Article contents
Electric field effect on chemical and phase equilibria in nano-TiB2–TiO2–TiBO3 system at <650 °C: an in situ time-resolved energy dispersive x-ray diffraction study with an ultrahigh energy synchrotron probe
Published online by Cambridge University Press: 19 December 2016
Abstract
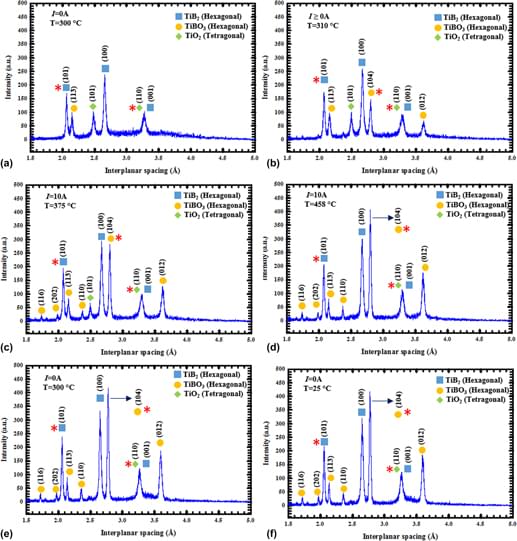
Nano-TiB2 powder of 58 nm size with TiO2 and TiBO3 as secondary phases was heated with 20 °C to <650 °C in argon while applying an electric field. The powder became conductive at 520 and 305 °C (T onset) for 16 and 40 V/cm, respectively, at which point current bursts of 4.5 and 10.0 A (peak value) were observed. Current bursts were accompanied by >1% TiB2 unit cell expansion, exceeding zero field thermally induced expansion. The current bursts also induced nonisothermal reaction between TiB2 and TiO2, yielding TiBO3 that is absent with no field. Increase from 16 to 40 V/cm shifts the TiB2 → TiBO3 reaction forward, decreases T onset but increases reaction rate. Analysis using Van’t Hoff relation, including electrochemical effects, precluded possibility of appreciable Joule heating, which was supported with adiabatic internal temperature calculations. The observed low temperature oxidation of TiB2 to TiBO3 that is electrochemically driven and is mediated by the TiO2 solid electrolyte.
- Type
- Articles
- Information
- Copyright
- Copyright © Materials Research Society 2016
References
REFERENCES
- 9
- Cited by



