Article contents
Monotherapy and combination chemotherapy for Chagas disease treatment: a systematic review of clinical efficacy and safety based on randomized controlled trials
Published online by Cambridge University Press: 12 August 2022
Abstract
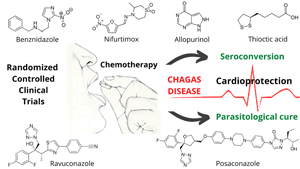
From a systematic review framework, we analysed the clinical evidence on the effectiveness and safety of monotherapy and combination chemotherapy for Chagas disease (ChD) treatment. The research protocol was based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses and patient, intervention, comparison and outcome strategy. Only randomized controlled trials (RCT) were retrieved from Embase, Medline, Scopus and Web of Science databases. Diagnostic tools, treatment protocols, seroconversion rates and adverse events were investigated. Fifteen RCT mainly concentrated in endemic countries were identified. ChD diagnosis was mainly based on haemagglutination, immunofluorescence, enzyme-linked immunosorbent assay and polymerase chain reaction. Benznidazole (BNZ), nifurtimox, fosravuconazole, posaconazole, allopurinol and thioctic acid were the identified drugs. The best negative seroconversion results (100, 96, 94 and 91.3%) were, respectively, based on BNZ (5 mg kg day−1, 200 mg day−1, 150 mg day−1 and 2.5 mg kg−1) administration for 60 days. Negative seroconversion was not achieved with allopurinol (300 mg day−1 for 60 days). Adverse reactions ranged from 5 to 73% in patients receiving antiparasitic chemotherapy. Treatment discontinuation (1.5–57%) was mainly associated with gastrointestinal, cutaneous and neurological manifestations. Current RCT-based evidence indicates that BNZ is the most viable option for ChD treatment. However, new protocols need to be developed to mitigate side effects and increase patient adherence to antiparasitic chemotherapy. Therefore, shorter regimens, lower concentrations and treatments combining BNZ with posaconazole, fosravuconazole or ravuconazole may be viable to ensure comparable efficacy to BZN-based monotherapy, contributing to reduce dose- and time-dependent toxicity reactions.
- Type
- Systematic Review
- Information
- Copyright
- Copyright © The Author(s), 2022. Published by Cambridge University Press
References
- 10
- Cited by



