INTRODUCTION
Toxoplasma gondii is a worldwide protozoan parasite that infects humans and warm-blooded animals. This parasite is of high veterinary and medical importance because it may cause abortion or congenital infection in its intermediate hosts, including humans [Reference Dubey and Beattie1, Reference Tenter, Heckeroth and Weiss2]. Felids, specifically the domestic cat (Felis catus), are the definitive hosts and shed millions of oocysts into the environment soon after primary infection [Reference Dubey and Beattie1]. Ingestion of oocysts is an important source of infection in intermediate hosts, as shown by the quasi-absence of T. gondii infection in areas without cats [Reference Wallace3]. It is also an important route of infection in humans, since 6–17% of infections in a European multicentre study were attributed to contact with soil [Reference Cook4]. In a recent study, Dabritz et al. [Reference Dabritz5] estimated that the annual burden of oocysts in the environment ranged between 94 and 4671 oocysts/m2, depending on the assumption of the number of oocysts excreted by an infected cat. This level is probably variable in space, and possibly over time, depending on cat density and incidence of T. gondii infection in cats. In order to assess the spatio-temporal variability of the risk of T. gondii infection for humans and intermediate hosts, it is necessary to estimate and understand the causes of variation of T. gondii infection incidence in cats.
Incidence of T. gondii in cats may depend on climatic factors, for several reasons. First, the life cycle of T. gondii includes a free-living phase and survival of oocysts may be affected by high temperatures and/or low humidity [Reference Dubey6, Reference Frenkel, Ruiz and Chinchilla7]. Other indirect mechanisms may play a significant role. Population dynamics of rodents, which are the main intermediate hosts of the parasite, are affected by climate-driven vegetation growth [Reference Stenseth8]. Specifically, when winter survival is high, rodent populations comprise many adult or old individuals, which are the age groups most often infected, thus the risk of encountering an infected prey should increase after mild winters.
Antibody prevalence in cats is known to vary among geographical areas [Reference Dubey9, Reference Silva10], but the relationship between risk of T. gondii infection and climatic conditions has not been investigated quantitatively. In a previous study, the highest seroprevalence of T. gondii in an urban population of domestic cats was observed in hot and moist years [Reference Afonso, Thulliez and Gilot-Fromont11].
This paper is based on long-term epidemiological monitoring in three populations of domestic cats in rural France. We wished to analyse the spatio-temporal variability in incidence risk of T. gondii infection in rural cats, taking into account climatic and other variables known to be related to infection. We also aimed to estimate the rate of soil contamination by oocysts and relate cat incidence rates to climatic conditions, spatial distribution and social organization of cats.
MATERIALS AND METHODS
Populations and sampling
Three cat populations in eastern rural France were monitored: Aimargues (AIM), Barisey-la-Côte (BLC) and Saint-Just-Chaleyssin (STJ) (Fig. 1). In general, maximum daily temperatures increased and rain decreased from north to south: maximum daily temperatures were highest and rain was lowest in the south (AIM; Fig. 2). Meteorological variables were obtained from MétéoFrance as follows: temperature was measured as the mean temperature of the hottest 10-day period in the preceding year, and rain was measured as the mean precipitation per 10-day period over the year. Because we hypothesized that the intensity of heat periods during each 10-day period should reflect the effect of high temperatures on oocyst infectivity, we took into account the maximal value of the mean daily temperature per 10-day period observed during summers of the study periods. Concerning precipitation, we hypothesized that moisture was a determinant for oocyst survival all year round and we thus used mean precipitations per 10-day period over the study periods.

Fig. 1. Location of the study sites in France.

Fig. 2. Mean temperature of the hottest 10-day period in the preceding year and mean precipitation per 10-day period over the preceding year. Aimargues (▵, 1991–1998), Barisey-La-Côte (▪; 1992–2004), Saint-Just-Chaleyssin (![]() , 1991–2005). (Data from MéteoFrance.)
, 1991–2005). (Data from MéteoFrance.)
Each population was stable in terms of size, density, sex ratio, and age structure but populations differed in size and density (range of 60–300 cats and 120–200 cats/km2) [Reference Courchamp12, Reference Pontier and Natoli13]. Most cats were fed by their owners but also preyed on small mammals and birds. Cats lived alone in the owners' homes or in small- to medium-sized groups (ranging generally from 2 to 16 cats), with mothers and kittens forming the core groups, which sometimes also included adult males.
An epidemiological study was carried out in the three populations for different durations: 1991–1998 at AIM, 1992–2004 at BLC and 1991–2005 at STJ (BLC and STJ were not investigated in 1995). Once yearly (in April at AIM, September at BLC and July at STJ), cats were either caught directly in the owner's house or trapped in baited cages. Cats were anaesthetized using a mixture of ketamin chlorhydrate (15 mg/kg Imalgène 1000®, Rhône Mérieux, France) and acepromazin (0·5 mg/kg Vétranquil®, Sanofi, France). Cats were then weighed and sexed, and blood samples were collected by jugular puncture. Cats were permanently marked with a subcutaneous electronic transponder at first capture for subsequent identification.
Age was known to the nearest month for individuals born during the study (60·2% of all cats) and was estimated to nearest year for other cats according to owners' information, general appearance, tooth development and body mass. During each trapping session, all untrapped cats that were seen on the site were recorded, particularly to estimate the number of cats in each group. Social groups are here defined as aggregations of mother–kitten groups around resource centres [Reference Turner and Bateson14]. The experimental protocol was approved by the Veterinary Services of the Rhône Department.
Serological method
Blood samples were centrifuged and stored at −20°C. Sera were diluted twofold, starting at a dilution of 1:20 and tested for anti-T. gondii IgG antibodies using the modified agglutination test [Reference Dubey and Desmonts15]. Cats were considered positive to anti-T. gondii antibodies when their titre was ⩾1:40 [Reference Afonso, Thulliez and Gilot-Fromont11].
Seroprevalence and incidence rate
Cats were recaptured (1–10 captures per cat) when they were present in the study site. Because cats having anti-T. gondii antibodies probably remain seropositive throughout life [Reference Dubey and Beattie1, Reference Afonso, Thulliez and Gilot-Fromont11], estimation of seroprevalence for T. gondii infection was calculated by taking into account the longitudinal aspect of this study. Seroprevalence in the three cat populations was thus estimated by randomly selecting one measurement for each cat. We then calculated the proportion of cats with an antibody titre of at least 1:40. In order to simulate a large number of the possible arrangements using one measurement per cat, this calculation was repeated 1000 times. Overall seroprevalence was then estimated as the mean of the proportions of positive cats in the 1000 re-samplings.
We also estimated incidence rate and rate of seroconversion in each population using the follow-up data. All cats were included in the estimation. The number of cats that seroconverted during the study period was recorded (i.e. those that were seronegative at the beginning of the study and became seropositive during the study) and the cumulative follow-up period for all initially negative cats was considered in order to obtain the annual incidence rate. The latter was then multiplied by cat density in each population to obtain an estimate of the expected rate of seroconversions per hectare per year.
In order to estimate the oocyst burden of T. gondii in the environment, we assumed that primary infected cats shed between 1 and 50 million oocysts, on average [Reference Dabritz5]. The annual burden/m2 was then estimated by multiplying the number of seroconversions/km2 per year in each population by 1 million or 50 million to obtain ranges.
Variables associated with incidence risk
We attempted to relate the probability of seroconversion between year t and year t+1 (i.e. incidence risk) to environmental factors that were measured during year t. Because trapping sessions occurred annually, seroconversions were considered as a change in antibody titre (from below to above 40) between two successive captures, based on a 1-year interval between captures. For this reason, the analysis of incidence risk was only possible for cats aged at least 1 year old.
The study of variables associated with incidence risk in recaptured cats was conducted as a case-control study. Positive cases were cats that seroconverted between two successive captures. Controls were cats that remained seronegative throughout their entire monitoring period. A logistic regression was used to relate seroconversion probability to predictor variables present at time t. For seroconverted cats, we used data from the capture that preceded the first positive test. For seronegative cats, we randomly selected one of the captures that preceded a seronegative result, and thus, conditions at the last seronegative capture were not used.
Several variables that are known to influence the risk of T. gondii infection in cats were taken into account in this study. These variables were total number of cats in the group (tnoc), number of kittens aged <6 months in the group (nok) and roaming habit (roam), a dichotomous variable (free-roaming vs. not allowed to roam). Because T. gondii infection seroprevalence increases linearly with age [Reference Afonso, Thulliez and Gilot-Fromont11], one could expect incidence risk not to be related to age; however, age (as a continuous variable) was tested to confirm this prediction. Moreover, cats living in social groups should be more exposed to parasites than solitary cats, because of a high risk of contact with soil or water that has been contaminated by a newly infected cat or ingestion of a contaminated prey [Reference Gauss16]. Further, the risk of acquiring T. gondii infection increases with level of predation in the diet, thus cats that are allowed outdoors are generally more often infected than cats remaining indoors [Reference Miró17].
Spatio-temporal variability in incidence risk
We estimated the level of spatio-temporal variability in incidence risk, and also quantified the contribution of meteorological factors to the variability of incidence risk. Thus, the effects of the studied population (pop) and trapping year were included in the logistic model as qualitative variables. Variables reflecting the variability of meteorological conditions were the mean temperature of the hottest 10-day period during the year following the capture (tmax) and mean precipitation per 10-day period (rain) during the year following the capture. In addition, we supposed that an interaction between tmax and rain could be detected: both hot and moist or moderate and dry weather have already been related to high antibody prevalence in cats [Reference Afonso, Thulliez and Gilot-Fromont11].
We then adjusted three models: the first one included no spatio-temporal variability and variables age, tnoc, nok and roam (model 0). The second model took into account variables from model 0 plus spatio-temporal variability as year and pop (model ST), while the third model included variables from model 0 plus the meteorological factors of tmax and rain and their two-way interaction (model M). The level of spatio-temporal variability was measured as the difference in deviance (called DevST) between model ST and model 0. The deviance explained by climatic variables was measured by the difference in deviance (DevM) between model M and model 0. The ratio DevM:DevST estimated the proportion of spatio-temporal variability explained by meteorological conditions. Moreover, because model M was included in model ST, we tested whether significant spatio-temporal variability remained in the dataset after taking into account meteorological factors using a likelihood ratio test (LRT) comparing model M and model ST.
Models were compared using Akaike's Information Criterion (AIC) [Reference Burnham, Anderson, McCullough and Barrett18]. AIC differences between the best model and all other models considered (Δi=difference between AIC and the lowest AIC value) were calculated in order to determine the relative ranking of each possible model. The model with the lowest AIC represented the best compromise between residual deviance and number of parameters [Reference Burnham, Anderson, McCullough and Barrett18]. When Δi<2, we selected the most parsimonious model, i.e. that with fewest parameters. We calculated odds ratios and 95% confidence intervals (CIs) to measure the strength of association between each variable and serological status while controlling for other variables. The overall fit of the final logistic equation was assessed using the Hosmer–Le Cessie test [Reference Hosmer19].
All statistical procedures were performed using R 2.7.1 software (R Development Core Team, 2005; http://www.R-project.org).
RESULTS
A total of 861 cats (458 females, 403 males) were trapped during the study (224 cats at AIM, 226 at BLC, 411 at STJ). Of the 1488 blood samples collected, 37% were from recaptures of cats (1–10 captures per cat). When we randomly selected one measurement per cat, age structure differed between the three populations (χ2, P<0·001): BLC and STJ contained younger cats than AIM (Table 1). A total of 302 cats were tested for T. gondii antibodies when they were aged <1 year (131 females, 171 males; median age 4 months). Of these, 100 juveniles carried antibodies against T. gondii: seroprevalence in juveniles was thus 33·1% (95% CI 28·1–38·6).
Table 1. Main characteristics of the studied cat populations
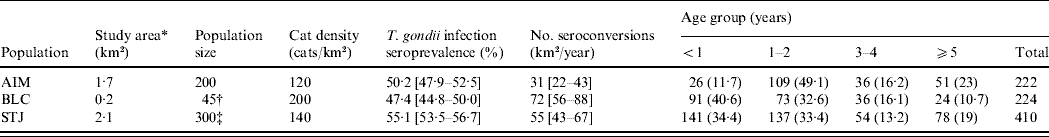
AIM, Aimargues; BLC, Barisey-La-Côte; STJ, Saint-Just-Chaleyssin.
Values under ‘Age group’ are the number (%) of cats in each age group. Values within square brackets are 95% confidence intervals.
* Surface area used by cats.
† See Pontier et al. [Reference Pontier, Rioux and Heizmann33].
‡ See Pontier & Natoli [Reference Pontier and Natoli13].
Prevalence, incidence and expected number of seroconversions
By randomly selecting one blood sample for each of the 861 tested cats, 454 cats showed an antibody titre of at least 40. Overall seroprevalence was estimated at 52·7% (95% CI 49·4–56·0). The proportion of seropositive cats did not differ significantly in the three populations (χ2, P=0·153). Seroprevalence was 50·2% (95% CI 47·9–52·5) at AIM, 47·4% (95% CI 44·8–50·0) at BLC and 55·1% (95% CI 53·5–56·7) at STJ (Table 1).
At AIM, 22/50 cats seroconverted during the cumulative monitoring period of 85 cat-years. The observed incidence at AIM was thus 0·26 seroconversions/cat per year (95% CI 0·18–0·36). At BLC, 49/71 cats seroconverted during a period of 136 cat-years, which represented 0·36 infections/cat per year (95% CI 0·28–0·44). The highest incidence was observed at STJ, as 52/138 cats seroconverted during a period of 132 cat-years, which represented 0·39 infections/cat per year (95% CI 0·31–0·48). Taking into account the density of cats, we expected the number of seroconversions/km2 per year to be 31 at AIM, 72 at BLC and 55 at STJ (Table 1). Using the same hypothesis as [Reference Dabritz5] concerning the quantity of oocysts shed by cats (1:50 million), the annual burden of T. gondii oocysts/m2 shed into the environment was thus calculated as 31–1550 at AIM, 72–3600 at BLC and 55–2750 at STJ.
Variables associated with incidence risk in adults
Seroconversion occurred between successive captures for 82 cats (19 at AIM, 27 at BLC, 36 at STJ), whereas 64 cats remained seronegative for all trappings (29 at AIM, 15 at BLC, 20 at STJ). Of the 146 cats followed, 89 were females and 57 were males. Cats were either solitary or lived in groups ranging from 2 to 16 individuals, with from 1 to 7 kittens. Most cats were free-roaming (n=128), while only 18 were not allowed to roam by their owner. Because of the unbalanced nature of this factor, interactions with roam were not included in models.
The model retained to explain incidence risk without any spatio-temporal variability (model 0) included two parameters: the number of kittens in the group (nok) and the roaming habit of cats (roam) (Table 2a). Cats living with many kittens appeared to have a high risk of seroconversion: the likelihood of seroconversion increased by 1·5 (95% CI 1·1–2·1) with each additional kitten in the social group. Moreover, although non-roaming cats were rare in the three cat populations, roaming strongly influenced incidence risk: cats not allowed to roam had a 4·1-fold lower probability of seroconversion than free-roaming cats (95% CI 2·2–7·6). Total number of cats in the group (tnoc) was not retained in the final model.
Table 2. Model comparisons of the incidence risk for Toxoplasma gondii infection in adult cats from rural populations
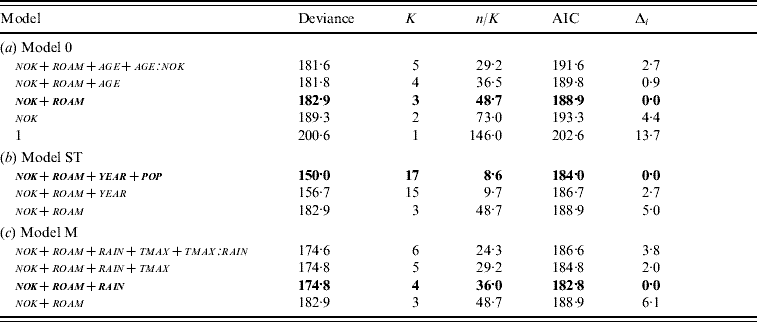
K, Number of estimated parameters; n/K, number of observations/K; AIC, Akaike's Information Criterion; Δi, difference between AIC and the lowest AIC value; nok, number of kittens; roam, roaming habit; age, age; tmax, maximum temperature; rain, mean precipitation.
Values in bold correspond to the selected models. (a) Model 0=the model without spatio-temporal variability; (b) model ST=the model including variables from model 0 plus spatio-temporal variables (year and population); (c) model M=the model including variables from model 0 plus meteorological factors (annual maximum temperature and mean precipitation).
The best model with spatio-temporal variables (model ST) included year of trapping (year) and population (pop), in addition to variables retained in model 0 (Table 2b). The probability of seroconversion was higher in BLC and STJ compared to AIM (Table 1). The interaction between year and population was not tested because the three populations were sampled in slightly different years.
The best model using climatic variables (model M) included variables retained in model 0 plus mean precipitation prior to a new seropositive capture (rain) (Table 2c). Every additional 1 mm in precipitation increased the chance of seroconversion by 1·1 (95% CI 1·0–1·1). Based on this value, we calculated that incidence risk (i.e. likelihood to seroconvert) between the least and the most rainy year increased by 24·9 (95% CI 23·7–26·2) at AIM (12·5 mm vs. 35·6 mm), by 11·0 (95% CI 10·5–11·6) at BLC (22·5 mm vs. 35·6 mm) and by 8·1 (95% CI 7·7–8·5) at STJ, which showed the least variation in precipitation (29·7 mm vs. 37·2 mm). In order to compare populations, we calculated odds ratios, based on average rainfall in each study site (24·1 mm at AIM, 27·6 mm at BLC, 33·5 mm at STJ). We thus determined that incidence risk was 3·8 times higher (95% CI 3·6–7·1) at BLC compared to AIM, 6·4 times higher (95% CI 6·1–6·7) at BLC than STJ, and 10·2 times higher (95% CI 9·7–10·7) at STJ than AIM. Moreover, the model predicted that almost all individuals would seroconvert in rainy years in the largest groups (seven kittens). Overall, incidence risk was high when rain was >25 mm per month and social groups comprised more than one kitten (Fig. 3). Maximum daily temperature (tmax) and the interaction rain∗tmax were not retained in model M. The Hosmer–Le Cessie test showed a good fit of the model selected (P=0·486). Because model M was more parsimonious than model ST, we selected the former model as the final model for the study of incidence risk in cats (Table 3).
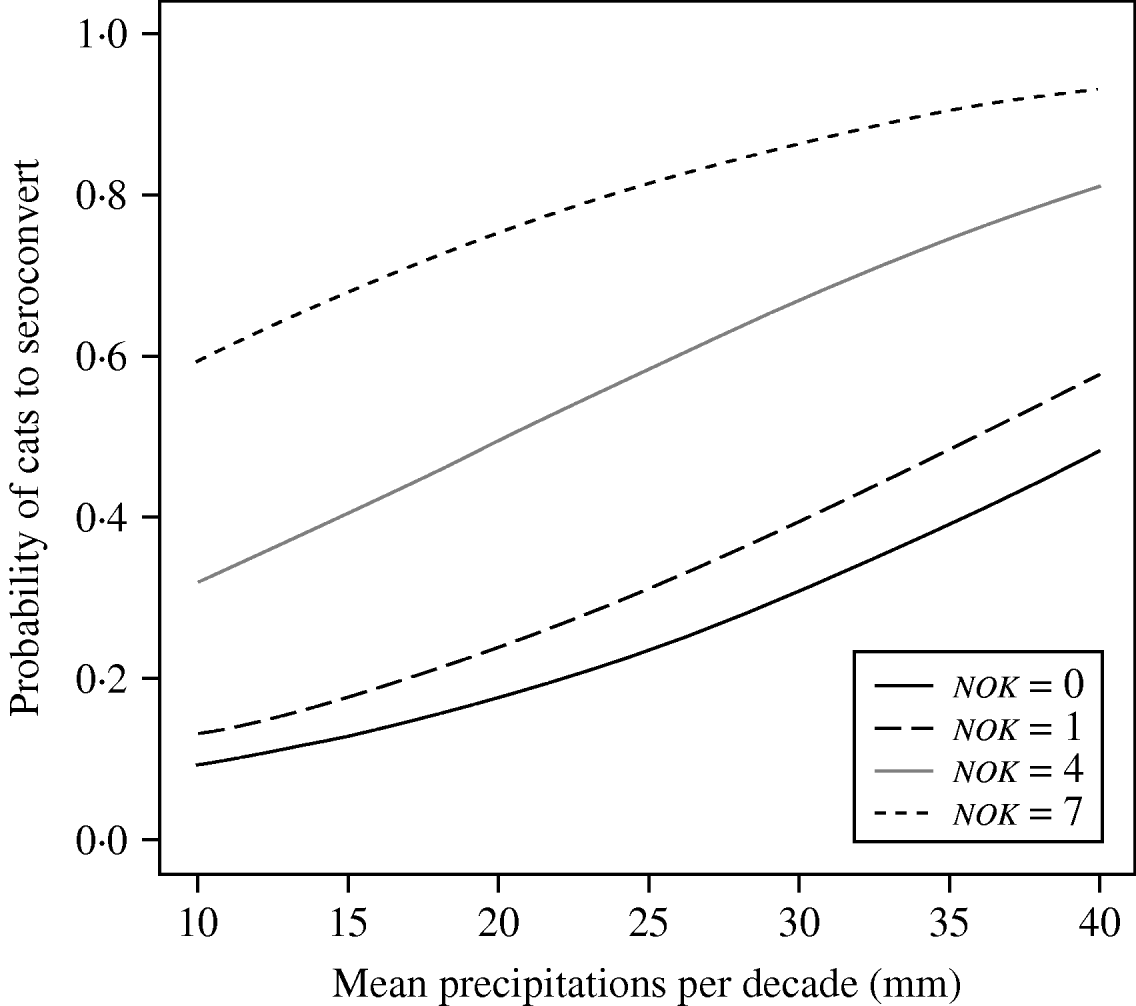
Fig. 3. Predictions of the final model for T. gondii infection incidence risk in free-roaming, adult cats. Mean precipitation was that observed in the three studied populations during the entire study period (1991–2005). Predictions were calculated for four different numbers of kittens (nok) in the cat group.
Table 3. Parameters of the meteorological model (model M) that were associated with the probability of seroconversion for anti-Toxoplasma gondii antibodies in recaptured cats

OR, Odds ratio; CI, confidence interval; nok, number of kittens; roam, roaming habit; rain, mean precipitation.
The differences in model deviances were assessed as DevST (34·3) and DevM (10·4). Therefore, using proportions, 30·4% of spatio-temporal variability of incidence risk could be explained by meteorological conditions. Finally, significant spatio-temporal variability remained in the dataset after taking into account meteorological factors (LRT, P=0·016): pop remained related to incidence risk (Δi=3·8), while the effect of year was no longer related to incidence risk (Δi=0·0).
DISCUSSION
This study constitutes a first estimate of annual T. gondii oocyst burden in a rural environment, based on incidence data in cats. It also represents the first quantitative measure of spatio-temporal variability in T. gondii infection risk and the role of meteorological conditions in explaining this variability.
Seroprevalence and incidence in rural cat populations
Previous studies have estimated T. gondii infection seroprevalence in cats to range between 9% and 74% [Reference Tenter, Heckeroth and Weiss2]. However, sampling methods were generally carried out at veterinary facilities and/or animal shelters, which made it difficult to compare seroprevalence levels because samples were biased in favour of one kind of individual. Our results were based on exhaustive censuses of the populations where high proportions were captured (15–60% according to the site and year) and representativeness of the samples was assessed for sex, age and neutering status [Reference Fromont, Artois and Pontier20]. This sampling plan thus provided an estimate of the spatio-temporal variability of seroprevalence for T. gondii infection in cat populations living in rural France. Seroprevalence ranged from 47·4% to 55·1% in our study, indicating that cats were often exposed to T. gondii.
The number of seroconversions per year and per surface area varied between the three studied populations: we estimated that 31 seroconversions/km2 per year occurred at AIM, 72 at BLC and 55 at STJ. Assuming that seroconverted cats shed 1 million to 50 million oocysts [Reference Dubey and Beattie1, Reference Dubey and Jones21], we estimated the rate of soil contamination in rural France to be 31–3600 oocysts/m2 per year. These values are close to those calculated by Dabritz et al. [Reference Dabritz5] in California, with an annual burden of 94–4671 oocysts/m2 under the same hypotheses. Our estimates were obtained using field data on cat incidence rather than coproscopic examination compared to cat faecal excretion rates. Although seroprevalence was higher in rural sites (52·7%) than in our previous urban study (18·6%) [Reference Afonso, Thulliez and Gilot-Fromont11], the predicted rate of seroconversion was higher in the urban site (400 compared to 31–72 seroconversions/km2 per year) due to higher cat density, thus we expect soil contamination to be higher in the urban than in the rural site.
Local variables associated with incidence risk
Taking advantage of long-term monitoring, we conducted an innovative analysis of T. gondii infection based on incidence risk in cats. Because we observed only 2–3 recent seroconversions per year in each cat population (19 at AIM during an 8-year survey, 27 at BLC during a 12-year survey, 36 at STJ during a 14-year survey), estimates for the spatio-temporal parameters may be not accurate. However, the strong associations between variables and incidence risk in adult cats and the similarity of results across the three populations lead us to propose that the detected effects are biologically significant.
As predicted, the number of kittens in the group and roaming habits were related to incidence risk in adult cats. Cats tend to use common defecation sites around feeding sites when they live in groups [Reference Turner and Bateson14, Reference Yamaguchi22]. Social cats thus have more opportunity to come in contact with areas contaminated by other cats than solitary cats, and so may have higher risks of ingesting oocysts or prey contaminated by concentrated oocysts in a restricted area. Kittens could represent a special exposure risk since they generally move only short distances away from feeding points to defecate [Reference Turner and Bateson14], and probably excrete oocysts in places heavily used by other cats. Moreover, kittens could be particularly affected by seroconversion because of high exposure rates. Further, the presence of kittens may encourage females to hunt intensively and share prey, in which case a single prey item may infect several cats.
Roaming habits are well known to increase risk of T. gondii infection in cats [Reference Tenter, Heckeroth and Weiss2, Reference Gauss16]. Free-roaming cats could be exposed to the parasite through contaminated intermediate hosts such as small mammals and birds, or infectious oocysts from the environment. Roaming may also reflect several at-risk behaviours: roaming, reproductive status and group size are all correlated in the studied populations [Reference Fromont, Artois and Pontier23]. Indeed, free-roaming cats are often not neutered and live in large social groups. Neutered cats have less motivation to venture outdoors than non-neutered cats [Reference Turner and Bateson14]. As a consequence, neutering of cats should reduce the risk of acquiring T. gondii infection.
Spatio-temporal variability of incidence
After accounting for local factors, both inter-annual and inter-population variability in incidence were significantly explained by mean precipitation. Incidence risk increased during rainy years, especially when mean precipitations per 10-day period were >25 mm. Such a relationship between infection and precipitation has also been proposed for another parasite in cats (Bartonella henselae [Reference Jameson24, Reference Maruyama25]), T. gondii infection in humans [Reference Berger26–Reference Sukthana28] and T. gondii infection in intermediate host species (in rabbits Oryctolagus cunniculus [Reference Almeria29], in roe deer Capreolus capreolus [Reference Gamarra30]), but has never been formally tested for T. gondii infection in cats. As stated earlier, rain may have several effects on exposure to T. gondii through increased oocyst survival [Reference Frenkel, Ruiz and Chinchilla7] and altered population dynamics of rodent hosts [Reference Stenseth8].
Based on our study of incidence risk, we assume that the level of soil contamination should be highest in rainy sites and years, and in sites where young and roaming cats are most abundant. This pattern may partly explain the geographical variation in risk for T. gondii infection humans. Thus, the lowest seroprevalences of T. gondii in humans have been observed in humans living in the most arid regions of the USA [Reference Vollaire, Radecki and Lappin31]. Level of T. gondii infection seroprevalence in humans is also related to rainfall in Guadeloupe [Reference Barbier, Ancelle and Martin-Bouyer32]. However, other environmental or cultural factors vary between the compared areas. More specifically, the risk related to contact with soil should be more variable in space and time since soil contamination is directly related to human risk, while the risk related to contaminated meat should be less variable since protozoan bradyzoites persist in the tissues of infected individuals. As a consequence, we expect high spatio-temporal variability in risk only in populations where contact with soil is a major route of infection. This hypothesis remains to be tested.
ACKNOWLEDGEMENTS
We thank Marc Artois, Dominique Pontier and all those who assisted in the field. We also thank Christiane Leprince for performing serological tests and Marie-Lazarine Poulle for helpful comments on the manuscript. This work was financially supported by the Agence Française de Sécurité Sanitaire de l'Environnement et du Travail, the Grünenthal France Laboratory, the Région Champagne-Ardenne and the Communauté de Communes de l'Argonne Ardennaise.
DECLARATION OF INTEREST
None.








