Introduction
The genus Echinochloa contains more than 250 species, and several are important troublesome weeds in agricultural systems (Gould et al. Reference Gould, Ali and Fairbrothers1972). Barnyardgrass [Echinochloa crus-galli (L.) P. Beauv.] is one of the most problematic weeds that is present in every continent where rice (Oryza sativa L.) is grown, leading to yield losses that can reach up to 80% (Bajwa et al. Reference Bajwa, Jabran, Shahid, Ali and Chauhan2015). At least three different varieties of E. crus-galli have been described, including var. crus-galli, var. formensis, and var. zelayensis, with the first being the most common in rice fields across the globe (Barrett and Wilson Reference Barrett and Wilson1981). As a prolific hexaploid and genetically diverse plant species, E. crus-galli tends to evolve resistance to weed control methods at a relatively rapid rate. The most classic example is a biotype of E. crus-galli that evolved a phenotype to mimic the appearance of rice to prevent visual identification after several generations of hand-weeding selection (Barrett Reference Barrett1983).
To date, 52 herbicide-resistance cases have been reported in E. crus-galli biotypes across 24 different countries (Heap Reference Heap2022). These cases involve single or multiple resistance to different herbicide modes of action (MOAs) such as 5-enolpyruvylshikimate-3-phosphate synthase, auxin mimics, acetyl-CoA carboxylase (ACCase), acetolactate synthase (ALS), microtubule assembly, PSII (photosystem II), and/or very-long-chain fatty-acid inhibitors. Propanil (PSII inhibitor) was the first herbicide to which E. crus-galli evolved resistance in rice fields (Baltazar and Smith Reference Baltazar and Smith1994). Consequently, rice growers shifted to quinclorac-based herbicide programs to manage resistance to PSII inhibitors, which led to the evolution of resistance to the auxinic herbicides and multiple resistance to both PSII inhibitors and auxin mimics in most populations (Talbert and Burgos Reference Talbert and Burgos2007). More recently, resistance to ALS-inhibiting herbicides (e.g., penoxsulam, bispyribac-sodium, and imazethapyr) evolved in the 2000s and after the commercialization of imidazolinone-resistant rice varieties (Bagavathiannan et al. Reference Bagavathiannan, Norsworthy, Smith and Neve2014).
Florpyrauxifen-benzyl (Loyant® herbicide with Rinskor™ active, Indianapolis, IN, USA) was introduced to the market in 2018 and was widely adopted for selective control of E. crus-galli in rice fields (Epp et al. Reference Epp, Alexander, Balko, Buysse, Brewster, Bryan, Daeuble, Fields, Gast, Green, Irvine, Lo, Lowe, Renga and Richburg2016). In the winter following the commercial launch of florpyrauxifen-benzyl, a greenhouse screen performed across multiple E. crus-galli populations from the midsouthern United States identified populations with resistance to florpyrauxifen-benzyl (Hwang et al. Reference Hwang, Norsworthy, González-Torralva, Priess, Barber and Butts2022b; Miller et al. Reference Miller, Norsworthy and Scott2018). A follow-up study associated the resistance mechanism to reduced foliar absorption and decreased activation of florpyrauxifen-benzyl into its acid form in resistant plants (Hwang et al. Reference Hwang, Norsworthy, González-Torralva, Piveta, Priess, Barber and Butts2022a, Reference Hwang, Norsworthy, González-Torralva, Priess, Barber and Butts2022b). The same study found no mutations in the auxin receptors TIR1 and AFB5, although most cases of target-site resistance to synthetic auxins are associated with the Aux/IAA co-receptor proteins (Figueiredo et al. Reference Figueiredo, Küpper, Malone, Petrovic, Figueiredo, Campagnola, Peersen, Prasad, Patterson, Reddy, Kubeš, Napier, Dayan, Preston and Gaines2022; Todd et al. Reference Todd, Figueiredo, Morran, Soni, Preston, Kubeš, Napier and Gaines2020).
Herbicide-resistance evolution in such a short period of time after commercialization indicates that resistant individuals were most likely already present at a high frequency in the field population, possibly due to a preexisting selection pressure by a different herbicide. For instance, rigid ryegrass (Lolium rigidum Gaudin) populations from Australia were cross-resistant to a newly introduced herbicide (pyroxasulfone) after preselection with older herbicides (prosulfocarb, triallate and trifluralin, S-metolachlor) (Busi and Powles Reference Busi and Powles2013, Reference Busi and Powles2016). Here, we hypothesized that florpyrauxifen-benzyl resistance in E. crus-galli evolved before the introduction of the herbicide to the market, likely due to the selection pressure imposed using other methoxy-containing herbicides such as penoxsulam and imazamox. We also discovered that a cytochrome P450 monooxygenase gene is likely associated with metabolic cross-resistance mechanism in E. crus-galli, challenging the preexisting understanding of florpyrauxifen-benzyl resistance.
Material and Methods
Seed Origin and Plant Growth
Two Echinochloa crus-galli biotypes were collected from commercial rice fields in Arkansas (AR-27) and Missouri (MO-18) where florpyrauxifen-benzyl had not been sprayed in prior years and poor efficacy was observed. Seeds were harvested from at least 10 random plants that were able to produce seeds during the rice-growing season. The collected seeds were planted in the greenhouse and sprayed with a field rate of florpyrauxifen-benzyl. Most plants survived (data not shown), and seeds from a single surviving plant were harvested for the subsequent studies. A third biotype was originally purchased from a commercial supplier (B&T World Seeds Route des Marchandes, Aigues-Vivesout, France) and increased to bulk up our seed inventory. Seeds from a single plant were used as a susceptible (S) standard for comparison purposes.
Plants were grown in a soil media composed of 90% by volume Pro-Mix® BX (Premier Tech Horticulture, Quakertown, PA, USA) and 10% Profile® Greens Grade™ (Profile Products, Buffalo Grove, IL, USA). Pro-Mix® BX contains approximately 83% sphagnum peat moss, 13% perlite, 20% vermiculite, and proprietary amounts of limestone, starter fertilizer, and wetting agent. Profile® Greens Grade™ is a sand-sized granule formed from calcining illite and montmorillonite clay. Several seeds of each biotype were planted in 10-cm2 pots and top watered twice daily. Plant material was propagated in a warm greenhouse with temperature ranging from 25 to 28 C and 50% to 60% relative humidity at the Corteva Global Headquarters (Indianapolis, IN, USA). Natural light was supplemented with 1,000-W metal-halide overhead lamps with an average illumination of 500 μE m−2 s−1 photosynthetic active radiation for 16 consecutive hours each day. Pots were thinned to 4 plants before spraying.
Spray Treatments and Tissue Sampling
Plants were grown as described earlier up to the 4-leaf stage. Treatments were applied with a research track sprayer (Generation III Research Sprayer manufactured by DeVries Manufacturing, Hollandale, MN, USA) calibrated to deliver a spray volume of 140 L ha−1. The track sprayer was fit with a TeeJet® 8003E nozzle (Spraying Systems, Wheaton, IL,USA), and used a spray pressure of 276 kPa and a speed of 3.1 km h−1. The nozzle height was 46 cm above the plant canopy. The experimental design used for the trial was a randomized complete block with four replications per treatment. After application, plants were placed on carts according to the randomization and returned to the greenhouse. All herbicides were obtained from Corteva Agriscience’s compound library. The herbicide treatments, doses, adjuvant system, and respective MOAs are described in Table 1. Each pot contained four plants and represented one biological replication. The entire shoot tissue (leaves + stem) from all plants was harvested at 1, 2, 3, and 7 d after treatment. The experiment was repeated.
Table 1. Treatments and their respective doses and herbicide modes of action.

a Methylated seed oil (MSO) at 1 L ha−1 was added to all treatments.
Metabolite Extraction and LC-MS/MS Analysis
Shoot samples were washed with 10 μl of 80:20 acetonitrile:water in 50-ml tubes for 1 min before extraction. After the washing step, the plant material was transferred into a new 20-ml homogenization tube containing two 5/16-inch stainless steel balls and 5 ml of extraction solution (30% water with 20 mM ammonium acetate pH 4.5, 15% 2-propanol, 25% methanol, and 30% acetonitrile). The tubes were vigorously shaken in a Geno/grinder® (SPEX SamplePrep LLC, Metuchen, NJ, USA) at 500 × g for 2 min or until all plant material turned into a finely ground homogenous solution. The homogenate was then centrifuged to pellet, and the supernatant transferred to 96-well plates to be analyzed by liquid chromatography-tandem mass spectrometry (LC-MS/MS).
Each sample had 10 µl injected into an UHPLC (Agilent Technologies, Santa Clara, CA, USA) attached to an AB Sciex X500R QToF system. The mobile phase consisted of 20 mM ammonium acetate pH 4.5 in de-ionized water (Solvent A) and 25% methyl tert-butyl ether + 75% methanol (Solvent B). The stationary phase was composed of an ACQUITY Premier CSH (Waters Corporation, Milford, MA, USA) Phenyl-Hexyl Column (1.7 µm, 2.1 by 150 mm). The total run time was 15 min at a flow rate of 0.25 ml min−1. The chromatography conditions were 2% at 0 to 2 min, 25% at 3.75 min, 100% at 11 to 12 min, 2% at 12.5 to 15 min, of Solvent B. QToF acquisition was in positive mode using information-dependent acquisition-mediated LC-MS/MS. Peak areas were quantified based on an external calibration curve when metabolite was available. Otherwise, peak areas were presented.
In Vitro ALS Enzyme Assays
Fresh tissue (2 g) from meristematic leaves was collected from each plant biotype (susceptible, MO-18, and AR-27) with 6 to 8 fully expanded leaves for in planta ALS activity assessment. Penoxsulam and imazamox were tested as 5-fold dilutions down from 1 mM to 0.1 nM. Samples were ground in liquid nitrogen and homogenized using a mortar and pestle with 9 ml of extraction buffer (4 mM thiamine pyrophosphate, 25 mM potassium phosphate, 5 mM magnesium chloride, 200 mM sodium pyruvate, and 20 mM flavin adenine dinucleotide) at pH 7.5. The crude extract was filtered through cheesecloth and miracloth into 50-ml tubes. Then, tubes were centrifuged at 16,000 × g for 15 min at 4 C. The crude extract was always maintained on ice. Reactions were prepared in deep-well plates by adding 62.5 μl of extraction buffer with herbicide and 62.5 μl of crude extracts and incubated at 37 C for 1 h. The reaction was stopped by adding 31.25 μl of 2 N sulfuric acid and incubated for 15 min at 60 C. After incubation, all samples received a fresh solution (217.5 μl) containing 1-naphthol (2.5%), creatine (0.25%), and ammonium hydroxide (2 N). Tubes were incubated for 15 min at 60 C and centrifuged at 4,000 × g for 15 min at room temperature. Two blanks were included for each extract, one with no herbicide (dose zero) and the other with no reaction (sulfuric acid added before the first incubation). An aliquot of 200 μl was transferred to microtiter plates and read at 535 nm absorbance. The blank samples were subtracted as background, and values were expressed in percentage relative to dose zero.
ALS Gene Sequencing
Approximately 100 mg of young leaf tissue was sampled from the three E. crus-galli biotypes: susceptible, MO-18, and AR-27. DNA was extracted using a DNeasy Plant Pro Kit (Qiagen, Germantown, MD, USA; CAS no. 69204). The full-length ALS gene was amplified by PCR using the forward primer: 5′-TTGCCACCCTCCCCAAACCC-3′ and reverse primer 5′-CGGATTTCGTGACCATTGC-3′. Fifty-microliter reactions were mixed following the standard protocol provided with SeqAmp DNA polymerase (Takara Bio, San Jose, CA, USA; CAS no. 638509) and 500 ng of template genomic DNA. A three-step PCR was run starting at 94 C for 1 min, followed by 30 cycles of 98 C for 10 s, 60 C for 15 s, and 68 C for 1 min. PCR products were run on a 0.7% agarose gel to verify single band amplification at 1,790 bp. The PCR products were sequenced using both the amplification primers listed above. Translated amino acid sequences obtained from these E. crus-galli plants were compared with a known susceptible E. crus-galli ALS amino acid sequence, GenBank protein accession LC006061.1.
Results and Discussion
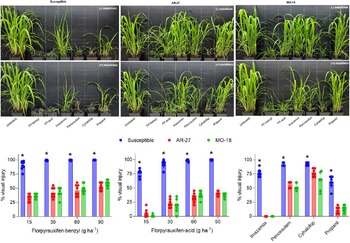
Overall, florpyrauxifen-benzyl provided higher visual control of all biotypes compared with florpyrauxifen-acid, but none of these compounds were able to control the resistant biotypes (Figure 1). In addition, while penoxsulam provided better control compared with imazamox, none of these ALS inhibitors were able to control the resistant biotypes. On the other hand, the susceptible E. crus-galli biotype was effectively controlled by florpyrauxifen-benzyl, florpyrauxifen-acid, penoxsulam, and cyhalofop. Florpyrauxifen-benzyl and florpyrauxifen-acid at the lowest rates (15 g ha−1) as well as imazamox and propanil, provided lower levels of control compared with the first group. Imazamox and propanil were tested at a lower than recommended rate, which may help to explain the lack of full control of the susceptible biotype, which does not rule out the potential presence of PSII resistance in the susceptible biotype. The fact that E. crus-galli control levels were substantially lower for resistant (AR-27 and MO-18) biotypes compared with the susceptible for all herbicides tested, indicates that these two biotypes are likely cross-resistant to at least four herbicide MOAs (auxin, ALS, ACCase, and PSII).

Figure 1. Visual injury (%) to susceptible (S) and resistant (AR-27, MO-18) Echinochloa crus-galli biotypes in response to florpyrauxifen-benzyl, florpyrauxifen-acid, penoxsulam, imazamox, cyhalofop, and propanil application. FP, florpyrauxifen. *Statistically significant difference between susceptible and resistant (AR-27, MO-18) biotypes (P < 0.05). Bar graphs represent average across two experimental runs with three replications each.
Malathion enhanced the control of E. crus-galli for all four compounds in both AR-27 and MO-18 biotypes (Figure 2). On average, malathion significantly increased E. crus-galli control levels from 20% to 50% for all herbicides, except cyhalofop and propanil. Malathion itself did not cause any phytotoxicity (data not shown). Both AR-27 and MO-18 biotypes responded similarly to the herbicides and the addition of malathion, suggesting that they may share a similar mechanism of resistance to these chemistries, possibly involving a cytochrome P450 monooxygenase enzyme. The fact that malathion treatment did not affect the response of AR-27 and MO-18 plants to cyhalofop and propanil, indicates that resistance to these two herbicides may result from a non–cytochrome P450 mechanism.

Figure 2. Effect of malathion on visual injury (%) provided by florpyrauxifen-benzyl, florpyrauxifen-acid, penoxsulam, or imazamox application to AR-27 and MO-18 Echinochloa crus-galli biotypes. *Statistically significant difference between no malathion and with malathion within each herbicide treatment (P < 0.05); ns, not significant (P > 0.05). Bar graphs represent average across two experimental runs with three replications each.
With regard to metabolite quantification, we focused on florpyrauxifen-benzyl, florpyrauxifen-acid, and penoxsulam, as malathion did not reverse resistance to propanil and cyhalofop. The levels of florpyrauxifen-acid were overall higher in the leaves of susceptible plants compared with the two resistant biotypes at all time points evaluated (Figure 3). In the absence of malathion, florpyrauxifen-acid levels tended to decrease over time in all biotypes, but susceptible plants showed a higher concentration compared with resistant ones at all time points. The addition of malathion caused these levels of florpyrauxifen-acid to remain higher for a longer period of time in all biotypes.

Figure 3. Levels of florpyrauxifen-acid in leaves of susceptible (S) and resistant (AR-27, MO-18) Echinochloa crus-galli (ECHCG) biotypes over time following florpyrauxifen-benzyl application without malathion. Data points represent the average and the respective standard error across two experimental runs with three replications each.
Resistant plants also showed a higher concentration of florpyrauxifen-hydroxyacid and its respective glucose conjugation metabolite compared with susceptible plants (Figure 4). At 24 h after treatment, these metabolites were up to 10 times higher in resistant plants. In addition, these metabolites were not present in malathion-treated plants, suggesting that the first reaction (demethylation) leading to the hydroxyacid metabolite is mediated by a cytochrome P450 monooxygenase.

Figure 4. Levels of florpyrauxifen-hydroxyacid and florpyrauxifen-hydroxy-glucose metabolites in leaves of susceptible (S) and resistant (AR-27, MO-18) Echinochloa crus-galli (ECHCG) biotypes following florpyrauxifen-benzyl application without malathion. These metabolites were not found in malathion-treated plants. Data points represent the average and the respective standard error across two experimental runs with three replications each.
A similar metabolism profile was observed for penoxsulam (Figure 5). Penoxsulam levels were up to three times higher in susceptible plants compared with those in resistant ones, even though these levels decreased over time in all biotypes. The main metabolite identified was a demethylated penoxsulam molecule that was 3- to 5-fold higher in resistant biotypes compared with the susceptible one. Glucose conjugation was also present in leaf extracts, and this metabolite was mostly found in resistant samples only.

Figure 5. Levels of pyroxsulam, pyroxsulam-hydroxyacid, and pyroxsulam-hydroxy-glucose metabolites in leaves of susceptible (S) and resistant (AR-27, MO-18) Echinochloa crus-galli (ECHCG) biotypes following florpyrauxifen-benzyl application without malathion. Data points represent the average and the respective standard error across two experimental runs with three replications each.
Finally, target-site resistance to penoxsulam and imazamox was assessed for the E. crus-galli biotypes (Figure 6). There were no differences among the three biotypes regarding ALS enzyme sensitivity to either herbicide, with IC50 (50% inhibitory concentration) values ranging around 0.001 and 1 μM for penoxsulam and imazamox, respectively. Therefore, these biotypes do not display target site–resistance mechanisms to penoxsulam and imazamox, and reduced sensitivity to these herbicides in AR-27 and MO-18 is due to cytochrome P450–mediated enhanced metabolism.

Figure 6. Acetolactate synthase (ALS) inhibition by penoxsulam and imazamox in susceptible (S) and resistant (AR-27, MO-18) Echinochloa crus-galli (ECHCG) biotypes. Data points represent the average and the respective standard error across two experimental runs with three replications each.
For a long time, herbicide-resistance management was thought to be mitigated by adopting herbicide mixtures and herbicide rotation with different MOAs (Gressel and Segel Reference Gressel, Segel, Green, LeBaron and Moberg1990). That concept can be applicable when managing target-site resistance to ALS inhibitors, for instance, with mixtures often providing a more favorable outcome (Beckie and Reboud Reference Beckie and Reboud2009). More recently, the rise of non–target site resistance has challenged this paradigm, because metabolic resistance can confer resistance to different MOAs that share similar chemical structures (Comont et al. Reference Comont, Lowe, Hull, Crook, Hicks, Onkokesung, Beffa, Childs, Edwards, Freckleton and Neve2020). Therefore, rotating or mixing herbicides of different MOAs can be ineffective if the two herbicides are metabolized by the same enzyme, which makes metabolic resistance a significant threat to sustainable weed management, mostly because cross-resistance issues are less predictable compared with target-site resistance (Yu and Powles Reference Yu and Powles2014).
Here, we discovered that florpyrauxifen-resistant E. crus-galli biotypes were cross-resistant to ALS-inhibiting herbicides (imazamox and penoxsulam). The addition of malathion enhanced the activity of all herbicides on resistant biotypes, suggesting that reduced sensitivity is mediated by a cytochrome P450 monooxygenase. Malathion is a well-known inhibitor of cytochrome P450 monooxygenase enzymes in plants and has been used in a number of metabolic herbicide-resistance studies involving these genes (Barrett Reference Barrett1995). Further metabolic analysis confirmed that resistant plants have enhanced ability to demethylate aryl-methoxy groups in these herbicides followed by glucose conjugation. We also show that these reactions are inhibited by the addition of malathion and that these biotypes do not contain a resistant version of the ALS gene.
Imazamox and penoxsulam are ALS-inhibiting herbicides that have been widely used in rice fields for E. crus-galli control for several years. The first is mostly adopted in Clearfield® (imidazolinone-resistant) cultivars (Bond and Walker Reference Bond and Walker2011), while the second is used for selective control of broadleaf and grass weeds in rice (Bond et al. Reference Bond, Walker, Webster, Buehring and Harrell2007). ALS resistance can be conferred by either target-site or non–target site resistance mechanisms (Fang et al. Reference Fang, Zhang, Liu, Yan, Li and Dong2019; Riar et al. Reference Riar, Norsworthy, Srivastava, Nandula, Bond and Scott2013). Florpyrauxifen-benzyl is a synthetic auxin herbicide that was first introduced to the rice herbicide market in 2018 (Epp et al. Reference Epp, Alexander, Balko, Buysse, Brewster, Bryan, Daeuble, Fields, Gast, Green, Irvine, Lo, Lowe, Renga and Richburg2016). Less than a year after its introduction, E. crus-galli resistance to this herbicide became a concern in the field. All these facts combined with the cross-resistance mechanism described in this research suggest that resistance may have evolved before the commercialization of florpyrauxifen-benzyl. Likewise, S-metolachlor-resistant L. rigidum in Australia was found to be cross-resistant to pyroxasulfone, a new herbicide from a different MOA that had never been sprayed on these plants (Busi and Powles Reference Busi and Powles2016).
In conclusion, we identified the florpyrauxifen-benzyl resistance mechanism in two E. crus-galli biotypes that were cross-resistant to imazamox and penoxsulam. Metabolic resistance was mediated (first step) by a cytochrome P450 monooxygenase enzyme that was inhibited in the presence of malathion. The second reaction involved a glucose conjugation to the hydroxylated metabolite in resistant plants. These findings lead one to speculate whether resistance evolved before the commercialization of florpyrauxifen-benzyl through repeated applications of imazamox and penoxsulam.
Acknowledgments
This research was funded by Corteva Agriscience, and no conflict of interest has been declared.