Introduction
Suicide is a serious public health problem and a leading cause of death in the United States. In 2021, 48183 Americans died by suicide (Stone, Mack, & Qualters, Reference Stone, Mack and Qualters2023). Major depressive disorder (MDD) is a major contributing factor (Cai et al., Reference Cai, Jin, Liu, Zhang, Zhang, Cheung and Xiang2021; Mann, Michel, & Auerbach, Reference Mann, Michel and Auerbach2021; Nordentoft, Mortensen, & Pedersen, Reference Nordentoft, Mortensen and Pedersen2011). An individual suffering from MDD is eight times more likely to die by suicide than those without the disorder (Moitra et al., Reference Moitra, Santomauro, Degenhardt, Collins, Whiteford, Vos and Ferrari2021).
While prompt diagnosis and treatment of MDD prevents suicide (Mann et al., Reference Mann, Michel and Auerbach2021), over half of those with MDD who receive antidepressant medications do not achieve remission (Rush et al., Reference Rush, Kraemer, Sackeim, Fava, Trivedi, Frank and Schatzberg2006). Failure to respond to first-line pharmacotherapy for MDD is associated with multiple negative health outcomes (Johnston, Powell, Anderson, Szabo, & Cline, Reference Johnston, Powell, Anderson, Szabo and Cline2019), including ongoing suicidal SI (Zisook et al., Reference Zisook, Trivedi, Warden, Lebowitz, Thase, Stewart and Rush2009), and the necessity of ‘next-step’ antidepressant treatments.
Whether next-step treatments exacerbate, ameliorate, or initiate suicidal thoughts and behaviors is largely unknown. Which patients are likely to experience these benefits or risks also is unknown. Addressing these clinical questions could better inform clinical decision-making and patient care.
The Veteran Administration (VA) Augmentation and Switching Treatments for MDD (VAST-D) study, a large multicenter, randomized, controlled next-step pragmatic treatment trial for patients with MDD, provides a unique opportunity to investigate treatment-related suicidal ideation (SI) and attempts (SA) among patients at high risk for suicide (Zisook et al., Reference Zisook, Johnson, Tal, Hicks, Chen, Davis and Mohamed2019; Nichter et al., Reference Nichter, Stein, Norman, Hill, Straus, Haller and Pietrzak2021). Unlike most controlled acute phase antidepressant treatment trials, VAST-D was broadly inclusive of patients with recent SI or suicidal behaviors and typical psychiatric and medical comorbidities (Mohamed et al., Reference Mohamed, Johnson, Vertrees, Guarino, Weingart, Young and Zisook2015).
This secondary analysis of VAST-D examines relationships between next-step antidepressant treatments and SI in a high-risk group of outpatients with MDD to address the following questions in depressed outpatients with inadequate benefit from at least one prior antidepressant medication:
1) What sociodemographic and clinical features are associated with lifetime SI and SA?
2) Are lifetime SI and SA associated with treatment outcomes?
3) What is the trajectory of SI during next-step antidepressant medication treatments?
4) What are the comparative effects of three commonly used next-step antidepressant treatments on SI in participants with and without current SI?
5) Which sociodemographic, clinical, and treatment features are associated with risk for new-onset or worsening of SI during the initial 12 weeks of a new course of treatment?
Methods
Study design
Complete descriptions of the study design, participant selection, methods and procedures are available (Mohamed et al., Reference Mohamed, Johnson, Chen, Hicks, Davis, Yoon and Little2017; Zisook et al., Reference Zisook, Tal, Weingart, Hicks, Davis, Chen and Mohamed2016). In brief, VAST-D was a multi-site randomized, single blind, parallel-assignment trial that included Veterans Health Administration (VHA) patients with nonpsychotic MDD from 35 VHA sites who were referred by their treating clinicians following an inadequate response (i.e. still moderately to very severely depressed) after at least one course of antidepressant treatment meeting minimal standards for dose and duration. Participants were randomly assigned to one of three treatment strategies for up to 36 weeks: switch to another antidepressant, bupropion-sustained release (S-BUP); combine current treatment with bupropion-SR (C-BUP); or augment current treatment with an antipsychotic, aripiprazole (A-ARI). This report focuses on the initial 12-week acute treatment period.
Participants
Participants were mostly male (85%) and White (69%) and most had recurrent MDD (63%); 35% had been treated with three or more antidepressant medication trials (mean = 2.4, s.d. = 1.7) and 15% with an antipsychotic medication.
Study measures
Baseline measures of suicidality
The baseline clinician-rated Columbia Suicide Severity Rating Scale (C-SSRS) (Posner et al., Reference Posner, Brown, Stanley, Brent, Yershova, Oquendo and Mann2011) determined the presence of lifetime or recent (past 3-months) suicidal thoughts and behaviors. Each of six yes/no questions addresses a different component of the respondent's lifetime or recent SI severity and behavior: (1) wish to be dead; (2) non-specific suicidal thoughts; (3) active SI with any methods (no plan) without intent to act (4) active SI with some intent to act, without specific plan; (5) active SI with specific plan and intent; (6) suicidal behavior (actual attempts, non-suicidal self-injurious behavior, interrupted attempts, aborted attempts, and preparatory acts or behaviors).
For this study, participants were characterized as having lifetime SI if they endorsed any of the five SI items and lifetime SA if they responded ‘yes’ to actual suicide attempts at the baseline evaluation. Severity of SI was scored as the number of the highest question endorsed (range: 0 to 5).
Suicidal ideation during the 12-week treatment period
The clinician rated Quick Inventory of Depression Severity (QIDS-C16) (Rush et al., Reference Rush, Trivedi, Ibrahim, Carmody, Arnow, Klein and Thase2003) was administered at baseline and at each treatment visit. QIDS-C16 item 12 was selected to measure current SI and changes over the course of treatment because the QIDS-C16 was administered at each visit and referenced the last week, whereas the C-SSRS was routinely administered only at baseline and week 12 (or last visit) and referenced the last 3-month period. Item 12 was scored on a 4-point scale from 0–3 (0 = ‘does not think of suicide or death’; 1 = ‘feels life is empty or is not worth living’; 2 = ‘thinks of suicide several times a week for several minutes’; 3 = ‘thinks of suicide/death several times a day in depth or has made specific plans for suicide or has attempted suicide’). Participants were characterized as having current SI if they endorsed any level of SI above 0. Those who endorsed a response of 1 were considered to have current passive SI, while those responding with 2 or 3 were considered to have current active SI. Online Supplementary Table 1 contains a glossary of ‘suicidality’ used in this study.
Other baseline measures
Information about age, gender, race, ethnicity, education, employment, marital status, and duration of current depressive episode were collected via patient interview. Childhood abuse was assessed with the 10-item short form of the Adverse Childhood Experiences Survey (ACE) (Bernstein et al., Reference Bernstein, Stein, Newcomb, Walker, Pogge, Ahluvalia and Zule2003; Felitti et al., Reference Felitti, Anda, Nordenberg, Williamson, Spitz, Edwards and Marks1998). A 9-item adaptation of the Brief Grief Questionnaire (Shear, Jackson, Essock, Donahue, & Felton, Reference Shear, Jackson, Essock, Donahue and Felton2006) was used to assess prolonged grief related to the death of a close relationship. Study staff administered the Mini-International Neuropsychiatric Interview (M.I.N.I.) (Sheehan et al., Reference Sheehan, Lecrubier, Sheehan, Amorim, Janavs, Weiller and Dunbar1998) to document posttraumatic stress disorder (PTSD) and to assess certain exclusionary criteria. To evaluate severity of depression at baseline, study staff administered the QIDS-C16 (Rush et al., Reference Rush, Trivedi, Ibrahim, Carmody, Arnow, Klein and Thase2003) and the Clinical Global Impression-Severity scale (CGI-S) (Guy, Reference Guy1976). Participants also competed the Patient Health Questionnaire (PHQ-9) (Kroenke, Spitzer, & Williams, Reference Kroenke, Spitzer and Williams2001). The 21-item self-report Beck Anxiety Inventory (BAI) (Beck, Epstein, Brown, & Steer, Reference Beck, Epstein, Brown and Steer1988) measured severity of anxiety symptoms. The 16-item Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form (Q-LES-Q-SF) (Endicott, Nee, Harrison, & Blumenthal, Reference Endicott, Nee, Harrison and Blumenthal1993) and the EuroQOL health state score (Group, Reference Group1990) measured quality of life and functioning. Mixed features were measured by a self-rated 12-item Mixed Features Scale, based on items taken directly from the DSM-5's criteria for mixed features (APA, 2013). Positive Mental Health was measured with a brief, 7-item, self-rated scale with items such as ‘I look forward to each day’ and ‘I am a good person.’
Treatment
1522 participants were randomly assigned to S-BUP (n = 511), C-BUP (n = 505) or A-ARI (n = 506). The most prescribed index antidepressants were, in descending order, sertraline, citalopram, fluoxetine, venlafaxine, paroxetine, duloxetine, mirtazapine, and escitalopram. Treatments included titration (cross-titration for the switch group) from standard starting doses of 150 mg of bupropion-SR to 300 mg or 400 mg daily; or from 2 mg of aripiprazole with titration to 5, 10, or 15 mg daily, until depressive symptoms remitted, or adverse effects were intolerable. Treatment visits occurred at baseline and weeks 1, 2, 4, 6, 8, 10, and 12.
Safety
A detailed safety plan was implemented to monitor and respond to SI. A response of 3 on QIDS-C16 item 12 triggered the administration of the C-SSRS to further evaluate the severity of SI. For participants determined to be at-risk based on the C-SSRS, clinical evaluation, or participant report, a clinical evaluation was conducted by the Site Investigator or mental health clinician. The participant was not left alone until the evaluation and disposition were completed. Additional visits were recommended for emergent or worsening SI. For participants remaining in the study after exhibiting potential suicide risk, a suicide risk management strategy, which included a detailed safety plan (Stanley & Brown, Reference Stanley and Brown2012) and weekly visits until symptoms improved or study treatment was discontinued, was implemented.
Statistical analysis
Descriptive statistics, including means and standard deviations for continuous variables and frequencies and percentages for categorical data, were computed for the different lifetime suicide risk subgroups from the C-SSRS (no lifetime SI or SA, lifetime SI only, lifetime SA). Parametric (e.g. t test, F-test) or non-parametric tests (e.g. chi-square tests) were used to compare distributions of the sociodemographic, psychiatric, and medical characteristics at baseline, and response and remission during the 12-week acute treatment period among the three SI categories. A significance level of 0.0167 (0.05/3) was set to evaluative the 3-way comparisons,
A descriptive exploration of change in SI from baseline to the first post-baseline treatment visit and to the 12-week or final visit was conducted by categorizing participants into 3 groups: (1) those with no SI at baseline (QIDS-C16 item 12 = 0; reference category); (2) those with baseline passive SI (QIDS-C16 item 12 = 1); and (3) those with baseline active SI (QIDS-C16 item 12 = 2 or 3). Only participants who competed at least one post baseline visit were included in this analysis. For those who dropped out before week 12, the last observed value was carried forward.
Generalized estimating equations (GEE) modeling using an alternating logistic regression (ALR) algorithm (Heagerty & Zeger, Reference Heagerty and Zeger1996) for multinomial ordinal data (PROC GEE, SAS version 9.4) was employed to model the association of baseline sociodemographic and clinical characteristics and treatment with repeated measures of SI based on QIDS-C16 item 12 responses at weeks 1, 2, 4, 6, 8,10, and 12. Baseline QIDS-C16 item 12 response was excluded from the repeated measures, but included as a baseline covariate to focus modeling on SI reported during the 12-week treatment period. The following baseline covariates were assessed individually in separate repeated measures models also including treatment and baseline QIDS-C16 item 12 score: age, gender, race, PTSD diagnosis, CGI severity, C-SSRS lifetime and recent severity, C-SSRS suicidal behaviors, QIDS-C16 total score, PHQ-9 total score, Q-LES-Q quality of life score, EuroQOL health state score, BAI total score, positive mental health score, prolonged grief score, ACE score, mixed features score and log transformed durations of present episode (in months) and current treatment trial (in months). A multivariable model was developed using a forward stepwise approach to add covariates significant at the p < 0.05 level and models improving QIC goodness of fit statistics.
Results
Of the 1522 trial participants, 1499 were administered the baseline C-SSRS to measure lifetime and recent (past 3-months) SI and behavior. Table 1 lists the frequencies of graded categories of lifetime and recent suicidal thoughts and behaviors. Almost half of the participants, 749 (47.6%), had lifetime SI without SA while 336 (22.4%) had SI + SA. The other 414 (27.6%) participants had no lifetime SI or SA.
Table 1. Lifetime and recent suicidal thoughts and behaviors (n = 1499)
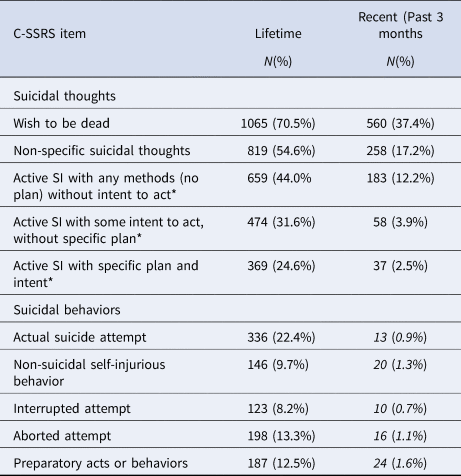
Percents are from total cohort with completed C-SSRS (n = 1499).
* N = 819 for lifetime SI and 258 for recent SI as only asked of those answering ‘yes’ to question 1 (wish to be dead) or 2 (non-specific suicidal thoughts) are asked about these items.
Sociodemographic and clinical features and treatment outcome associated with lifetime SI and SA
Table 2 shows that participants with either lifetime SI only or SI + SA were more likely than those with neither SI or SA (lifetime) to report current SI on the QIDS16 and PHQ-9 SI items, were more markedly or severely ill (CGI-S), had more severe depressions and anxiety, more prolonged grief, adverse childhood events, and diagnosis with PTSD, and less positive mental health. Compared to those with lifetime SI only, participants with lifetime SI + SA were more likely to be female, unmarried, unemployed, and to endorse recent SI (within 3 months) on the C-CSSR, being moderately to severely ill, and to have experienced more early life adversities. There were no differences between the 3 groups on remission to next-step study treatments, but participants with SI + SA where more likely than those with no lifetime SI or SA and those with SI only to respond to next-step treatments.
Table 2. Association of baseline characteristics and response/remission with history of suicide ideation or suicide attempts prior to treatmenta
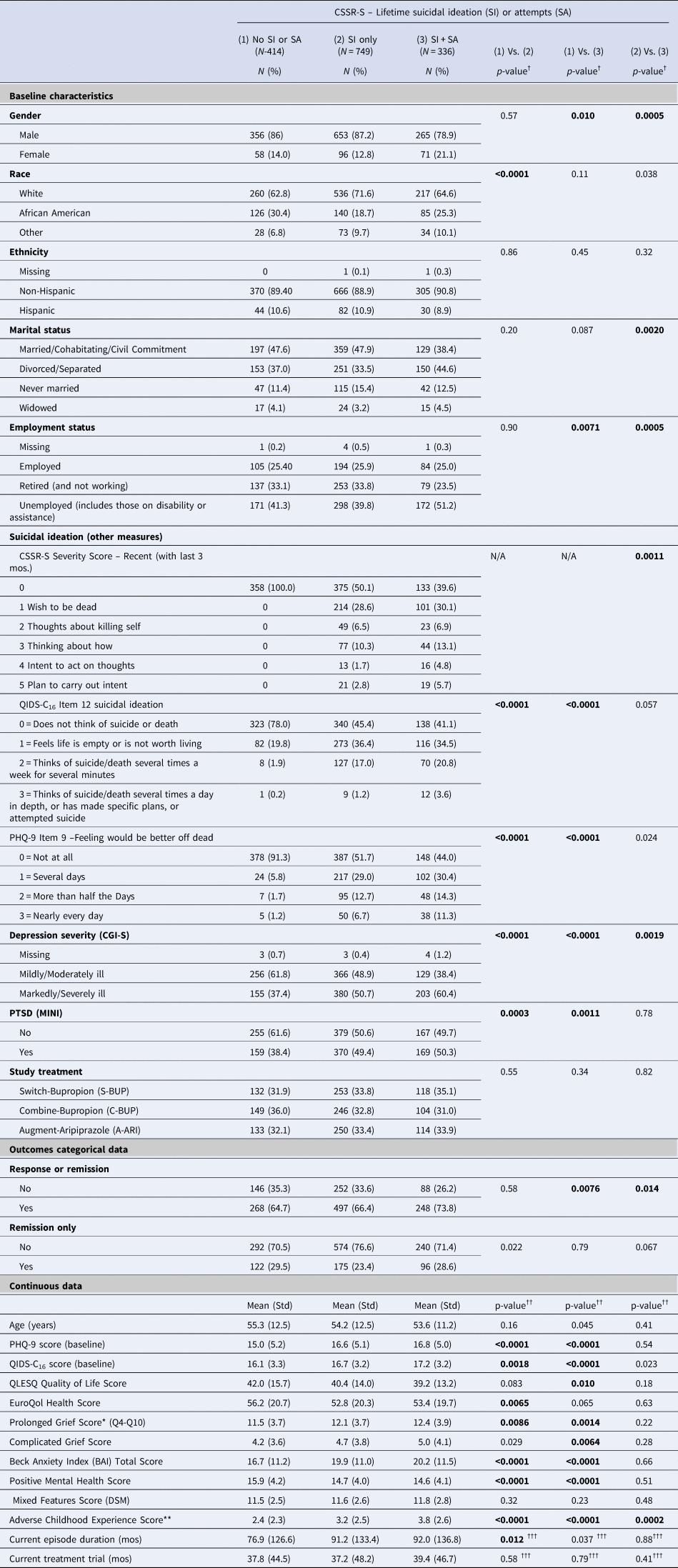
a Values based on available data.
† p-values for categorical measures are from chi-square tests.
†† p-values for continuous measures are from F-tests for one-way analyses of variance.
††† F-tests for duration of current episode and duration of current treatment trial were performed on log transformed data.
†††† −p-values for comparison of severity score levels 0–5 between those with SI and history of SA and those without SA, excluding those with no history of SI.
p-values in bold indicate meeting a significance level p < 0.0167 with no adjustment for multiple comparisons to the values in the table.
* Prolonged grief score was by total score for 7 items from the 9-item Brief Grief Questionnaire (Zisook et al., Reference Zisook, Mohamad, Johnson, Tal, Villarreal, Wilcox and Shear2021).
** Mean score on the 10-item Childhood Adversity Scale (Bernstein et al., Reference Bernstein, Stein, Newcomb, Walker, Pogge, Ahluvalia and Zule2003).
CGI-S = Clinical Global Impression – Severity rating scale.
SI during treatment by baseline SI
Participants who completed at least one QIDS-C16 at a scheduled visit (N = 1492) were included in the analysis of SI reported during follow-up. QIDS-C16 item 12 was used as the measure of SI. At baseline, over half (53.5% or 798/1492) of the participants were categorized as having ‘no SI’, almost a third (31.4% or 468/1492) were categorized as ‘passive SI’ and 15.1% (226/1492) were categorized as having ‘active SI’.
Overall, the number of participants with no SI increased from 798 (53.5%) at baseline to 1065 (71.4%) at the first treatment visit and to 1177 (78.9%) at the final visit. In contrast, the number with passive SI decreased from 468 (31.3%) to 305 (20.4%) at the first visit and to 210 (14.1%) at the final visit; and the number with active SI decreased from 226 (15.1%) to 122 (8.2%) at the first visit and to 105 (7.0%) at the final visit. Emergent SI occurred in 209 participants with no SI at baseline (26.2%), worsening SI in 88 participants with passive SI at baseline (18.8%) and 14 with active SI at baseline (6.8%). Online Supplemental Figure 1 illustrates the overall decline in SI, albeit with fluctuations in the occurrence and intensity of SI during the 12-week treatment period.
SI during treatment by baseline SI and medication groups
Figure 1a shows most participants who start with no SI at baseline do not experience treatment emergent SI, and if they do, it is more likely to be passive than active SI. When emergent SI occurred, it was generally by the first week of treatment and the rate waxed and waned over time. Those treated with A-ARI appeared to have the lowest rates of both passive SI (2.7%) and active SI (0.5%) at the final visit.
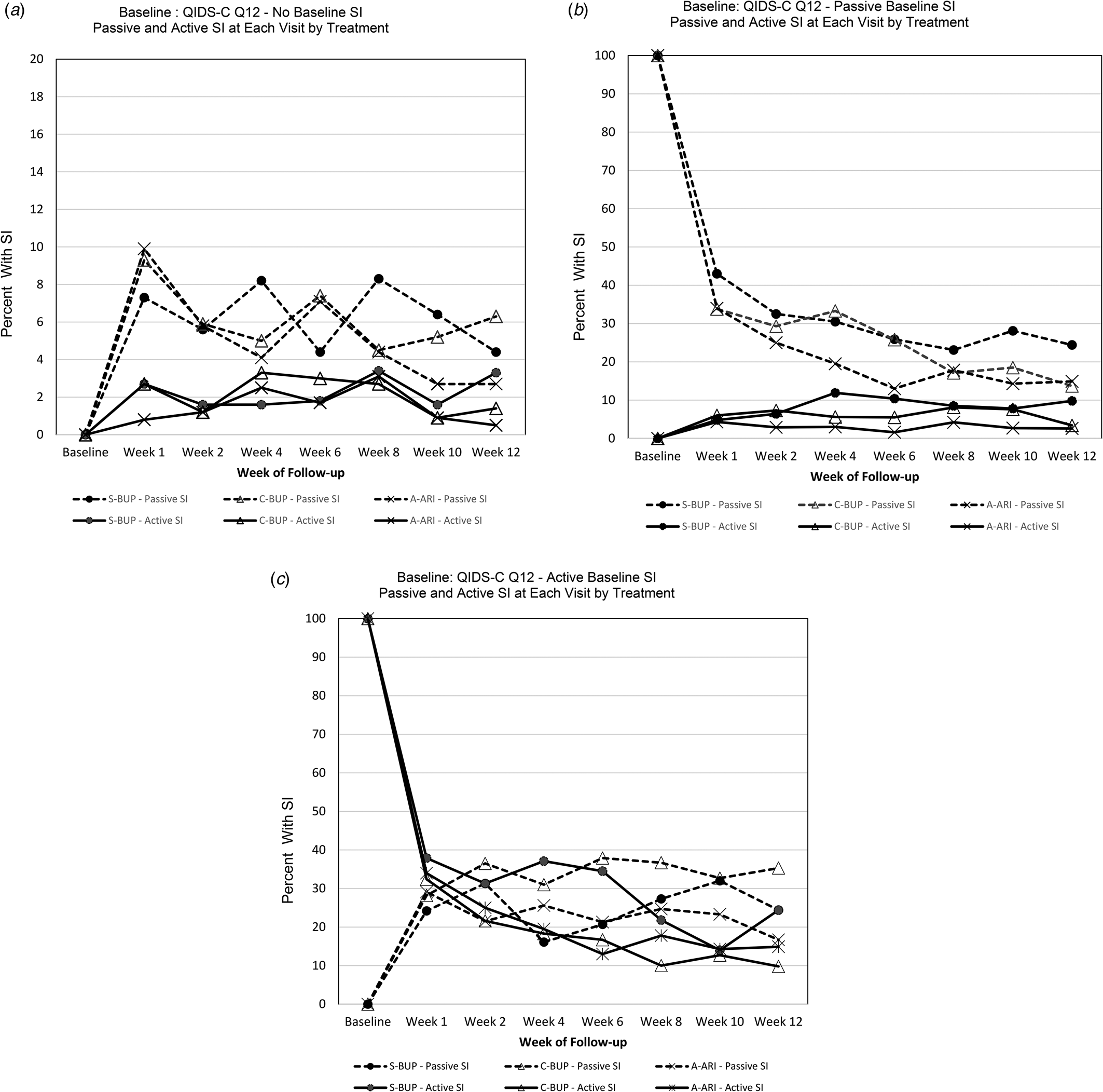
Figure 1. (a–c) Passive and active SI (by item 12 on QIDS-C16) at each visit by treatment from baseline to each week of follow-up by treatment group.
(a) Passive and active SI at each visit by treatment for those with No SI at baseline (Baseline QIDS-C16 = 0) (N = 818).
(b) Passive and active SI at each visit by treatment for those with Passive SI at baseline (Baseline QIDS-C16 = 1) (N = 476).
(c) Passive and active SI at each visit by treatment for those with Active SI at baseline (Baseline QIDS-C16 = 2 or 3) (N = 228).
Percent of SI is the proportion of participants with SI (passive or active) within each treatment group among the participants in follow-up at that visit in that baseline SI group. At baseline percent is therefore either 100 or 0 percent.
Figure 1b illustrates that most participants who started with passive SI at baseline experienced a drop in SI as early as week 1 regardless of treatment group. A smaller proportion of participants began to experience active SI by week 1 and the proportion of those with active SI remained fairly consistent over the 12-weeks of treatment. By the end of treatment, those treated with S-BUP appeared to have the highest rates of both passive SI (24.4%) and active SI (9.8%).
Figure 1c shows most participants who start with active SI at baseline experience a drop in SI as early as week 1 regardless of treatment group. Rates continued dropping through most of the 12-week treatment period. Participants treated with S-BUP had the highest rate of active SI throughout follow-up.
Online Supplementary Table 1 provides week to week data on SI by the three categories of SI and three treatment groups.
Sociodemographic and clinical features and treatment group associated with risk of emergent or worsening SI during 12-week treatment
The evaluation of individual baseline covariables using alternating logistic regression (ALR) models with responses to item 12 on the QIDS-C16 as the outcome measure for SI showed that several demographic factors and most measures of depression severity and associated features were associated with SI reported during follow-up (Table 2). All ALR logistic regression models included baseline QIDS-C16 item 12 response, treatment and visit week to parameterize the effects for repeated SI outcome measures. With no covariate added to the base model, the risk of SI in follow-up was more than 10 times greater for those with active SI (OR 10.64, 95% CI 8.36–13.55) and more than five times greater for passive SI (OR 5.50, 95% CI 4.45–6.81) compared to no SI, and also significantly greater for active SI compared to passive SI (OR 1.94, 95% CI 1.55–2.42). When individual baseline characteristics were added to the model, African American participants showed a significantly lower risk of SI compared to White participants (OR 0.78, 95% CI 0.62–0.99), and females had a lower risk of SI than males (OR 0.76, 95% CI 0.59–0.99). Higher (worse) QIDS-C16 total scores, PHQ-9 score, CGI-S score, BAI score, ACE score, prolonged grief score, and PTSD all demonstrated an increased risk of SI. Lower (worse) PMH score, EuroQOL and Q-LES-Q scores also were significantly associated with greater risk of SI. Compared to participants with no lifetime SI, those with lifetime SI had greater odds of either passive (OR 2.16, 95% CI 1.61–2.90) or active (OR 2.56, 95% CI 1.94–3.39) SI during treatment. Similarly, participants with recent SI were at greater risk for either passive (OR 2.24, 95% CI 1.78–2.82) or active (OR 2.76, 95% CI 2.06–3.68) SI during treatment. Two characteristics of the current MDD were significantly associated with SI: greater duration of episode (OR 1.22, 95% CI 1.07–1.40) and shorter duration of treatment (OR 0.73, 95% CI 0.62–0.87) (Table 3).
Table 3. Evaluation of individual baseline covariates as predictors of suicide ideation during study treatment
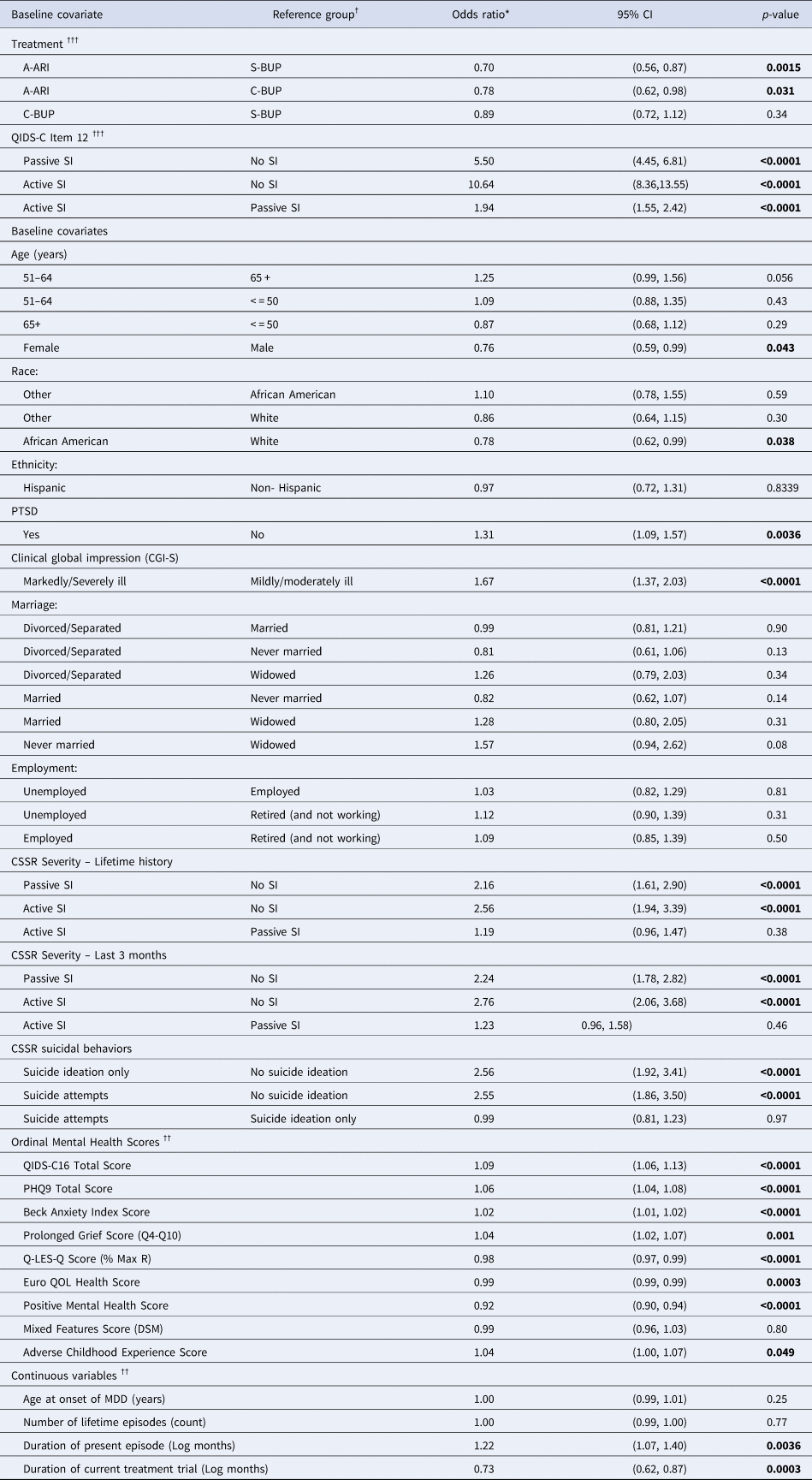
Footnotes:
Results of ALR model predicting QIDS Question 12 score over follow-up.
Model structure: QIDS 12 suicide CAT = Trtcode + Visit week ([repeated] + QIDS12BASE + Covariate.
QIDS12 Base = QIDS-C16 Item 12 response at baseline.
Visit week represents QIDS-C16 Item 12 response recorded at each follow-up visit (weeks 1, 2, 4, 6, 8, 10, 12).
* = Odds ratio represents the relative odds of suicide ideation being reported during follow-up.
† = For categorical variables, odds ratio represents relative increase or decrease in risk of SI within a category relative to the reference category.
†† = For ordinal and continuous variables, odds ratio represents relative increase or decrease in risk of SI for 1 unit increase (i.e. in score, in years, in count, or in log[months]).
††† = Effects for treatment and baseline QIDS-C16 Item 12 in the base ALR model without any additional baseline covariate.
95% CI = 95 percent confidence interval for odds ratio.
Covariates with significant (p < 0.05) increase or decrease in relative odds of suicide ideation relative to reference category were considered for inclusion in the multivariable model.
A multivariable ALR logistic regression model including all covariates that were still significant at p < 0.05 level after adjustment for all other significant variables was developed using a forward stepwise variable selection strategy. Figure 2 illustrates the odds (risk) of SI were still significantly lower in females relative to males (OR 0.65, 95% CI 0.51–0.85, p < 0.005); African Americans relative to Whites (OR 0.71, 95% CI 0.58–0.96, p < 0.05) and other racial groups relative to Whites (OR 0.74, 95% CI 0.52–0.96, p < 0.05); and those with higher positive mental health scores (OR 0.95, 95% CI 0.92–0.97, p < 0.0001). The multivariable also demonstrated the risk of SI was significantly higher in those indicating active lifetime SI on the C-CSSR (OR 2.75, 95% CI 2.04–3.70, p < 0.0001) and active current SI on the QIDS-C16 (OR 4.96, 95% CI 3.74–6.58, p < 0.0001), as well as passive lifetime SI on the CSSR (OR 2.30, 95% CI 1.69–3.12, p < 0.0001) and passive current SI at baseline on the QIDS-C16. (OR 3.74, 95% CI 2.98–4.68, p < 0.0001) relative to no SI during those time periods. Other factors associated with increased risk of SI were more severe depression symptoms assessed by QIDS-C16 (OR 1.07, 95% CI 1.03–1.11, p < 0.005), higher BAI (anxiety) scores (OR 1.01, 95% CI 1.00–1.02, p < 0.05), longer duration of current episode of depression (OR 1.15, 95% CI 1.00–1.32, p < 0.05) and shorter duration of current treatment trial at randomization (OR 0.83, 95% CI 0.70–0.99, p < 0.05). After adjusting the model with the covariates above, co-occurring PTSD, quality of life, general health, childhood adversity, grief score, and mixed symptoms were not significant.
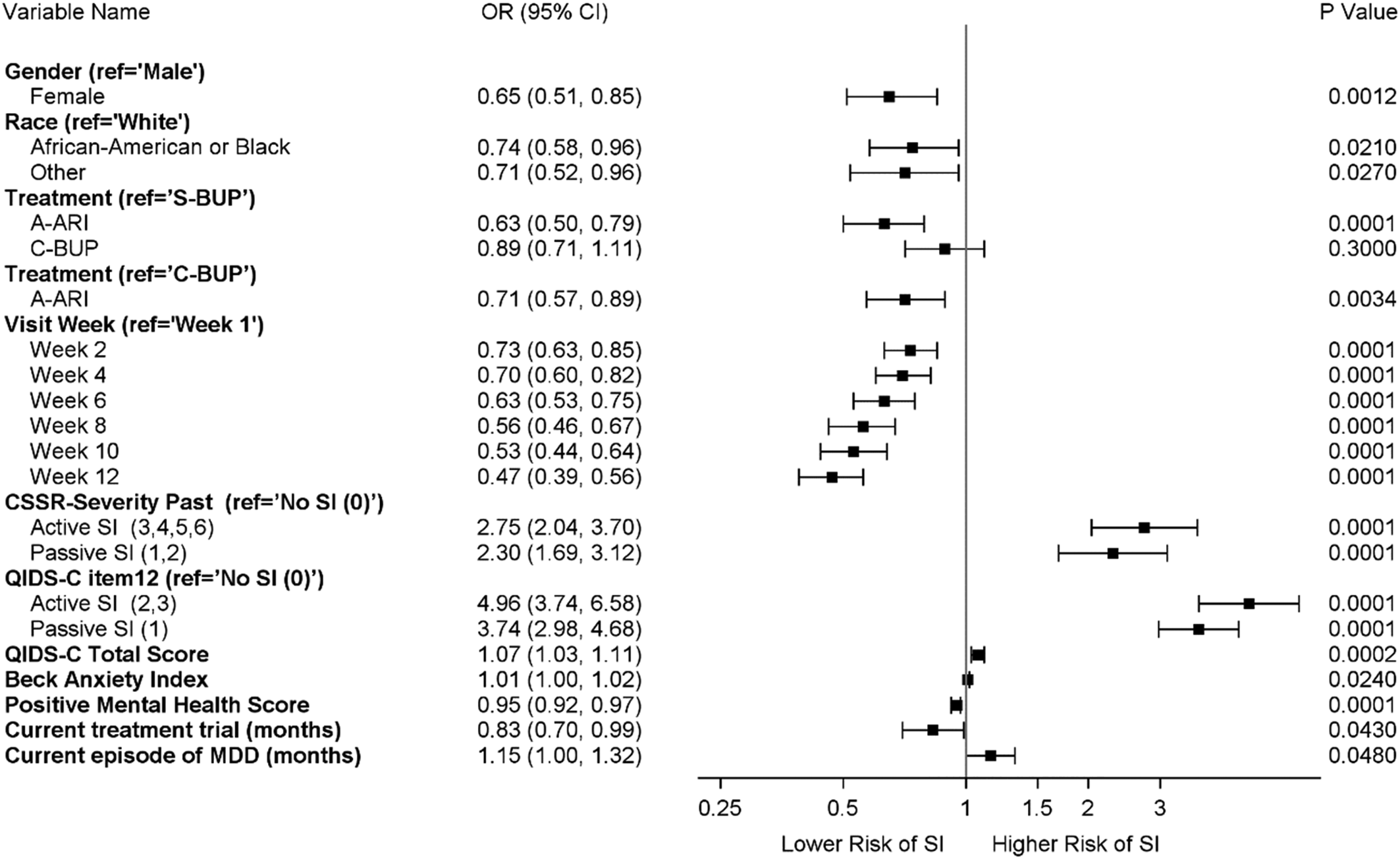
Figure 2. Association of baseline demographic and clinical features with risk of suicide ideation reported during treatment- all participants (n = 1499).
Footnote:
Results of multivariable repeated measures model using an alternating logistic regression (ALR) methodology (Heagerty & Zeger) to accommodate for three ordinal levels of response [0, 1, 2, or 3] to QIDS-C16 Item 12 (suicide ideation).
CSSR-severity past = Highest Columbia Suicide Severity Rating Scale Score for Lifetime or Recent (within last 3 months).
QIDS-C Item 12 = QIDS-C16 Item 12 response at baseline.
Visit week represents QIDS-C16 Item 12 response at each follow-up visit where week 1 is the reference category.
Ref = Reference group.
OR = Odds ratio for relative odds of suicide ideation reported during follow-up within a category relative to the reference category.
95% CI = 95 percent confidence interval for odds ratio.
Risk of SI occurring during treatment was significantly lower for treatment with A-ARI compared to both S-BUP (OR 0.63, 95% CI 0.50–0.79, p < 0.0001) and C-BUP (OR 0.71, 95% CI 0.57–0.89, p < 0.0034). The relative odds of SI decreased each week relative to week 1, indicating improvement over time, with magnitude of SI risk decreasing from 0.73 to 0.47 from week 2 to week 12, p < 0.0001.
Safety
New active SI and SA were reported as serious adverse events. Among the full sample of 1522 participants, there were 25 reports of SI in 24 participants, seven SAs by seven participants, and one suicide. The participant who completed suicide had withdrawn from the study.
Discussion
This large sample of Veteran outpatients who require ‘next-step’ treatments for MDD after a suboptimal response to at least one antidepressant demonstrates remarkably common suicidal thoughts and behaviors in this population and how treatment affects SI: over two-thirds reported a lifetime history of ‘wishing they were dead’; almost one-fourth had made at least one suicide attempt; and SI generally lessened with treatment, albeit with fluctuations in the prevalence and severity over the 12-week course of treatment. We compared features associated with lifetime SI and SA, trajectories of SI during treatment and risk factors for SI occurring during treatment.
We found a number of overlapping features that differentiated those with no lifetime SI or SA from those with SI only or SI + SA, and fewer features that differentiated those with SI only from those with SI + SA. Most of these findings are consistent with previous reports of factors associated with SI and SA in depressed patients: co-occurring PTSD, anxious features, quality of life, severity, and chronicity of MDD, and demographic features (Bachmann, Reference Bachmann2018; Borges, Angst, Nock, Ruscio, & Kessler, Reference Borges, Angst, Nock, Ruscio and Kessler2008; Claassen et al., Reference Claassen, Trivedi, Rush, Husain, Zisook, Young and Alpert2007; Spijker, de Graaf, Ten Have, Nolen, & Speckens, Reference Spijker, de Graaf, Ten Have, Nolen and Speckens2010; Zisook et al., Reference Zisook, Lesser, Lebowitz, Rush, Kallenberg, Wisniewski and Trivedi2011). This study also confirmed previous reports on the importance of childhood adversity (Tunnard et al., Reference Tunnard, Rane, Wooderson, Markopoulou, Poon, Fekadu and Cleare2014; Zisook et al., Reference Zisook, Lesser, Lebowitz, Rush, Kallenberg, Wisniewski and Trivedi2011) and prolonged grief (Zisook et al., Reference Zisook, Mohamad, Johnson, Tal, Villarreal, Wilcox and Shear2021) as risks for adult SI and SA, reinforcing the value of obtaining information about childhood neglect, abuse, trauma, and loss throughout the life cycle in adult patients with MDD and providing additional scrutiny for suicide risk in those with these life experiences. The few studies that have examined differences between those with SI alone from those with SI + SA also reported childhood trauma, anxiety, and PTSD being more prevalent in those with SA (May & Klonsky, Reference May and Klonsky2016; Wiebenga, Eikelenboom, Heering, van Oppen, & Penninx, Reference Wiebenga, Eikelenboom, Heering, van Oppen and Penninx2021). Why is this important? Both SI and SA are risk factors for suicide (Bostwick, Pabbati, Geske, & McKean, Reference Bostwick, Pabbati, Geske and McKean2016; Large, Corderoy, & McHugh, Reference Large, Corderoy and McHugh2021). But most persons with SI do not go on to make attempts (May & Klonsky, Reference May and Klonsky2016), and SA has been identified by the World Health Organization as being the single most robust risk factor for suicide (https://www.who.int/news-room/fact-sheets/detail/suicide). Thus, it is important to identify those who turn thoughts into actions to inform clinical practice and identify targeted suicide prevention interventions (Wiebenga et al., Reference Wiebenga, Eikelenboom, Heering, van Oppen and Penninx2021).
MDD, especially untreated or inadequately treated, is another well documented risk for suicide, and effective treatment of MDD has been demonstrated to reduce risk (Mann et al., Reference Mann, Michel and Auerbach2021; Zalsman et al., Reference Zalsman, Hawton, Wasserman, van Heeringen, Arensman, Sarchiapone and Zohar2016). Our novel finding that SI + SA was associated with a better response to next-step treatments compared to either of the other groups was unexpected. Some previous studies have found that patients with MDD who have SI and a history of SA are less likely to respond to treatment than non-suicidal patients with MDD (Lopez-Castroman, Jaussent, Gorwood, & Courtet, Reference Lopez-Castroman, Jaussent, Gorwood and Courtet2016). Esketamine nasal spray, given in conjunction with an oral antidepressant, is the only approved treatment for MDD with SI or SA (Canuso et al., Reference Canuso, Ionescu, Li, Qiu, Lane, Turkoz and Fu2021). This study potentially broadens the scope of effective treatments for patients with MDD and lifetime SA.
Similar to findings in persons undergoing their first step treatment (Zisook et al., Reference Zisook, Trivedi, Warden, Lebowitz, Thase, Stewart and Rush2009), only a small minority of these next-step participants who had no current SI at baseline developed emergent SI (which, if occurring, was much more likely to be passive than active SI). For those with either passive or active SI at the initiation of next-step treatment, there was a dramatic positive effect within one week for all three treatments. This underscores the clinical importance of next-step medications for patients who fail to have an optimal response to initial trials.
With the possible exception of clozapine for patients with schizophrenia or schizoaffective disorder, the effects of most atypical antipsychotic drugs in reducing suicidal risk have not been adequately tested (Pompili et al., Reference Pompili, Baldessarini, Forte, Erbuto, Serafini, Fiorillo and Girardi2016). The results of this study provide important impetus for further testing. For those with passive SI at treatment initiation, aripiprazole seemed to have an advantage over bupropion whether switched to, or added on to, the prior treatment. The logistic regression analysis confirmed the overall advantage of A-ARI over S-BUP and C-BUP. Perhaps the partial antagonist effect of aripiprazole on the dopamine system accounts for this difference, or conversely if there is a modest dopaminergic agonist effect with bupropion, it may be expressed as a difference in SI during treatment for those with SI at the onset of treatment. Whether these differences in SI are clinically meaningful over time cannot be addressed with this study. Potential benefit must be weighed against the risk of other side effects from aripiprazole.
Although clinical markers of suicidal thoughts and behaviors occurring during antidepressant treatment are not well studied, we were not surprised to find that SI occurring during treatment was associated with SI at baseline, lifetime and recent SI, low positive mental health, anxiety, high severity, and long duration of current episode of MDD. We were not expecting to see females, who are at greater risk for SA in the general population and in this study population, had a reduced risk of SI during treatment. The association of gender with treatment emergent or worsening SI during treatment bears further study. We also were surprised that some of the clinical features that have been associated with SI or SA, such as PTSD, lifetime SA, mixed symptoms, childhood adversity, and prolonged grief did not make it into the final multivariable model. This may be due to the overriding effect of depression severity, the lack of power to fully explore the effects of these important features, or possibly that they are not independent triggers of antidepressant-related emergent or worsening SI.
The effect of all 3 study arms on reducing SI was evident after only 1 week despite low doses used during the first week and meaningful antidepressant effects often lagging by several weeks. This suggests the antidepressant medication effects on SI maybe at least somewhat independent of their antidepressant effects, as has been shown with other agents (Hochschild, Grunebaum, & Mann, Reference Hochschild, Grunebaum and Mann2021). Doses continued to be raised as the treatment target was depression and the management plan was to optimize the dose based on response and side effects. Finding optimal doses to reduce SI may be a worthwhile endeavor for future studies.
Limitations
Study limitations include the naturalistic design comparing three next-step treatment options without a placebo control group. Using SI as a proxy for suicide risk during treatment has limitations given the relatively high prevalence of SI amongst clinical and community samples, and its limited predictive value on its own for suicidal behavior. That all participants were Veterans is both a limitation and a strength: a limitation regarding generalizability to other populations and a strength regarding Veterans being a high-risk group for suicide. Since we did not have data on fluctuations of participant SI prior to study entry, we could not distinguish newly emergent SI from longer-term fluctuations in SI prior to baseline. Only outpatients were included, eliminating the likelihood of enrolling the most severely depressed participants. Several potentially important clinical features, such as personality traits and disorders, were not measured. As the mean duration of the depressive episodes in this trial was >24 months, many participants may have suffered from chronic depression rather than episodic or recurrent MDD.
Conclusions
In these outpatients with MDD of whom close to 25% had previously attempted suicide, SI was substantially reduced in all three 12 week ‘next step’ treatments. However, about one in five either experienced a new onset SI (from no SI to mainly passive SI) or SI worsening which could occur throughout treatment; thus, vigilance for suicide risk through the entire 12-week acute treatment period is necessary. Baseline SI; low positive mental health, anxiety, severity, and duration of current MDD episode; being male and White were associated with a greater risk of emergent or worsening SI. Treatment selection may affect the risk of SI as augmentation with aripiprazole was associated with a lower risk of SI occulting during treatment than either a switch to or a combination with bupropion.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S0033291723003008.
Disclosures and acknowledgements
Dr Zisook reports research support and consultation fees from Compass Pathways, Ltd. Dr Rush reports: Patent Holder: Methods to Identify Patients at Risk of Developing Adverse Events During Treatment with Antidepressant Medication [Patent 7 906 283]; Methods to Predict the Outcome of Treatment with Antidepressant Medication [Patent 7 795 033]. Consultant/Advisory Boards: Compass [Psilocybin]; Emmes Corp [Substance Use Disorder]; Evecxia Therapeutics, Inc [Depression]; Holmusk Technologies, Inc [Depression]; ICON plc [Depression]; Johnson and Johnson [Ketamine]; Liva-Nova [Depression]; MindStreet [Depression]; Neurocrine Biosciences Inc [Depression]; Otsuka-US [Depression]. Speaker's Bureau: Johnson and Johnson [Depression]; Liva-Nova [Depression]. Other Financial Interest: Guilford Press [Royalties]; UT Southwestern [Royalties]. All the relevant financial relationships listed have been mitigated. Mr. Johnson reports his spouse being an employee of and owning stock in Bristol- Myers Squibb. Dr Davis reports receiving grant funding from Tonix and personal fees from Otsuka, Tonix, Lundbeck, and Bracket.
The authors thank the VAST-D staff and patient participants at each of our 35-sites without whom this study could not have been done.
Author's contributions
Each author has read and approved the submitted manuscript. In addition, each author made significant intellectual contributions to the development and completion of the manuscript. This includes involvement in the conception, design, data acquisition, analysis, and interpretation of the data. Each author has agreed to be accountable for all aspects of this work, ensuring there is accuracy and integrity.
Previous presentation
None.
National clinical trials
NCT01421342.








