Crossref Citations
This article has been cited by the following publications. This list is generated based on data provided by
Crossref.
García-Romero, E.
Suárez Barrios, M.
and
Bustillo Revuelta, M. A.
2004.
Characteristics of a Mg-palygorskite in Miocene rocks, Madrid Basin (Spain).
Clays and Clay Minerals,
Vol. 52,
Issue. 4,
p.
484.
Kuang, Wenxing
Facey, Glenn A.
and
Detellier, Christian
2004.
Dehydration and rehydration of palygorskite and the influence of water on the nanopores.
Clays and Clay Minerals,
Vol. 52,
Issue. 5,
p.
635.
Suárez, M.
and
García-Romero, E.
2006.
FTIR spectroscopic study of palygorskite: Influence of the composition of the octahedral sheet.
Applied Clay Science,
Vol. 31,
Issue. 1-2,
p.
154.
Huang, Yan-Jun
Li, Zhen
Li, Shu-Zhen
Shi, Zi-Liang
Yin, Lin
and
Hsia, Yuan-Fu
2007.
Mössbauer investigations of palygorskite from Xuyi, China.
Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms,
Vol. 260,
Issue. 2,
p.
657.
Post, James L.
and
Crawford, Susan
2007.
Varied forms of palygorskite and sepiolite from different geologic systems.
Applied Clay Science,
Vol. 36,
Issue. 4,
p.
232.
García-Romero, E.
Suárez, M.
Santarén, J.
and
Alvarez, A.
2007.
Crystallochemical characterization of the palygorskite and sepiolite from the Allou Kagne deposit, Senegal.
Clays and Clay Minerals,
Vol. 55,
Issue. 6,
p.
606.
Li, Z.
He, K.
Yin, L.
Xiong, F.
and
Zheng, Y. C.
2007.
Crystallochemistry of Fe-rich palygorskite from eastern China.
Clay Minerals,
Vol. 42,
Issue. 4,
p.
453.
Cai, Yuanfeng
Xue, Jiyue
and
Polya, D.A.
2007.
A Fourier transform infrared spectroscopic study of Mg-rich, Mg-poor and acid leached palygorskites.
Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy,
Vol. 66,
Issue. 2,
p.
282.
Bayram, H.
Önal, M.
Üstünışık, G.
and
Sarıkaya, Y.
2007.
Some thermal characteristics of a mineral mixture of palygorskite, metahalloysite, magnesite and dolomite.
Journal of Thermal Analysis and Calorimetry,
Vol. 89,
Issue. 1,
p.
169.
Gionis, Vassilis
Kacandes, George H.
Kastritis, Ioannis D.
and
Chryssikos, Georgios D.
2007.
Combined near-infrared and X-ray diffraction investigation of the octahedral sheet composition of palygorskite.
Clays and Clay Minerals,
Vol. 55,
Issue. 6,
p.
543.
Tao, CHEN
Hejing, WANG
Xiaoping, ZHANG
and
Nan, ZHENG
2008.
SAED and HRTEM Investigation of Palygorskite.
Acta Geologica Sinica - English Edition,
Vol. 82,
Issue. 2,
p.
385.
Burzo, E.
2009.
Phyllosilicates.
Vol. 27I5b,
Issue. ,
p.
444.
Boudriche, L.
Hamdi, B.
Kessaïssia, Z.
Calvet, R.
Chamayou, A.
Dodds, J. A.
and
Balard, H.
2010.
An Assessment of the Surface Properties of Milled Attapulgite Using Inverse Gas Chromatography.
Clays and Clay Minerals,
Vol. 58,
Issue. 2,
p.
143.
Frost, Ray L.
Xi, Yunfei
and
He, Hongping
2010.
Synthesis, characterization of palygorskite supported zero-valent iron and its application for methylene blue adsorption.
Journal of Colloid and Interface Science,
Vol. 341,
Issue. 1,
p.
153.
Madejová, Jana
Balan, Etienne
and
Petit, Sabine
2010.
Advances in the characterization of industrial minerals.
p.
171.
Garciá-Romero, E.
and
Suárez, M.
2010.
On the Chemical Composition of Sepiolite and Palygorskite.
Clays and Clay Minerals,
Vol. 58,
Issue. 1,
p.
1.
Frankovská, J.
Andrejkovičová, S.
and
Janotka, I.
2010.
Effect of NaCl on hydraulic properties of bentonite and bentonite–palygorskite mixture.
Geosynthetics International,
Vol. 17,
Issue. 4,
p.
250.
Tlili, Ali
Felhi, Mongi
Fattah, Nabil
and
Montacer, Mabrouk
2011.
Mineralogical and geochemical studies of Ypresian marly clays and silica rocks of phosphatic series, Gafsa-Metlaoui basin, southwestern Tunisia: implication for depositional environment.
Geosciences Journal,
Vol. 15,
Issue. 1,
p.
53.
Eren, E.
and
Gumus, H.
2011.
Characterization of the structural properties and Pb(II) adsorption behavior of iron oxide coated sepiolite.
Desalination,
Vol. 273,
Issue. 2-3,
p.
276.
Suárez, Mercedes
and
García-Romero, Emilia
2011.
Developments in Palygorskite-Sepiolite Research.
Vol. 3,
Issue. ,
p.
33.
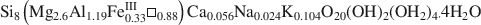 , was studied by FTIR spectroscopy. In both OH-stretching and OH-bending regions, there is evidence of dioctahedral Al2□OH, AlFe□OH and trioctahedral Mg3OH features, leading to a di-trioctahedral crystallochemical model of octahedral site occupancies in ribbons of Ganntour palygorskite.
, was studied by FTIR spectroscopy. In both OH-stretching and OH-bending regions, there is evidence of dioctahedral Al2□OH, AlFe□OH and trioctahedral Mg3OH features, leading to a di-trioctahedral crystallochemical model of octahedral site occupancies in ribbons of Ganntour palygorskite.