, Yadav, Redden, Chen and Sharma2007). This emphasizes the urgent requirement to focus on increasing the chickpea productivity under drought stress.
Reproductive stage is the most significant growth stage in chickpea affected by the terminal drought stress (Kushwah et al., Reference Kushwah, Bindra, Singh, Dixit, Sharma, Srinivasan, Gaur, Singh, Gosal and Wani2020b). Drought stress is well-known for reducing the crop growth duration in various crops, thus affecting yield components, i.e. total biomass, pod number, seed number, seed weight, seed quality and yield per plant (Toker et al., Reference Toker, Lluch, Tejera, Serraj, Siddique, Yadav, Redden, Chen and Sharma2007; Krishnamurthy et al., Reference Krishnamurthy, Kashiwagi, Upadhyaya, Gowda, Gaur, Singh, Purushothaman and Varshney2013). Breeding for drought tolerance requires an understanding of the genetic basis of the numerous morphological and physiological traits responsible for drought tolerance (Purushothaman et al., Reference Purushothaman, Krishnamurthy, Upadhyaya, Vadez and Varshney2017). Despite several studies in this area, the significance of the traits responsible for drought tolerance remains unclear, resulting in poor exploitation of critical characteristics in drought tolerance breeding programmes. Despite of this, genotypes having low yield potential under non-stress condition generally depict high tolerance under stress condition, which also causes problems in selection of tolerant genotypes precisely.
Drought tolerance is a complex quantitative trait which is highly influenced by genotype by environment (G × E) interactions (Kashiwagi et al., Reference Kashiwagi, Krishnamurthy, Upadhyaya and Gaur2008). Thus, high G × E interactions largely hampered the direct selection of genotypes with high yields under stress conditions in the field (Kushwah et al., Reference Kushwah, Bhatia, Singh, Singh, Bindra, Vij and Singh2021b). Due to the presence of various underlying mechanisms such as drought escape, drought avoidance and drought tolerance, understanding the yield stability under drought stress becomes more difficult (Tuberosa and Salvi, Reference Tuberosa and Salvi2006). Genetic improvement in the drought tolerance-related traits can be an enduring strategy for high and stable yield under drought stress. Considering this, a trait-based breeding strategy can be preferred over yield-based breeding approach, as seed yield is profoundly affected by high G × E interactions and low heritability (Ludlow and Muchow, Reference Ludlow and Muchow1990; Kushwah et al., Reference Kushwah, Bhatia, Singh, Thudi, Singh, Bindra, Vij, Gill, Bharadwaj, Singh and Varshney2021c). But successful exploitation of trait-based breeding strategy under drought stress in chickpea is still doubtful.
Yield-based selection indices involve the comparison of crop performance under stress and non-stress environment, thus facilitate the selection of tolerant genotypes efficiently (Porch, Reference Porch2006). Thus, an inter-specific population from a cross between GPF 2 (C. arietinum) and ILWC 292 (C. reticulatum) has been used in the present study to identify drought-tolerant lines based on breeder-friendly yield-based selection indices in chickpea.
Material and methods
Plant materials and experimental sites
A set of 202 recombinant inbred lines (RILs; F8-generation) segregating for drought tolerance-related traits from an inter-specific cross of drought-tolerant genotype GPF 2 (C. arietinum L.) with drought-sensitive accession ILWC 292 (C. reticulatum) were developed using single seed descent method. Chickpea cultivar GPF2 is a semi-erect, medium tall cultivar recommended for cultivation in Punjab state and in North Western Plains Zone of India. This is a drought-tolerant high-yielding chickpea cultivar resistant to fusarium wilt and ascochyta blight. Another parent of RILs, ILWC292 (C. reticulatum) has semi-prostrate growth habit. It is sensitive to drought and susceptible to ascochyta blight disease, but resistant to botrytis grey mould disease and chickpea cyst nematode. In spite of drought sensitivity, it possesses some desirable drought-related traits such as more root length density, root-to-shoot ratio and membrane permeability index. The RIL population and the parents were planted in 2017 in an alpha lattice design (17 × 12) under irrigated (non-stress) and rainfed (drought-stress) conditions at two locations (Ludhiana and Faridkot) with three replications. Each RIL was planted in 2 m long paired-rows at 30 cm × 10 cm spacing. The Ludhiana (30.9010°N, 75.8573°E) and Faridkot (30.6769°N, 74.7583°E) sites are categorized as a semi-arid sub-tropical region and semi-arid dry region, respectively. Both sites comprise loamy sand with 59.8% sand and 16.5% clay (Typic Ustorthents). The average annual rainfall is 700 mm at Ludhiana and 450 mm at Faridkot, of which more than 70% occurs from July to September.
Phenotyping and statistical analysis
Sowing was done on the residual moisture on 30 October which was sufficient for good germination, as recommended for chickpea sowing in this region. Essential irrigation was applied to the irrigated treatment at regular intervals, while no irrigation was applied to the rainfed treatment. In case of rainfed plots, the soil moisture was ideal for drought conditions for chickpea crop. Drastic reduction in soil moisture content at 90, 110 and 130 days of planting in rainfed plots as compared to irrigated plots induced sufficient drought stress at reproductive stages (flowering, pod formation and development) at both locations.
A total of six yield-based selection indices, i.e. drought tolerance index (DTI), drought sensitivity index (DSI), tolerance index (TI), mean productivity (MP), geometric mean productivity (GMP) and harmonic mean productivity (HMP), were calculated by the following formulae:
-
Drought tolerance index (DTI): [(Y p) × (Y s)/(Y AP)2] (Fernandez, Reference Fernandez1992)
-
Drought sensitivity index (DSI): $[ { { 1\hbox{-} (Y_{\rm s} {\rm /}{Y}_{\rm p}} )] {/[1\hbox-(Y_{\rm A}}{_{\rm S}}/Y{_{\rm A}}{_{\rm P}})} ]$
 (Fisher and Maurer, Reference Fisher and Maurer1978)
(Fisher and Maurer, Reference Fisher and Maurer1978) -
Tolerance index (TI): Y p–Y s (Rosielle and Hamblin, Reference Rosielle and Hamblin1981)
-
Mean productivity (MP): $( {Y_{\rm p}{\rm} + Y_{\rm s}} ) {\rm /2}$
 (Rosielle and Hamblin, Reference Rosielle and Hamblin1981)
(Rosielle and Hamblin, Reference Rosielle and Hamblin1981) -
Geometric mean productivity (GMP): ${\rm GMP = }\left( {Y_{\rm s} \times Y_{\rm p}} \right)^{1/2}$
 (Fernandez, Reference Fernandez1992)
(Fernandez, Reference Fernandez1992) -
Harmonic mean productivity (HMP): $2( {Y_{\rm p} \hbox{-} Y_{\rm s}} ) {\rm /}( {Y_{\rm p}{\rm} + Y_{\rm s}} )$
 (Baheri et al., Reference Baheri, Javanshir, Kazemi and Aharizad2003)
(Baheri et al., Reference Baheri, Javanshir, Kazemi and Aharizad2003)
where, Y s is the seed yield from stressed plot of a given genotype; Y p is the seed yield from non-stressed plot of the same genotype. DII is the drought intensity index, which was calculated by the following equation:
where, Y AS is the average seed yield of all genotypes from the stressed plot; Y AP is the average seed yield of all genotypes from the non-stressed plot.
In each plot, five randomly taken plants were used to record seed yield observations in each plot under stressed and non-stressed conditions.
Analysis of variance (ANOVA) was undertaken for individual environments using a mixed-model analysis to estimate the contribution of each factor to total variation using SAS-software version 9.3 (SAS Institute, Cary, NC, USA). The variability of each trait was estimated by simple statistical measures, such as mean, range, phenotypic and genotypic variances and coefficients of variation. Variances and coefficients of variation were calculated as per Singh and Chaudhary (Reference Singh and Chaudhary1985). A matrix of simple correlation coefficients between seed yield and its components was computed using SAS-software version 9.3 to determine the relationship between the examined traits and seed yield. Principal component analysis (PCA) was performed using SAS-software version 9.3 in which all the traits were considered as independent variables while yield was taken as dependent variable.
Results
Phenotypic evaluation of selection indices of RIL population including parents
The RILs, along with parents, were evaluated for all the yield-based selection indices in the irrigated (non-stress) and rainfed (drought-stress) treatments at two research locations (Ludhiana and Faridkot) in the state of Punjab, India. Significant variations were observed in the RILs and their parents for all the selection indices and yield under irrigated (non-stress) and rainfed (stress) environment (Table 1). The analysis of parents with contrast reaction to drought for all the selection indices and yield were showing highly significant differences between parents at both locations. The rainfed treatment had significantly lower mean values of RILs for yield (YLD) (44.18%) than the irrigated treatment. The pooled ANOVA for all the yield-based selection indices including yield showed highly significant differences between genotypes at both the locations. Significant differences were also observed for genotype × location (G × L) interactions for all the selection indices (Table 1). Even though there was significant G × L interaction, the scatter plots showed highly significant correlation between locations (Ludhiana and Faridkot) for almost all of the selection indices including yield (Fig. 1). The frequency distributions for most of the yield-based selection indices were normal at both locations (Fig. 2).
Table 1. Mean performance of chickpea RIL population including parents for various quantitative drought tolerance selection indices and yield at Ludhiana and Faridkot locations
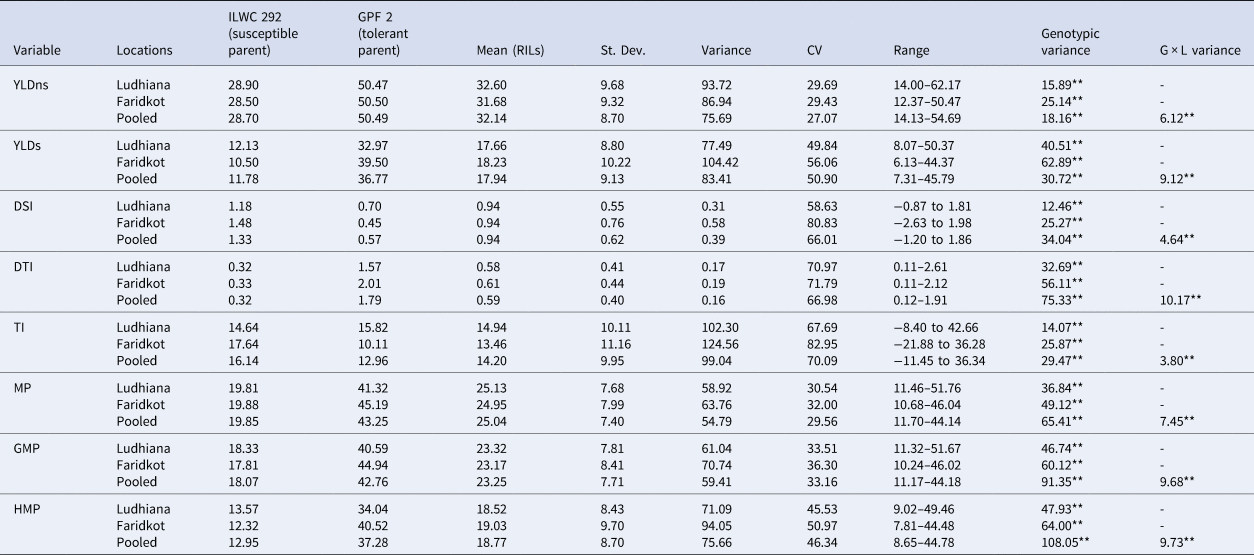
**, Highly significant at 1% probability level; YLDns, yield non-stress (irrigated); YLDs, yield stress (rainfed); DTI, drought tolerance index; DSI, drought susceptibility index; TI, tolerance index; MP, mean productivity; GMP, geometric mean productivity; HMP, harmonic mean productivity; G × L, genotype × location interaction.
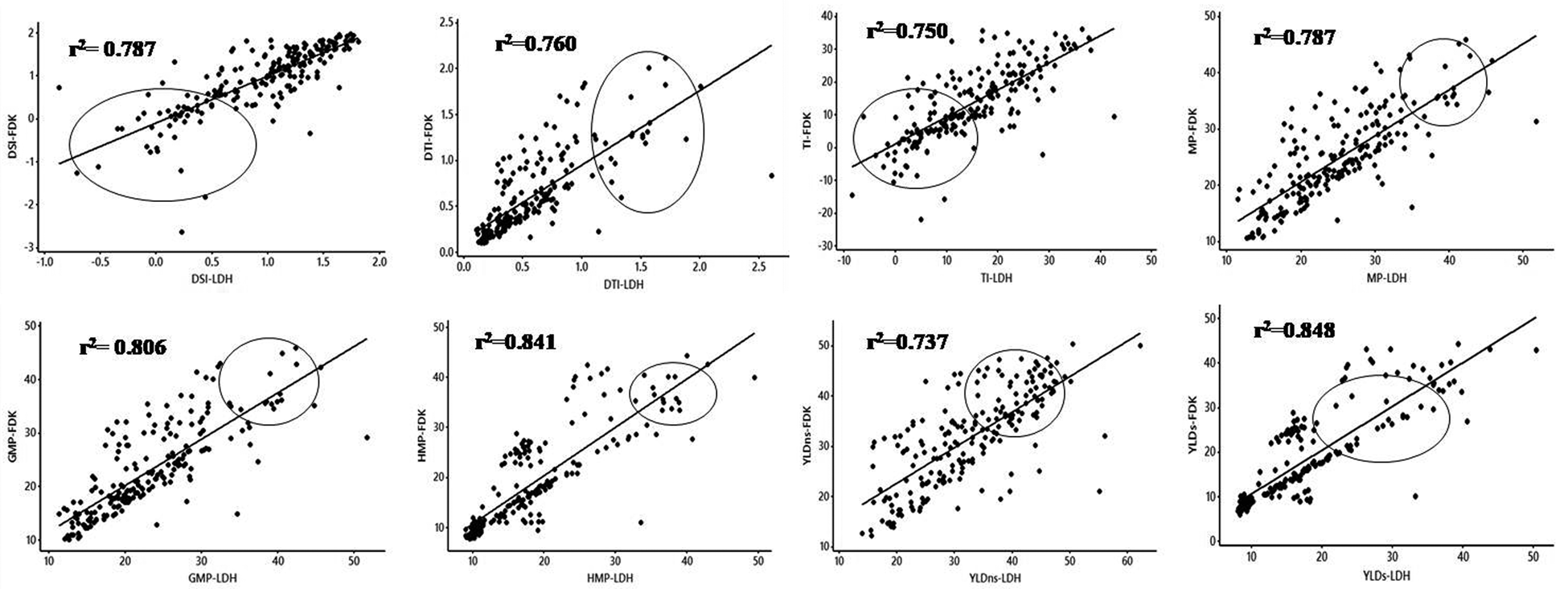
Figure 1. The scatter plots of various quantitative yield-based selection indices showing relationship between both of the locations, i.e. Ludhiana and Faridkot. The straight was plotted as regression line.
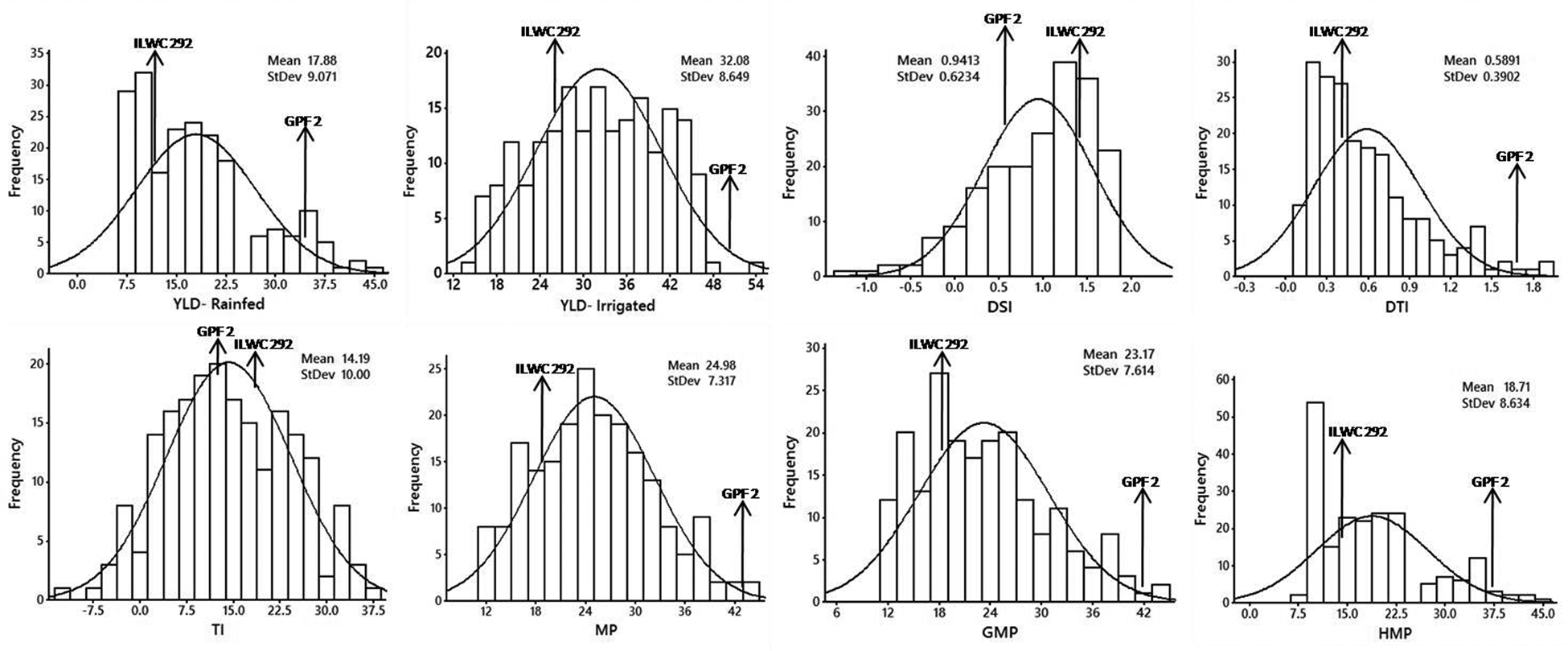
Figure 2. Graphical representations of RILs for the various quantitative yield-based selection indices and yield under rainfed and irrigated conditions in chickpea using pooled phenotypic data of Ludhiana and Faridkot locations.
Association analysis and principal component analysis
The value of independent secondary traits in the selection process can be identified through associations with dependent traits such as yield. The association analysis indicated that yield under control condition had a significant positive correlation with all the yield-based selection indices. In contrast, yield under stress condition had significant negative correlation with DSI and TI. DSI and TI depict significant negative correlation with all the yield-based selection indices, while the remaining selection indices depict the significant positive correlation with each other (Table 2).
Table 2. Phenotypic correlation coefficient among various quantitative drought tolerance selection indices and yield in chickpea at Ludhiana and Faridkot locations
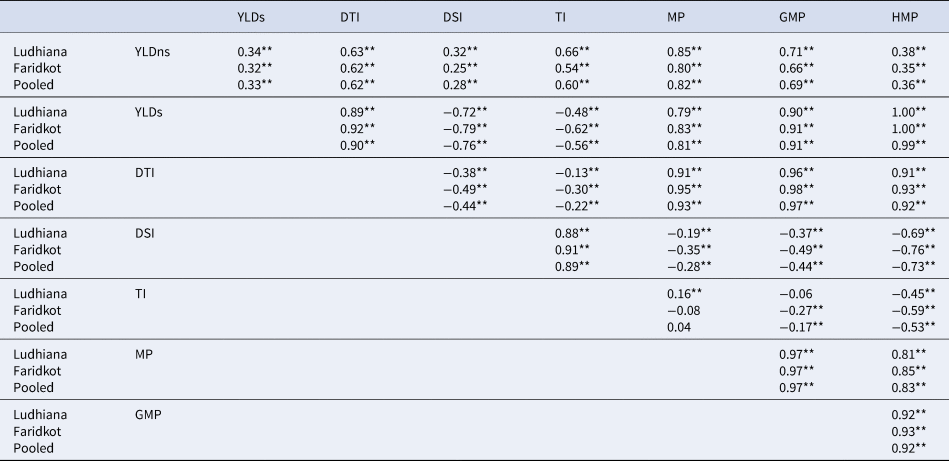
**, Highly significant at 1% probability level; YLDns, yield non-stress (irrigated); YLDs, yield stress (rainfed); DTI, drought tolerance index; DSI, drought susceptibility index; TI, tolerance index; MP, mean productivity; GMP, geometric mean productivity; HMP, harmonic mean productivity.
The PCA provides information on the measured traits to elucidate the maximum variability present in the population under specific environments. The PCA explained that the first two principal components explained 97.53 and 98.27% of the total phenotypic variability at Ludhiana and Faridkot locations, respectively (Table 3). The PCA revealed that DTI, MP, GMP and HMP were the main contributing selection indices in PC1 at both the locations, while DSI and TI were the other selection indices that occurred in a negative direction (Supplementary Fig. 1).
Table 3. Eigen values, proportion of variability and quantitative drought tolerance selection indices that contributed to the two principal components in RILs of chickpea at Ludhiana and Faridkot locations

DTI, drought tolerance index; DSI, drought susceptibility index; TI, tolerance index; MP, mean productivity; GMP, geometric mean productivity; HMP, harmonic mean productivity.
Overall, on the basis of these yield-based selection indices, a total of 15 promising RILs were identified as drought tolerant (Table 4). These lines were showing high values of DTI, MP, GMP and HMP while low values of DSI and TI as compared to both the parents as well as remaining RILs. These RILs could be used in chickpea breeding programme for developing drought-tolerant cultivars.
Table 4. List of promising recombinant inbred lines showing drought tolerance based on yield-based selection indices

DTI, drought tolerance index; DSI, drought susceptibility index; TI, tolerance index; MP, mean productivity; GMP, geometric mean productivity; HMP, harmonic mean productivity.
Discussion
Drought stress is one of the most significant abiotic stresses which reduces chickpea yields by up to 50% alone (Kumar et al., Reference Kumar, Bharadwaj, Rizvi, Sarker, Tripathi, Alam and Chauhan2015). Drought tolerance is highly influenced by several morphological and physiological traits, which directly or indirectly participated in various unknown mechanisms implying in drought tolerance (Kushwah et al., Reference Kushwah, Bhatia, Barmukh, Singh, Singh, Bindra, Vij, Chellapilla, Pratap, Roorkiwal, Kumar, Varshney and Singh2022a). Due to this highly complex nature of drought tolerance, it is imperative to study various yield-based selection indices onto which any breeder can rely for selection of drought-tolerant genotypes. Therefore, evaluation of various yield-based selection indices and identification of molecular markers tightly linked to selection indices will facilitate the selection of drought-tolerant genotypes and for introgression of drought tolerance into other genetic background in marker-assisted breeding programme. Thus, an inter-specific RIL mapping population from a cross between GPF 2 (C. arietinum) and ILWC 292 (C. reticulatum) was developed and a rainfed (drought-stress) treatment was used to evaluate RILs and parents for mapping of various selection indices using an irrigated (non-stress) treatment as a control.
Significant variation was observed in the RILs and their parents for all the yield-based selection indices and yield under irrigated (non-stress) and rainfed (stress) environment. It indicated that the genetic components in the RILs were well segregated and normally distributed, which provides ample opportunity for fine mapping of QTLs of these selection indices. However, RILs also depict the presence of transgressive segregants for all the selection indices explained that genes having both positive and negative effects were dispersed between the parents. The pooled ANOVA for all the yield-based selection indices including yield showed highly significant differences for genotypes and genotype × location (G × L) interactions at both locations. Combined ANOVA in several studies also showed significant differences for various morphological and physiological traits (Hamwieh et al., Reference Hamwieh, Imtiaz, Hobson and Ahmed2013; Pang et al., Reference Pang, Turner, Khan, Du, Xiong, Colmer and Siddique2017; Purushothaman et al., Reference Purushothaman, Krishnamurthy, Upadhyaya, Vadez and Varshney2017; Sachdeva et al., Reference Sachdeva, Bharadwaj, Sharma, Patil, Soren, Roorkiwal, Varshney and Bhat2018).
Considering DSI as an important selection index, parent ILWC 292 showed higher DSI value than the other parent GPF 2 at both locations. In contrast, GPF 2 exhibited higher value for DTI than the other parent ILWC 292. This clearly indicated that both the parents were contrasting in nature in terms of drought tolerance. GPF 2 was the tolerant parent while ILWC 292 was the sensitive parent. Similarly, considering MP, GMP and HMP as other important selection indices, GPF 2 exhibited higher value for all these selection indices than the other parent ILWC 292 at both locations. These results indicated that mapping of QTLs using RILs developed from these contrasting parents was much more precise and can be easily exploited in further drought tolerance breeding programmes in chickpea. These yield-based selection indices were already exploited for selection of drought tolerance genotypes previously in chickpea (Sabaghnia and Janmohammadi, Reference Sabaghnia and Janmohammadi2014; Jha et al., Reference Jha, Basu, Shil and Singh2016), rice (Khan and Dhurve, Reference Khan and Dhurve2016) and wheat (Anwar et al., Reference Anwar, Subhani, Hussain, Ahmad, Hussain and Munir2011). Thus, the higher value of DTI, MP, GMP and HMP indicated the presence of high level of drought tolerance as compared to others and can be efficiently used in selection of superior genotypes under drought stress. While, in context of DSI, genotypes showing <1 value of DSI can be considered as drought tolerant, also reported previously (Porch, Reference Porch2006; Yucel and Mart, Reference Yucel and Mart2014).
Selection indices having significant positive correlation with yield under both control and stress conditions can be effectively used for selection of drought-tolerant genotypes. In the present study, DTI, MP, GMP and HMP are the selection indices that showed significantly high positive correlation with YLDns and YLDs at both the locations. These results were in accordance with the previous studies in chickpea under drought stress (Sabaghnia and Janmohammadi, Reference Sabaghnia and Janmohammadi2014; Jha et al., Reference Jha, Basu, Shil and Singh2016) and heat stress (Jha et al., Reference Jha, Jha, Singh, Shil and Kole2018). Similar results were also obtained in other crops such as wheat (Drikvand et al., Reference Drikvand, Doosty and Hosseinpour2012) and maize (Parihar et al., Reference Parihar, Godawat, Singh, Parihar and Jat2012). DSI and TI were the selection indices that showed significant negative correlation with yield under stress condition in the present study which is in agreement with Sabaghnia and Janmohammadi (Reference Sabaghnia and Janmohammadi2014) and Jha et al. (Reference Jha, Basu, Shil and Singh2016). Considering these results, genotypes having high values of DTI, MP, GMP and HMP and low values of DSI and TI can be the most tolerant against drought stress. Considering MP, GMP and HMP as an important selection index for drought tolerance, promising RILs having drought tolerance showed higher value than sensitive RILs (Table 4). Importantly, these selection indices were also observed as an important selection parameter for drought tolerance in other crops such as rice (Sabouri et al., Reference Sabouri, Dadras, Azari, Kouchesfahani, Taslimi and Jalalifar2022) and wheat (Mahdy et al., Reference Mahdy, Althagafi, Al-Zahrani, Aloufi, Alsalmi, Abeed, Mahdy and Tammam2022).
Various selection indices have been suggested on the basis of seed yield under stress and non-stress conditions for identification of superior genotypes (Singh et al., Reference Singh, Singh, Tyagi, Singh and Kumar2017). Importantly, the combination of these selection indices developed by different mathematical models will be more appropriate for the selection of the promising genotypes under both stress and non-stress conditions (Sallam et al., Reference Sallam, Alqudah, Dawood, Baenziger and Börner2019). Multivariate analysis such as PCA could also give an idea of simultaneous analysis of several variables for improving the ranking accuracy of the selected genotypes (Yang et al., Reference Yang, Wang, Chen, Li and Cao2019). Indeed, a combination of specific selection indices may offer an appropriate criterion for selection in abiotic stress tolerance breeding programmes (Kushwah et al., Reference Kushwah, Bhatia, Singh, Singh, Vij, Bindra, Siddique, Nayyar and Singh2022b). In the present study, PCA reduced all the selection indices into two principal components and concluded that DTI, MP, GMP and HMP were the main contributing selection indices at both the locations, while DSI and TI were the other selection indices that occurred in a negative direction. Similar results were also obtained in some previous studies in chickpea (Sabaghnia and Janmohammadi, Reference Sabaghnia and Janmohammadi2014; Jha et al., Reference Jha, Basu, Shil and Singh2016, Reference Jha, Jha, Singh, Shil and Kole2018). On the basis of these yield-based selection indices, a total of 15 promising RILs were identified as drought tolerant having high values of DTI, MP, GMP and HMP while low values of DSI and TI as compared to both the parents as well as remaining RILs, which could be used in chickpea breeding programme for developing drought-tolerant cultivars.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S1479262123001107.
Acknowledgements
The INSPIRE research grant provided to A. K. by the Department of Science and Technology (DST), New Delhi, India and research grant provided under the project ‘Consortia Research Platform on Molecular Biology’ by the Indian Council of Agricultural Research, New Delhi to S. S. for carrying out the research are highly acknowledged.
Author contributions
A. K., G. S., S. S., I. S. and S. B. designed and conducted the experiments. D. B., A. K. and S. V. performed the data acquisition and data analysis. A. K., N. J., K. K., S. V. and S. S. prepared and edited the final manuscript. All authors reviewed the manuscript critically and approved for submission.
Competing interests
None.








