The identification of neurophysiological endophenotypes associated with psychiatric disorders may help in determining the causal relationship between clinical phenomena associated with the disorder and basic molecular processes that are in large part determined by genetic factors. One psychophysiological measure that has been used as a potential endophenotype for a number of psychiatric disorders is the acoustic startle reflex (ASR) and pre-pulse inhibition of the startle (PPI). The startle reflex is a constellation of responses usually indexed by eye blink responses (Swerdlow et al., Reference Swerdlow, Caine, Braff and Geyer1992), but also by electrophysiological recordings from cortical areas that may index cognitive responses to startle (Ehlers et al., Reference Ehlers, Phillips, Criado and Gilder2011; Ford et al., Reference Ford, White, Lim and Pfefferbaum1994, Reference Ford, Roth, Menon and Pfefferbaum1999; Putnam & Roth, Reference Putnam and Roth1990). PPI refers to the fact that if a weak stimulus is presented prior to the presentation of the startle stimuli (pre-pulse) the response to the startle is reduced in amplitude. It has been suggested that pre-pulse inhibition is an index of automatic sensorimotor gating (Geyer & Swerdlow, Reference Geyer and Swerdlow2001). In pre-pulse facilitation (PPF), the response to the startle is enhanced by short or long delay pre-pulses. PPF has been suggested to reflect a combination of alerting, attention and/ or arousal (Filion et al., Reference Filion, Dawson and Schell1998; Hsieh et al., Reference Hsieh, Swerdlow and Braff2006; Ludewig et al., Reference Ludewig, Ludewig, Seitz, Obrist, Geyer and Vollenweider2003).
The anatomical substrates of the neurobehavioral responses (ASR/PPI/PPF) to the presentation of the startle stimuli have been extensively investigated in clinical and pre-clinical studies (Braff et al., Reference Braff, Geyer, Light, Sprock, Perry, Cadenhead and Swerdlow2001a, Reference Braff, Geyer and Swerdlow2001b; Kumari et al., Reference Kumari, Antonova, Zachariah, Galea, Aasen, Ettinger and Sharma2005; Swerdlow et al., Reference Swerdlow, Braff, Taaid and Geyer1994). Startle responses involve a complex neural network extending from brainstem nuclei via the thalamus to higher order cortical areas that may regulate cognitive responses to startle (Campbell et al., Reference Campbell, Hughes, Budd, Cooper, Fulham, Karayanidis and Schall2007; Fendt et al., Reference Fendt, Li and Yeomans2001; Kumari et al., Reference Kumari, Antonova, Zachariah, Galea, Aasen, Ettinger and Sharma2005; Neuner et al., Reference Neuner, Stocker, Kellermann, Ermer, Wegener, Eickhoff and Shah2010; Schall et al., Reference Schall, Catts, Karayanidis and Ward1999). There is some evidence that the cognitive response to ASR/PPI may share a common underlying neurophysiology with some behavioral and clinical measures of cognition that require response inhibition (Filion et al., Reference Filion, Kelly, Hazlett, Dawson, Schell and Bohmelt1999). For instance, both performance on the Wisconsin Card Sorting Task and PPI of startle have been suggested to reflect pre-frontal cortical function and dysfunction (Filion et al., Reference Filion, Kelly, Hazlett, Dawson, Schell and Bohmelt1999; Swerdlow & Geyer, Reference Swerdlow, Geyer, Dawson, Schell and Bohmelt1999). Impairments in frontal lobe function and associated behaviors, such as executive functioning, have been an important theoretical construct in the understanding a number of behavioral disorders, such as schizophrenia (Bagney et al., Reference Bagney, Rodriguez-Jimenez, Martinez-Gras, Sanchez-Morla, Santos, Jimenez-Arriero and Parg2013; Chan et al., Reference Chan, Chan, Hui, Wong, Chang, Lee and Chen2014; Eisenberg & Berman, Reference Eisenberg and Berman2010; Holmen et al., Reference Holmen, Juuhl-Langseth, Thormodsen, Ueland, Agartz, Sundet and Melle2012; Owens et al., Reference Owens, Johnstone, Miller, Macmillan and Crow2010), bipolar disorders (Erol et al., Reference Erol, Kosger, Putgul and Ersoy2014; Kulkarni et al., Reference Kulkarni, Jain, Reddy, Kumar and Kandavel2010; Yen et al., Reference Yen, Cheng, Huang, Ko, Yen, Chang and Chen2009; Zimmerman et al., Reference Zimmerman, DelBello, Getz, Shear and Strakowski2006) and substance use disorders (Fernandez-Serrano et al., Reference Fernandez-Serrano, Perez-Garcia, Schmidt Rio-Valle and Verdejo-Garcia2010; Gierski et al., Reference Gierski, Hubsch, Stefaniak, Benzerouk, Cuervo-Lombard, Bera-Potelle and Limosin2013; Loeber et al., Reference Loeber, Vollstadt-Klein, von der Goltz, Flor, Mann and Kiefer2009; Maurage et al., Reference Maurage, de Timary, Billieux, Collignon and Heeren2014; van der Plas et al., Reference van der Plas, Crone, van den Wildenberg, Tranel and Bechara2009; Zorko et al., Reference Zorko, Marusic, Cebasek-Travnik and Bucik2004). If frontal cortical responses to the startle stimuli index some aspect of frontal lobe functioning involving higher cognition, then psychiatric disorders postulated to involve some aspects of frontal lobe dysfunction should also have startle deficits, such as anxiety disorders (De Pascalis et al., Reference De Pascalis, Cozzuto and Russo2013), schizophrenia (De Koning et al., Reference De Koning, Bloemen, Van Duin, Booij, Abel, De Haan and Van Amelsvoort2014; Swerdlow et al., Reference Swerdlow, Light, Sprock, Calkins, Green, Greenwood and Braff2014), post-traumatic stress disorder (Grillon et al., Reference Grillon, Morgan, Southwick, Davis and Charney1996), and alcoholism (Ehlers et al., Reference Ehlers, Phillips, Criado and Gilder2011; Marin et al., Reference Marin, Ponce, Martinez-Gras, Koeneke, Curivil, Jimenez-Arriero and Rubio2012).
The present investigation used a startle paradigm with short delay pre-pulse-plus-startle stimuli, which elicits a large frontal negative slow wave designated the N4S component (Ehlers et al., Reference Ehlers, Phillips, Criado and Gilder2011). In the present study, we used this endophenotype in an association analysis using an Affymetrix Axiom Exome1A array to explore potential genetic factors underlying the N4S component response to pre-pulse startle stimuli in a young adult MA cohort that has been previously well-characterized clinically (Criado & Ehlers, Reference Criado and Ehlers2007; Criado et al., Reference Criado, Gizer, Edenberg and Ehlers2014; Ehlers & Phillips, Reference Ehlers and Phillips2007; Ehlers et al., Reference Ehlers, Gilder, Criado and Caetano2009, Reference Ehlers, Gilder, Criado and Caetano2010, Reference Ehlers, Phillips, Criado and Gilder2011, Reference Ehlers, Liang and Gizer2012, Reference Ehlers, Stouffer and Gilder2014).
Materials and Methods
Sample Ascertainment
To investigate risk and protective factors for the pre-pulse inhibition and startle response in a select population of MA young adults, we investigated a cohort of 420 (age 18–30 years) MA men (n = 170) and women (n = 250). Participants were recruited using a commercial mailing list that provided the addresses of individuals with Hispanic surnames in 11 zip codes in San Diego County. The mailed invitation stated that potential participants must be of MA heritage, between the ages of 18 and 30 years, residing in the United States legally, and able to read and write in English. Based on a phone interview, participants were excluded if they were pregnant, were nursing, or currently had a major medical or neurological disorder or head injury. All participants were identified as having over 20% Hispanic heritage, with 92% reporting over 50% Hispanic heritage. The Semi-Structured Assessment for the Genetics of Alcoholism (SSAGA; Bucholz et al., Reference Bucholz, Cadoret, Cloninger, Dinwiddie, Hesselbrock, Nurnberger and Schuckit1994) was used to make lifetime substance use and other psychiatric disorder diagnoses, according to DSM-III-R and DSM-IV criteria. There have been several studies that have evaluated the concurrent diagnostic validity of the SSAGA across alcohol and drug dependencies, major depression, anxiety disorders, and antisocial personality disorder (Bucholz et al., Reference Bucholz, Cadoret, Cloninger, Dinwiddie, Hesselbrock, Nurnberger and Schuckit1994; Hesselbrock et al., Reference Hesselbrock, Easton, Bucholz, Schuckit and Hesselbrock1999). These findings indicate that the SSAGA is a highly reliable and valid instrument for use in studies of psychiatric disorders, including substance dependence. The protocol for the study was approved by the Institutional Review Board (IRB) at the Scripps Research Institute, and written consent was obtained for all participants. Participants were asked to refrain from alcohol and drug usage for 24 hours prior to the testing.
Startle ERP Collection and Analysis
Recordings were obtained from participants who were seated on a hospital bed in a sound-attenuated room. Acoustic startle stimuli were presented binaurally through headphones. The behavioral response to the startle (eye blink) is recorded using electrodes placed below and lateral to the eye as described (Braff et al., Reference Braff, Geyer and Swerdlow2001b). The auditory stimuli consist of 45 trials. These trials include randomly presented startle stimuli (115 dB white noise burst for 40 msec n = 30) and pre-pulse-startle stimuli (85 dB white noise burst for 20 msec-duration) immediately (<5 msec) followed by the startle (115 dB white noise burst for 40 msec n = 15). Each individual startle and/or pre-pulse startle trial is separated by an interval of 15 seconds. Background white noise was presented for the entire session at a level of 60 dB. The behavioral variables assessed included: ASR magnitude on startle trials and pre-pulse trials as determined by quantification of the eye blink response as described below.
Seven channels of ERP data (FZ, CZ, PZ, F3, F4, F7, and F8, referenced to linked ear lobes with a forehead ground, international 10–20 system) were obtained using gold-plated electrodes with impedance held below 5 KΩ. Frontal electrodes were emphasized in the montage as previous data had suggested that ERP decrements in frontal areas distinguished subjects with a risk for alcohol dependence (Bauer, Reference Bauer1997). An electrode placed left lateral infraorbitally and reference to the left earlobe was used to monitor both horizontal and vertical eye movements. ERP signals were amplified (time constant 0.3 s, 35 Hz low pass) using a Nihon Kohden EEG machine and were transferred online to a PC. The combined gain of the EEG amplifiers and the analog-to-digital multiplexer amplifier was 50 K.
The eye blink and ERP trials were simultaneously digitized at a rate of 256 Hz (bandwidth 0.5–35 Hz). Individual trials where the EEG or eye blink exceeded ±250 microvolts (<5% of the trials) were eliminated before averaging. The N4S component of the ERP was quantified using a computerized peak detection routine that identifies baseline-to-peak amplitudes (in μV) within the specified latency window (350–500 msec). The eye blink amplitude was also assessed using this routine. The latency window for the eye blink was 50–120 msec. The baseline was determined by averaging the 150 ms of pre-stimulus activity obtained for each trial. The routine is user-driven, and each peak detection must be verified by the user. All peaks were quantified by one investigator, and verified by a second investigator, both of whom were blind to participants’ characteristics.
The N4S component of the ERP was also evaluated to determine if it was altered as a function of alcohol dependence, antisocial personality disorder/conduct disorder (ASPD/CD), affective/anxiety disorders (ANYAXAF), and any other drug dependence (AnyDrugDep). In this analysis, regionally averaged N4S component responses to startle and pre-pulse/startle were compared between those participants with and without the psychiatric disorders using ANCOVA (co-varying for gender).
Sample Preparation and Genotyping
For all subjects, DNA was extracted from blood samples, followed by genotyping using an Affymetrix Exome1A chip. The DNA samples were prepared and the exome chip genotyping was performed on the Affymetrix Axiom Exome 1A Array according to the Affymetrix Axiom 2.0 Assay Manual Workflow documentation. The Affymetrix Exome 1A chip contains 247,222 markers. Variant quality from the exome chip genotyping was initially assessed according to Affymetrix best practices (Affymetrix, 2011). Plink version 1.07 (Purcell et al., Reference Purcell, Neale, Todd-Brown, Thomas, Ferreira, Bender and Sham2007) was used to calculate Hardy-Weinberg (HWE) p values on the set of unrelated samples, followed by the removal of 653 variants with an HWE p < 10−10.
Association Analysis
PLINK was used to test for genome-wide association for the N4S component of the ERP in response to acoustic startle stimuli following the presentation of a pre-pulse. PLINK was run with linear regression model parameters and with one million permutations. Gender and age were included as covariates. To determine the effect of extreme outliers in the phenotypic values, custom R code was written to generate winsorized phenotype values at 5% and 95% cut-offs, which were then used as the phenotype values in PLINK. Manhattan plots were generated using Manhattan R library (Stephen Turner, http://gettinggeneticsdone.blogspot.com/2011/04/annotated-manhattan-plots-and-qq-plots.html). Annotations of the variants were obtained from the Affymetrix Exome 1A chip description file. Multiple test correction p-value thresholds were calculated for the Affymetrix Exome1A chip using the Genetic Type 1 Error Calculator (GEC) software (Li et al., Reference Li, Yeung, Cherny and Sham2012). The UCSC Genome Browser (Kent et al., Reference Kent, Sugnet, Furey, Roskin, Pringle, Zahler and Haussler2002) was also utilized to visualize the genomic region containing the significant SNPs.
In order to determine if the SNPs that were significantly associated with the pre-pulse response phenotype were shared with the alcohol dependence trait, we used the multivariate version of the PLINK software (Ferreira & Purcell, Reference Ferreira and Purcell2009) with covariates age and gender.
LD Analysis
In order to check for linkage disequilibrium (LD) across the CCKAR and CCKBR gene regions, PLINK was utilized to extract a subset of variants for analysis based on the physical position of these genes on chromosomes 4 and 11, respectively. In particular, a region of chromosome 4 from genomic location 25,450,000–28,000,000 and chromosome 11 from genomic location 6,100,000–6,500,000, were extracted from the data set. Haploview (Barrett et al., Reference Barrett, Fry, Maller and Daly2005) was used to calculate the LD statistics and visualize the haplotype block structure of the gene regions.
Results
Demographics of the Mexican American Population
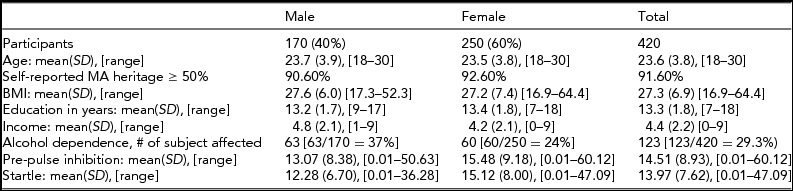
The demographics for the full sample of individuals (N = 420) that were included in the association analysis are shown in Table 1. The subjects were a mean age of 23.6 (range 18–30) years at the time of interview, with 40% of the sample being male and 60% of the sample being female. Participants had a mean of 13.3 years of education (SD = 1.8), and a mean income of $30,000–$49,000. Using self-reported ancestry based on grandparent origin, 92% of the participants reported at least 50% Hispanic heritage. The mean BMI was 27 (SD = 7, range 17–64). Approximately 29% (n = 123) of the participants were diagnosed with alcohol dependence according to DSM-III-R guidelines, indicating that these subjects had symptoms from three or more symptom groups, out of nine possible symptom groups. Eleven percent (n = 45) of the participants were diagnosed with conduct disorder or ASPD/CD. Thirty-one percent (n = 132) of the participants has a lifetime diagnosis of affective and/or anxiety disorder. Twenty-seven percent (n = 115) of the participants had a diagnosis of another drug disorder (nicotine, cannabis, hallucinogens, stimulants, sedatives, opioids).
TABLE 1 Demographics for Mexican American Study Participants
The mean N4S amplitude was evaluated using ANCOVA (co-varying with gender) as a function of alcohol dependence, ASPD/CD, any affective and/or anxiety disorder, and any other drug dependency. Participants with a lifetime diagnosis of alcohol dependence had significantly increased amplitude N4S responses to pre-pulse/startle stimuli as compared to participants with no alcohol dependence diagnoses (F = 6.535; p = .011). There were no significant associations between the N4S amplitude to pre-pulse stimuli phenotypic trait and ASPD/CD, ANYAXAF, or AnyDrugDep diagnoses.
Association Analysis
Figure 1 contains the Manhattan plot for the amplitude of the N4S response to pre-pulse stimuli phenotypic trait tested in the association analysis across the entire genome, using covariates age and gender, and applying a minor allele cut-off of 0.01. Although there were five total variants which exhibited p values ≤ E-05 after association, as shown in Table 2, there were only two variants (rs2171755 and rs58905541) that showed suggestive significance and possessed plausible gene functions for this phenotype. One protective variant (rs2171755; NC_000004.12:g.26502338T>C) is located 12 kb upstream from the CCKAR gene in a MIRb class SINE repetitive element, and is common in our sample, with an allele frequency of 0.36. The rs58905541 variant is a risk variant (NC_000011.10:g.6296157C>T) located 24 kb downstream from the CCKBR gene in a DNaseI hypersensitivity cluster, with an allele frequency of 0.018. The variant retained its significance through permutation and winsorization and the exclusion of covariates age and gender. The association values for these two variants, along with the allele and genotype frequencies, are shown in Table 3. The minor allele frequencies of these variants in the 1,000 Genomes project was obtained from the dbSNP website (http://www.ncbi.nlm.nih.gov/SNP/).
TABLE 2 Significant Variants for Pre-pulse Phenotype

TABLE 3 Allele Frequencies, Genotype Frequencies, and Association Analysis Results
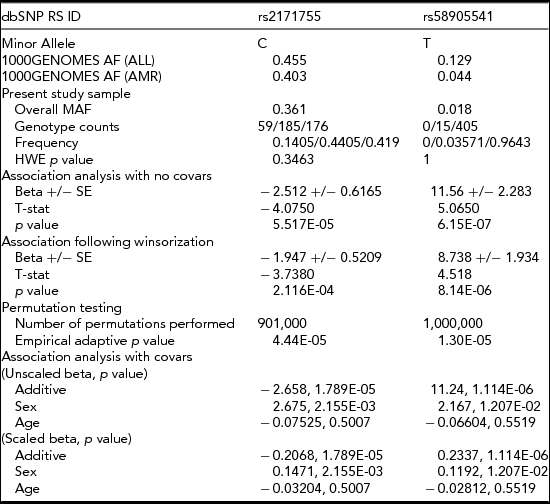
The minor allele frequencies from the 1000 Genomes project, 1000GENOMES AF (ALL = total subjects and AMR = Admixed American subjects) were obtained from the 1000Genomes website (http://www.1000genomes.org/). Abbreviations: MAF = minor allele frequency, HWE = Hardy–Weinberg equilibrium, SE = standard error.

FIGURE 1 Manhattan plot for pre-pulse phenotype.
From the multivariate association analysis of the N4S response to pre-pulse stimuli phenotypic trait and alcohol dependence diagnosis phenotype, we found that the inclusion of the alcohol dependence phenotype did not significantly alter the association of the SNPs with the pre-pulse phenotype. That is, the p values were significant whether we used univariate PLINK (rs2171755 p value = 1.79E-05; rs58905541 p value = 1.11E-06), or multivariate PLINK (rs2171755 p value = 9.36E-05; rs58905541 p value = 5.77E-06). Additionally, the weights determined by multivariate PLINK demonstrated that the pre-pulse phenotype had a strong correlation to the SNPs (rs2171755 weight for pre-pulse = 0.995; rs58905541 weight for pre-pulse = 0.999), while the alcohol dependence phenotype had a very weak correlation (rs2171755 weight for alcohol dependence = 0.026; rs58905541 weight for alcohol dependence = 0.109). These results suggest that the pre-pulse phenotype is driving the significance of the association for these two SNPs.
Multiple Test Correction
Multiple test correction p value thresholds were calculated for the Affymetrix Axiom Exome1A chip using the GEC software (Li et al., Reference Li, Yeung, Cherny and Sham2012), and the thresholds generated were thereby used to determine that the variants could be characterized to possess suggestive significance. In particular, for the Affymetrix Axiom Exome1A chip at a minor allele frequency of 0.01 or greater, the threshold for a suggestive p value was calculated as 2.53E-05, significant p value as 1.27E-06, and highly significant p value as 2.53E-08.
LD Analysis
The LD was calculated across the CCKAR and CCKBR gene regions. The average D’ value across the SNP pairs in the CCKAR region was 0.77 in this data set. No other SNPs on the Affymetrix chip were found to be in high LD with the variant near the CCKAR gene. The average D’ value across the SNP pairs in the CCKBR region was 0.85 in this data set. While no SNP was found to be in complete LD with rs58905541, the SNP with highest LD was rs1462983 (Dʹ = 0.837, r2 = 0.023, LOD = 2.19, CI = 0.37–0.95). This SNP was located in OR56B4 (olfactory receptor, family 56, subfamily B, member 4) and possessed a positive beta value, suggesting it is a risk factor. However, rs1462932 was not significantly associated with the pre-pulse inhibition response phenotype in our sample when using PLINK for the analysis.
Discussion
The present study confirmed previous observations in this population of an increase in PPF of the N4S ERP component to the acoustic pre-pulse stimuli (Ehlers et al., Reference Ehlers, Phillips, Criado and Gilder2011). In the present study, we used this endophenotype in an association analysis using an Affymetrix Axiom Exome1A array to explore potential genetic factors underlying the N4S component response to pre-pulse startle stimuli in this young adult MA cohort. The results of the present study suggest that variants located near CCKAR and CCKBR (cholecystokinin A and B receptor) genes are suggestive to be associated with the N4S ERP response to pre-pulse startle stimuli in this MA cohort. The rs2171755 variant (NC_000004.12:g.26502338T>C) is located 12 kb upstream from the CCKAR gene in a MIRb class SINE repetitive element. Interestingly, a variant in a MIRb class SINE repetitive element has been reported to be associated with human cognition (Gosso et al., Reference Gosso, de Geus, Polderman, Boomsma, Posthuma and Heutink2007). The rs58905541 variant (NC_000011.10:g.6296157C>T) is located 24 kb downstream from the CCKBR gene in a DNaseI hypersensitivity cluster. Variants in regulatory regions of the genome, such as DNase I hypersensitive sites, have been found to be associated with a number of diseases (Encode Project Consortium, 2012; Maurano et al., Reference Maurano, Humbert, Rynes, Thurman, Haugen, Wang and Stamatoyannopoulos2012). Additionally, both SNPs show phylogenetic conservation in the UCSC Genome Browser alignment tracks in Figure 2. Since, it has been reported that phenotype-associated variants occur more frequently in evolutionarily constrained regions of the genome (Parker et al., Reference Parker, Hansen, Abaan, Tullius and Margulies2009), it suggests that these SNPs are likely to be real and functional. Therefore, it is feasible that the non-coding variants found in this present study could potentially play a role in the N4S ERP response to pre-pulse stimuli.

FIGURE 2 UCSC Genome browser view of the SNP locations.
The neurotransmitter cholecystokinin (CCK) is widely present in the human body and in the central nervous system, where it modulates the dopaminergic system (Crawley & Corwin, Reference Crawley and Corwin1994). Because of its potential modulation of the dopaminergic system and associated reward pathways, CCK has been investigated in a number of studies as a candidate gene for substance dependence and other behavioral disorders. CCK, along with its two receptors, CCKAR and CCKBR, have been previously associated with anxiety and panic disorders (Maron et al., Reference Maron, Toru, Tasa, Must, Toover, Lang and Shlik2008; Wilson et al., Reference Wilson, Markie and Fitches2012), schizophrenia (Christoforou et al., Reference Christoforou, Le Hellard, Thomson, Morris, Tenesa, Pickard and Evans2007; Sanjuan et al., Reference Sanjuan, Toirac, Gonzalez, Leal, Molto, Najera and De Frutos2004), nicotine dependence (Takimoto et al., Reference Takimoto, Terayama, Waga, Okayama, Ikeda, Fukunishi and Iwahashi2005), and alcohol dependence (Miyasaka et al., Reference Miyasaka, Yoshida, Matsushita, Higuchi, Maruyama, Niino and Funakoshi2004; Okubo & Harada, Reference Okubo and Harada2001; Okubo et al., Reference Okubo, Harada, Higuchi and Matsushita2002). Although one study reported no association between CCK and CCKBR with alcohol dependence (Vanakoski et al., Reference Vanakoski, Virkkunen, Naukkarinen and Goldman2001), the study was performed using a Finnish population and did not examine the same significant SNPs that were determined in our study. Besides possible neurological influences of CCK on alcohol addiction, it has also been found that CCK through activation of CCKA receptors protects the gastric mucosa against ethanol-induced gastric damage in rats (Konturek et al., Reference Konturek, Brzozowski, Pytko-Polonczyk and Drozdowicz1995). In the context of alcoholism, variation in CCK activity in the gut could allow for the ingestion of greater or lesser amounts of alcohol, which in turn could influence liability towards alcoholism.
Further evidence of the role of CCK in the regulation of startle responses is supported by studies where the administration of CCK-related peptides has been found to influence the startle response in rats and humans. In particular, infusion of CCK has been found to enhance the acoustic startle response in rats (Feifel & Swerdlow, Reference Feifel and Swerdlow1997; Feifel et al., Reference Feifel, Priebe and Shilling2001; Fendt et al., Reference Fendt, Koch, Kungel and Schnitzler1995), and some CCK antagonists attenuate startle responses (Feifel et al., Reference Feifel, Reza, Wustrow and Davis1999; Josselyn et al., Reference Josselyn, Frankland, Petrisano, Bush, Yeomans and Vaccarino1995). In human subjects, CCK infusion has also been demonstrated to increase eye-blink startle as well as produce a mild increase in anxiety and heart rate as well as increases in plasma concentrations of ACTH, cortisol, prolactin, and growth hormone (Shlik et al., Reference Shlik, Zhou, Koszycki, Vaccarino and Bradwejn1999). Since, CCK peptides have been demonstrated to enhance anxiety, several studies have investigated the role of the CCK system in anxiety and panic disorders, using CCK-related peptide administration as a challenge method (Koszycki et al., Reference Koszycki, Prichard, Fiocco, Shlik, Kennedy and Bradwejn2012; Maron et al., Reference Maron, Toru, Tasa, Must, Toover, Lang and Shlik2008). These CCK challenge studies were able to explore genetic factors in panic disorders by performing candidate gene analyses to find alleles that may be associated with greater sensitivity to the CCK peptides. One study found an association of the tryptophan hydroxylase gene isomer 2 (TPH2) with subjects experiencing panic attacks after CCK infusion (Maron et al., Reference Maron, Toru, Tasa, Must, Toover, Lang and Shlik2008). Another study found an association to the CCKBR gene in subjects with greater pre-challenge anxiety (Koszycki et al., Reference Koszycki, Prichard, Fiocco, Shlik, Kennedy and Bradwejn2012). Additional studies have utilized CCK-peptide infusion to determine its effects on EEG and ERP measures in healthy human subjects (Knott et al., Reference Knott, Mahoney, Gunnarsson, Bradwejn and Shlik2002, Reference Knott, Mahoney, Bradwejn, Shlik and Gunnarsson2003). CCK-4 infusion was found to delay the latencies of N100 and P200 components of the ERP that was elicited during an auditory oddball task (Knott et al., Reference Knott, Mahoney, Gunnarsson, Bradwejn and Shlik2002). During EEG recording of resting subjects, CCK-4 infusion was also found to increase asymmetry and reduce coherence of the slow-wave activity at mid-temporal recording sites (Knott et al., Reference Knott, Mahoney, Bradwejn, Shlik and Gunnarsson2003). Taken together, these studies suggest a plausible role for CCK variants in the regulation of brain activity and behavior.
Impairments in frontal lobe function and associated behaviors such as executive functioning have been suggested to underlie anxiety disorders (Castaneda et al., Reference Castaneda, Tuulio-Henriksson, Marttunen, Suvisaari and Lonnqvist2008), as well as a number of other behavioral disorders such as schizophrenia (Bagney et al., Reference Bagney, Rodriguez-Jimenez, Martinez-Gras, Sanchez-Morla, Santos, Jimenez-Arriero and Parg2013; Chan et al., Reference Chan, Chan, Hui, Wong, Chang, Lee and Chen2014; Eisenberg & Berman, Reference Eisenberg and Berman2010; Holmen et al., Reference Holmen, Juuhl-Langseth, Thormodsen, Ueland, Agartz, Sundet and Melle2012; Owens et al., Reference Owens, Johnstone, Miller, Macmillan and Crow2010), bipolar disorders (Erol et al., Reference Erol, Kosger, Putgul and Ersoy2014; Kulkarni et al., Reference Kulkarni, Jain, Reddy, Kumar and Kandavel2010; Yen et al., Reference Yen, Cheng, Huang, Ko, Yen, Chang and Chen2009; Zimmerman et al., Reference Zimmerman, DelBello, Getz, Shear and Strakowski2006), and substance use disorders (Fernandez-Serrano et al., Reference Fernandez-Serrano, Perez-Garcia, Schmidt Rio-Valle and Verdejo-Garcia2010; Gierski et al., Reference Gierski, Hubsch, Stefaniak, Benzerouk, Cuervo-Lombard, Bera-Potelle and Limosin2013; Loeber et al., Reference Loeber, Vollstadt-Klein, von der Goltz, Flor, Mann and Kiefer2009; Maurage et al., Reference Maurage, de Timary, Billieux, Collignon and Heeren2014; van der Plas et al., Reference van der Plas, Crone, van den Wildenberg, Tranel and Bechara2009; Zorko et al., Reference Zorko, Marusic, Cebasek-Travnik and Bucik2004). Interestingly, schizophrenia and bipolar disorders also demonstrate significant clinical comorbidity with substance use disorders (Regier et al., Reference Regier, Farmer, Rae, Locke, Keith, Judd and Goodwin1990) and may also share genetic susceptibility factors (Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2014; Schuckit et al., Reference Schuckit, Kelsoe, Braff and Wilhelmsen2003). There is also some data to suggest that they may share common endophenotypes such as deficits in some aspects of responses to startle (Kohl et al., Reference Kohl, Heekeren, Klosterkotter and Kuhn2013), although this hypothesis requires further testing and confirmation.
In summary, in the present study, associations between the N4S ERP responses to acoustic pre-pulse startle stimuli were determined using association analyses. Our results suggest that variants located in regulatory non-coding regions near the cholecystokinin A and B receptors may play a role in the pre-pulse/startle response. However, the results of this study should be interpreted in the context of several limitations. First, the findings may not generalize to the general American population of mixed heritage or all MAs, or all Hispanic young adult Americans. Over half of the participants in the present were women and thus findings many not generalize to previous studies that have focused on samples of entirely male participants. Second, the study was limited to young adults between the ages of 18 and 30 years, and the sample size may limit the interpretation of the results. Despite these limitations, this report represents an important step in an ongoing investigation to determine risk and protective factors associated with development of substance use disorders in this select MA population.
Acknowledgments
We would like to thank and acknowledge the following people for their role in (1) the genotyping effort: Chris Bizon, Scott Chasse, Piotr Mieczkowski, Ewa Patrycja Malc, Joshua Sailsbery, and Phil Owens; and (2) for recruiting participants and collecting the clinical data: David Gilder, Susan Lopez, and Linda Corey. Funding for this study was provided by grants from the National Institutes of Health (NIH); from the National Institute on Alcoholism and Alcohol Abuse (NIAAA) and the National Center on Minority Health and Health Disparities (NCMHD) 5R37 AA010201 and P60 AA006420 (CLE) and the National Institute of Drug Abuse (NIDA) 5 R01 DA030976 (CLE, IRG, KCW, & NJS). NIAAA, NCMHD and NIDA had no further role in study design; in the collection, analysis and interpretation of data; in the writing of the report; or in the decision to submit the paper for publication. NJS and his lab are supported in part by NIH grants 5 UL1 RR025774, R21 AI085374, 5 U01 DA024417, 5 R01 HL089655, 5 R01 AG035020, 1 R01 MH093500, 2 U19 AI063603, 2 U19 AG023122, 5 P01 AG027734, 1 R21 DA033813.
Conflict of Interest
NJS is a founder and stock holder in Cypher Genomics and paid consultant for the following companies: Human Longevity, Inc., MD Revolution, and Click Therapeutics. All of the authors declare that they have no conflicts of interests.







