Article contents
Optical biosensors utilizing graphene and functional DNA molecules
Published online by Cambridge University Press: 03 April 2017
Abstract
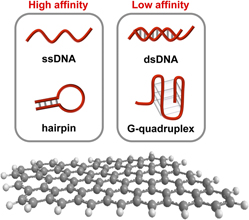
Single-stranded DNA molecules capable of molecular recognition and catalysis can now be routinely generated via the technique of in vitro selection. When coupled with adequate signal transduction modes, these synthetic functional DNA species represent a potential paradigm shift in the research and development of biosensors to meet the challenges of our rapidly changing world. Coupling functional DNA molecules with graphene materials for the design of optical biosensors has become an exciting research area in recent years, mostly because graphene materials are not only excellent quenchers of fluorescence, but they also display considerably different affinities for free and ligand-bound functional DNA molecules. We will discuss notable progress in this area in this mini-review by highlighting representative studies.
Keywords
- Type
- Invited Articles
- Information
- Journal of Materials Research , Volume 32 , Issue 15: Focus Issue: Two-Dimensional Nanomaterials for Biosensors , 14 August 2017 , pp. 2973 - 2983
- Copyright
- Copyright © Materials Research Society 2017
Footnotes
Contributing Editor: Venkatesan Renugopalakrishnan
References
REFERENCES
- 7
- Cited by



