Infective endocarditis is a life-threatening bacterial infection of the heart that affects approximately 0.3–3.3 per 100,000 children, or 5–12 per 100,000 paediatric admissions, each year. Reference Pasquali, He, Mohamad, McCrindle, Newburger and Li1–Reference Gupta, Sakhuja, McGrath and Asmar3 Risk factors in children include congenital heart disease (CHD), previous cardiac surgery, Reference Rushani, Kaufman, Ionescu-Ittu, Mackie, Pilote and Therrien4–Reference Baddour, Wilson, Bayer, Fowler, Tleyjeh and Rybak6 the presence of central venous catheters, Reference Stockheim, Chadwick, Kessler, Amer, Abdel-Haq and Dajani7 and rheumatic heart disease. Mortality in paediatric infective endocarditis ranges from 5 to 10%. Children at highest risk for infective endocarditis-associated mortality are those with cyanotic CHD, infective endocarditis caused by Staphylococcus aureus, and children who develop cardiac or extracardiac infective endocarditis complications, such as heart failure, perivalvular abscess, or septic emboli. Reference Pasquali, He, Mohamad, McCrindle, Newburger and Li1,Reference Gupta, Sakhuja, McGrath and Asmar3,Reference Day, Gauvreau, Shulman and Newburger8,Reference Ware, Tani, Weng, Wilkes and Menon9
Neurologic complications of infective endocarditis, such as arterial ischaemic stroke, infectious intracranial aneurysms, intracranial haemorrhage, and intracerebral abscesses, occur in over half of adults with infective endocarditis, Reference Snygg-Martin, Gustafsson, Rosengren, Alsio, Ackerholm and Andersson10,Reference Heiro, Nikoskelainen, Engblom, Kotilainen, Marttila and Kotilainen11 likely as a direct result of septic material from cardiac vegetations embolising to the intracranial vasculature. Risk factors for neurologic complications of infective endocarditis in adults include vegetations larger than 30 mm, left-sided vegetations, particularly those affecting the anterior leaflet of the mitral valve, anticoagulation, and infection due to Staphylococcus aureus. Reference Stockheim, Chadwick, Kessler, Amer, Abdel-Haq and Dajani7 The frequency of and risk factors for neurologic complications of infective endocarditis have not been well described in children and may differ from those in adults. One recent study of 31 children under 14 years of age identified neurologic complications in 23%, with possible risk factors including lower body weight, left-sided valvular lesions, and higher C-reactive protein. However, this group did not comment on differences between children with and without CHD. Reference AlBassri, Sheikho, Chaikhouni, Al Habshan and Kabbani12 Abnormal cardiac anatomy that allows for right-to-left shunting and surgically created aberrant connections between systemic and venous vasculature (e.g., single ventricle, systemic-pulmonary artery shunts, ductus arteriosus stents) may lead to unique structural risk factors for intracranial embolisation in children with CHD and infective endocarditis. Reference Bayer, Bolger, Taubert, Wilson, Steckelberg and Karchmer13 Moreover, the presence of implanted foreign materials as part of CHD repair may alter the typical microbiology associated with paediatric infective endocarditis. Reference Gupta, Sakhuja, McGrath and Asmar3 A better understanding of risk factors for neurologic complications of infective endocarditis in children with structurally normal hearts and in children with CHD is critical to guide diagnostic surveillance and management strategies in this vulnerable population.
We therefore aimed to 1) define the frequency and characteristics of acute neurologic complications in a cohort of children with infective endocarditis hospitalised at a tertiary care facility and 2) identify risk factors for the development of acute neurologic complications.
Patients and methods
Study design and data collection
We performed a retrospective review of the electronic medical record at Children’s Hospital of Philadelphia to identify patients with infective endocarditis. The Institutional Review Board approved this study. Informed consent and Health Insurance Portability and Accountability Act (HIPAA) authorisation were waived. We queried the electronic medical record for all children aged 0–18 years hospitalised at Children’s Hospital of Philadelphia between 1 January, 2008 and 31 December, 2017 with an International Classification of Disease, Ninth or Tenth Revision (ICD-9 or -10) discharge diagnosis code for infective endocarditis (A01.02, A18.84, A32.82, A39.51, A52.03, A54.83, B33.21, B37.6, B39, B49, I01.1, I08, I09.1, I26.01, I26.90, I33, I35.8, I38, I39, I76, M05.03, M32.11, Q24, and Z86.79). To ensure complete ascertainment of all eligible cases, we cross-referenced this list with institutional contributions to national databases (Society for Thoracic Surgeons (STS), Pediatric Cardiac Critical Care Consortium (PC4), and IMproving Pediatric and Adult Congenital Treatment (IMPACT) databases). We then manually reviewed data from each child’s medical record to confirm the diagnosis of infective endocarditis by Duke criteria (see Study Definitions, Table 1) and abstracted demographic and clinical data for confirmed cases.
Table 1. Modified Duke criteria for infective endocarditisReference Durack, Lukes and Bright14

a Requires typical pathogens from ≥2 separate cultures.
b Heart condition or intravenous drug use.
c Major arterial emboli, septic pulmonary infarcts, infectious intracranial aneurysm, intracranial haemorrhage, conjunctival haemorrhages, or Janeway lesions.
d Glomerulonephritis, Osler nodes, Roth spots, or rheumatoid factor.
e Positive blood cultures that do not meet Duke major criteria, or serologic evidence of active infection with an organism consistent with infective endocarditis.
Study definitions
Infective endocarditis was defined as “definite” or “possible” using the revised Duke criteria. The Duke criteria use clinical, imaging, and laboratory evidence of intracardiac infection to define endocarditis. Reference Baltimore, Gewitz, Baddour, Beerman, Jackson and Lockhart15 While originally established for the diagnosis of adult infective endocarditis, these criteria have subsequently been modified for application to the paediatric population (Table 1). Reference Durack, Lukes and Bright14,Reference Li, Sexton, Mick, Nettles, Fowler and Ryan16,Reference Bendig, Singh, Butler and Arrieta17 Vegetations with the potential for embolisation to systemic (rather than pulmonary) circulation included those present in the left side of the heart in any child, and those present on the right side of the heart in a child with CHD and the anatomic potential for a right-to-left shunt. Presence of foreign material included prosthetic valves, central venous catheters, conduits, or recent surgery or catheterisation in the 30 days prior to endocarditis symptom onset. We defined endocarditis symptom onset as the first days of symptoms attributed to infective endocarditis.
Neurologic complications of endocarditis were defined as neurologic signs or symptoms that were directly attributed to infective endocarditis or to a related intervention (e.g., cardiac surgery for infective endocarditis), either during acute hospitalisation or follow-up at our institution (e.g., a related subsequent valve repair). Arterial ischaemic stroke, intracranial haemorrhage, cerebral sinus venous thrombosis, intracranial cerebral aneurysm, intracranial abscess, and other cerebral structural abnormalities were defined based on head computed tomography (CT) and/or brain magnetic resonance imaging (MRI). Endocarditis-associated seizures were defined as any clinical or electrographic seizure occurring in a child without a known seizure history, or increased seizures from baseline frequency in a child with known seizures. Encephalopathy was defined as documented parental or clinician concern for altered mental status, confusion, or irritability out of proportion to medical illness. Meningitis and encephalitis were defined as the presence of pleocytosis (cerebrospinal fluid white blood count >7 cells/high powered field), or MRI findings suggestive of brain parenchymal inflammation (T2 signal abnormalities) or leptomeningeal inflammation (post-contrast T1-weighted MRI enhancement), in the absence of alternative explanations. Cranial nerve palsies were defined based on clinical examination findings. Headache, the aetiology of which can be multi-factorial, was not considered a neurological complication and was not used to define the time of onset of neurologic symptoms unless it was temporally associated with a focal neurologic symptom or abnormal neuroimaging findings.
We classified neurological deficits at discharge based on the neurological examination documented in the discharge summary with additional input from the last neurology progress note, when available. We defined mild deficits as abnormalities on neurological examination that did not interfere with function, moderate deficits as neurological abnormalities that interfered with function, and severe deficits as abnormalities on neurological examination that resulted in significant loss of language, motor, or cognitive function.
Data analysis and statistical methods
Data were analysed using STATA version 14.2 (StataCorp, College Station, TX, USA). In order to minimise the effect of outliers on statistical associations in this small sample size, we described continuous variables using medians and interquartile ranges (IQRs) and compared inter-group differences using Wilcoxon rank-sum tests. We described categorical variables using counts and frequencies and compared inter-group differences using the χ2 test or Fisher’s exact test when n < 5 in any cell of a contingency table. The Kaplan–Meier estimate of survival was calculated to determine the neurologic complication-free survival of the cohort. Survival time was calculated from the date of endocarditis symptom onset or endocarditis diagnosis in those without clinical endocarditis symptoms. For those with neurologic complications, censor time was the date of neurologic complication onset or the date of imaging on which the neurologic complication was identified in those with asymptomatic neurologic complications. For those without neurologic complications, censor time was the date of hospital discharge or the date of the last examination by a neurologist, whichever was later. Statistical significance was considered a two-tailed p-value <0.05.
Results
One hundred forty-one potentially eligible children were identified by electronic medical record query, 68 of whom (48%) met Duke criteria for infective endocarditis on manual chart review. Forty-three met criteria for definite infective endocarditis and 25 for possible infective endocarditis (Table 1 and Fig 1). The remaining 73 children had reference to infective endocarditis or infective endocarditis prophylaxis in their electronic medical record but did not meet criteria for definite or possible infective endocarditis. Demographic and clinical characteristics of the cohort and the subgroups of children with and without neurological complications are summarised in Table 2. The median age at infective endocarditis symptom onset was 9.0 years (IQR 0.3, 14.6 years); about one-third of children (33%, 22/66) were less than 1-year-old at infective endocarditis symptom onset. Forty-one children (60%) with infective endocarditis had CHD (Supplementary Tables 1 and 2), nine of whom had a prosthetic valve (mechanical, n = 3 [aortic, n = 2, mitral, n = 1]; homograft/bioprosthetic, n = 6 [mitral, n = 1, pulmonary, n = 5]). There were no cases of rheumatic heart disease. Thirty-six children (53%) with infective endocarditis had foreign material present. Three children (two with CHD) had a recent dental procedure. Vegetations were present in 42 children, and 28 children had a vegetation with potential for systemic embolisation. Vegetations were located on the aortic valve in 8, mitral valve in 14, pulmonary valve in 4, tricuspid valve in 8, ventricle or ventricular septum in 5, atrium or atrial septum in 6, common atrioventricular valve in 1, and in other locations in 7 including 2 in right ventricle-pulmonary artery conduits, 1 in the pulmonary artery, 1 in a Sano conduit, 1 in a right pulmonary artery near a Blalock–Taussig shunt, 1 in a right ventricular outflow track, and 1 unspecified. Six children had vegetations in multiple locations. Figure 2 shows an example of a vegetation.

Figure 1. Summary of infectious endocarditis (IE) cohort.
Table 2. Demographic and clinical features of children with infective endocarditis, by neurologic complication
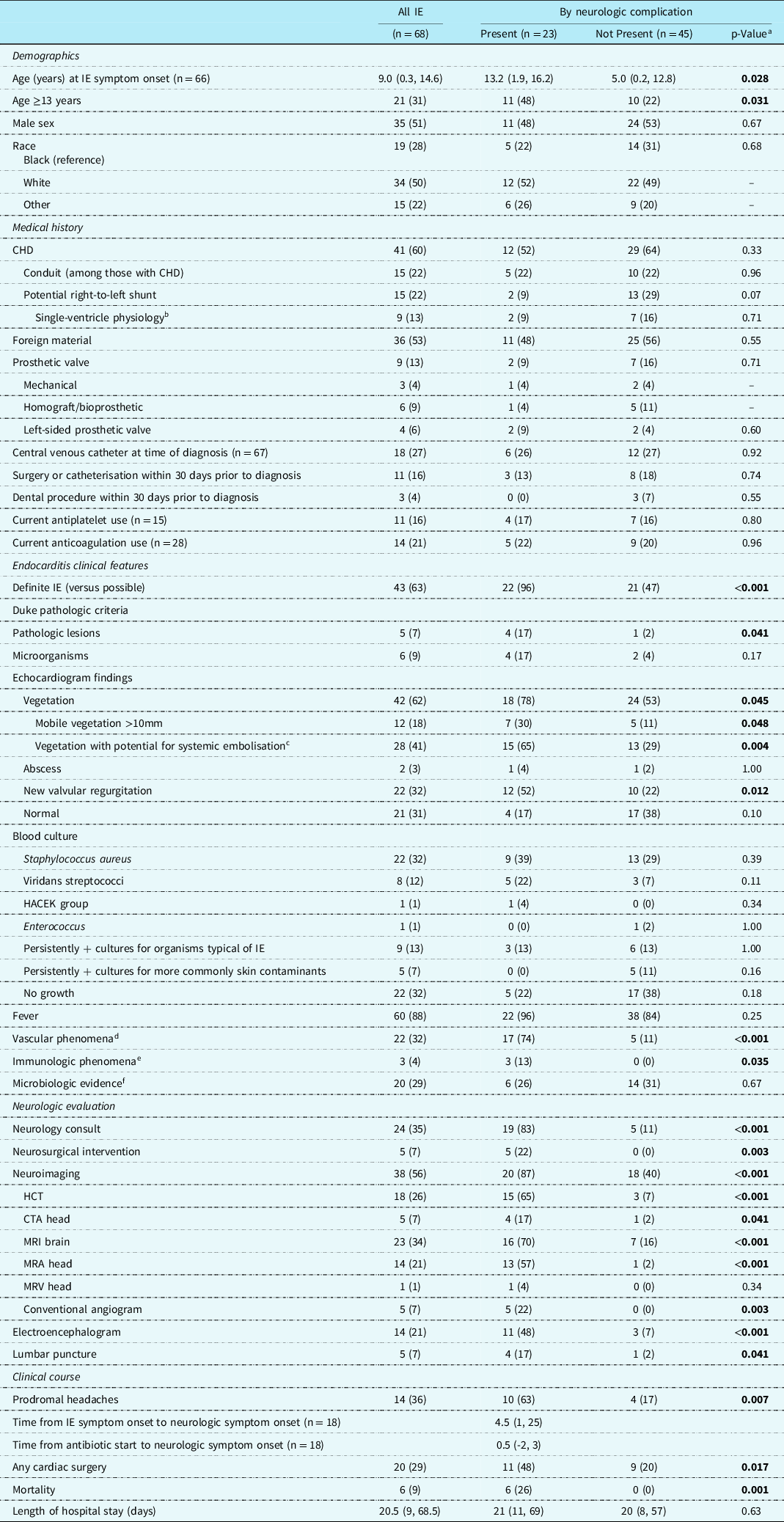
Bold p-values indicate statistical significance.
Categorical variables are described using n (%). Continuous variables are described using median (IQR).
CTA, computed topography angiogram; HACEK, Haemophilis species, Actinocacillus actinomycetemcomitans, Cardiobacterium hominis, Eikenella species, and Kingella kingae; HCT, head computed topography; IE, infective endocarditis; MRA, magnetic resonance angiography; MRV, magnetic resonance venography.
a p-Values were calculated using chi-squared tests or Fisher’s exact test for categorical variables and Wilcoxon rank-sum tests for continuous variables.
b All children with fenestrated Fontan or stage I palliation. There were no children with stage II palliation.
c All children with a left-sided vegetation or those with a right-sided vegetation with potential for a right-to-left shunt and systemic embolisation.
d Vascular phenomena include major arterial emboli, septic pulmonary infarcts, infectious intracranial aneurysm, intracranial haemorrhage, conjunctival haemorrhages, or Janeway lesions.
e Immunologic phenomena include glomerulonephritis, Osler nodes, Roth spots, or rheumatoid factor.
f Microbiologic evidence includes positive blood cultures that do not meet Duke major criteria, or serologic evidence of active infection with organism consistent with IE.
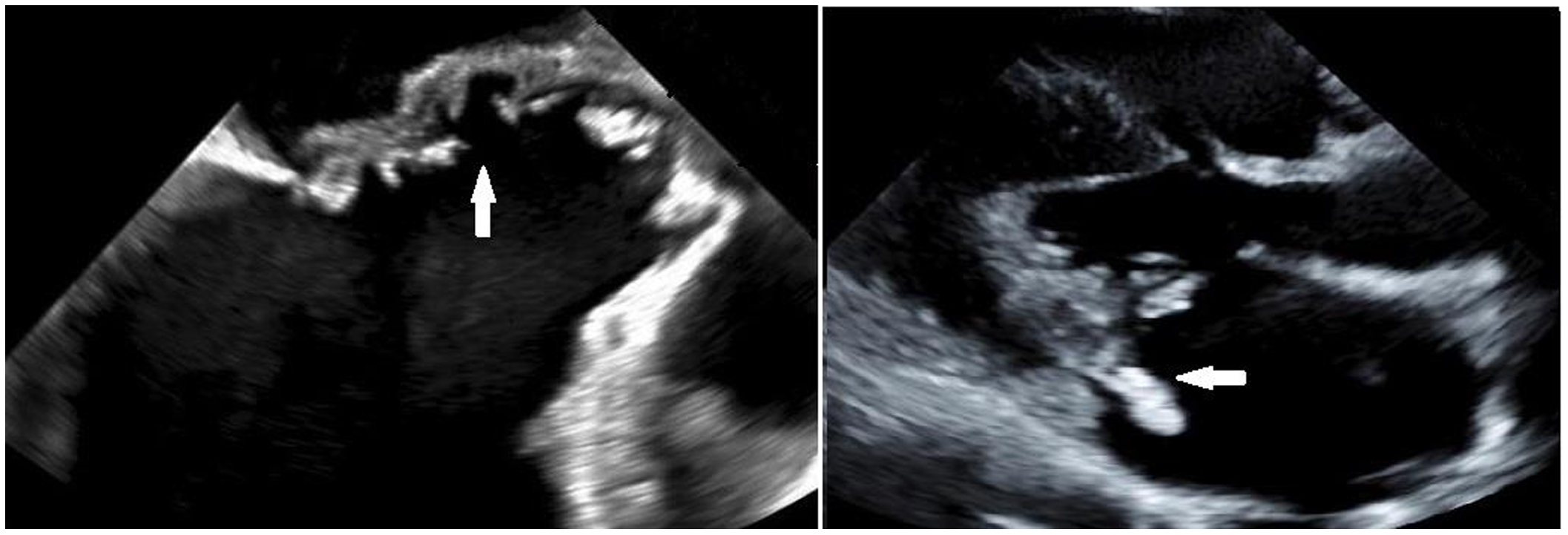
Figure 2. Trans-esophageal echocardiogram demonstrates erosion of the mitral to aortic fibrous continuity in a child with infective endocarditis (left). A vegetation on the posterior leaflet of the mitral valve seen on trans-thoracic echocardiogram in a different child with IE (right).
Neurologic complications
Neurologic complications of infective endocarditis were common, identified in 23 of 68 children (34%, 95% CI 23–46%) (Tables 2 and 3). Neurologic complication-free survival was 70% at 30 days (95% CI 57-80%), 63% at 90 days (95% CI 49–75%), and 58% at 1 year (95% CI 42–72%) (Fig 3). The proportion of children with neurologic complications did not differ between those with and without a diagnosis of CHD (12/41 [29%] versus 11/27 [41%], p = 0.33). Neither children with CHD and a right-to-left shunt (compared to those with CHD without right-to-left shunt) nor children with a systemic prosthetic valve (compared to children without systemic prosthetic valve) had higher proportions of neurologic complications. Two children had bovine jugular vein prosthetic materials (both with Melody valves). Neither of these children had residual shunts or neurologic complications.
Table 3. Frequency of neurologic complications in children who met modified Duke criteria for infective endocarditis
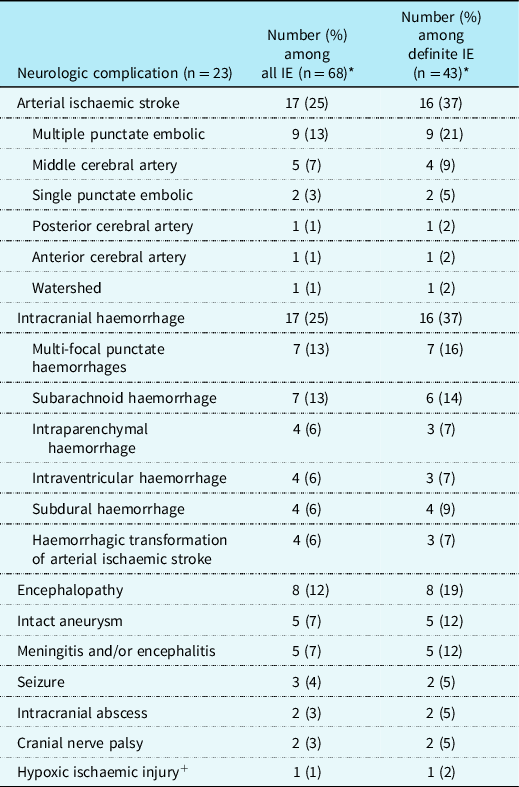
* Some children had more than one neurologic complication.
+ Occurred after conduit replacement.
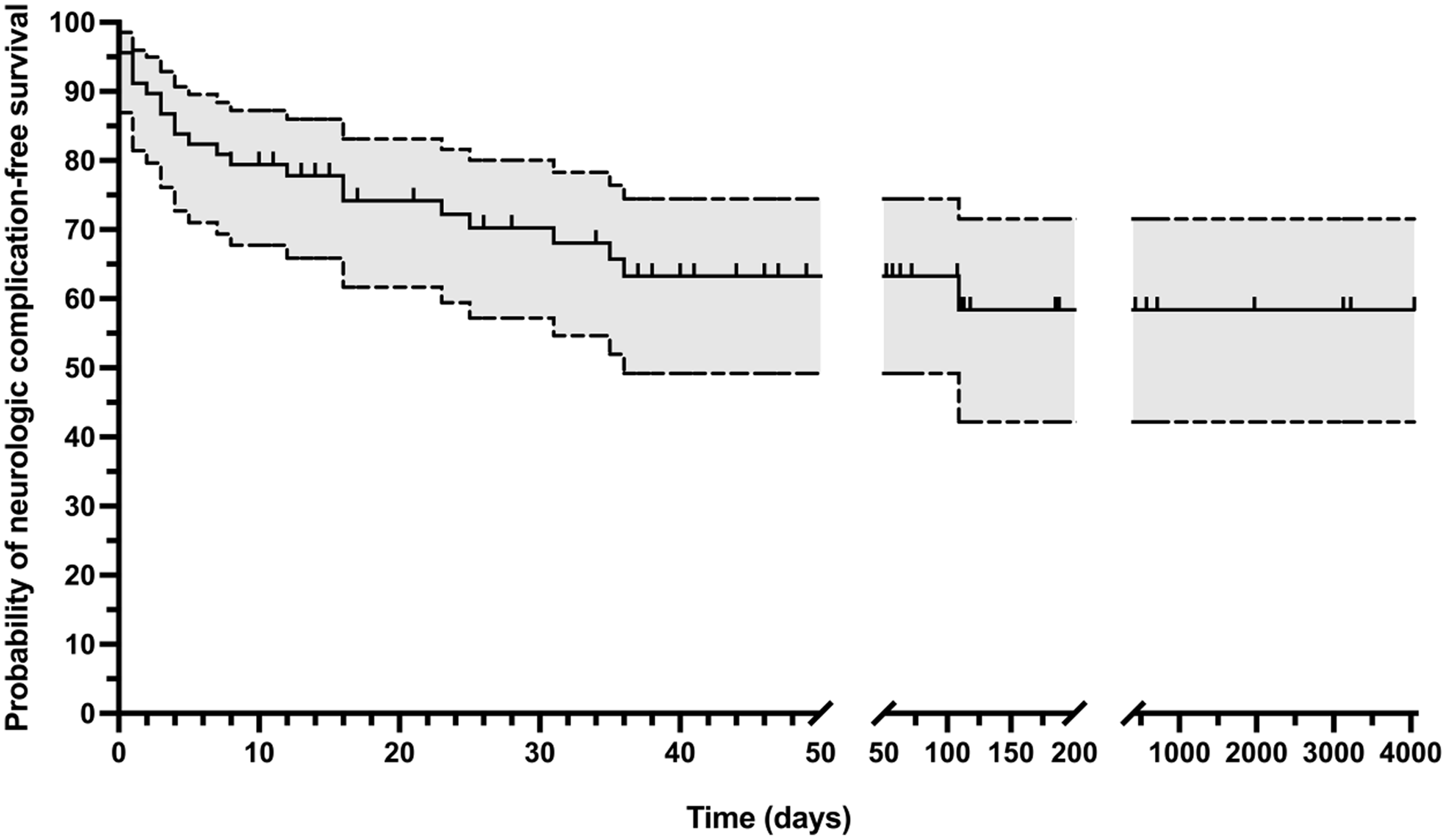
Figure 3. Kaplan–Meier survival curve with 95% confidence interval showing neurologic complication-free survival. Vertical tick marks indicate censor time for children without identified neurologic complications.
Almost all children with identified neurologic complications (22/23) had these occur during the acute hospitalisation. Eighteen of the 23 children with a neurologic complication (78%, 95% CI 56–93%) had clinical neurologic symptoms; five had asymptomatic neurologic diagnoses identified on screening neuroimaging only. Symptoms began a median of 4.5 days (IQR 1, 25; n = 18) after the onset of endocarditis symptoms and a median of 0.5 days (IQR -2, 3; n = 18) after antibiotic initiation. Ten children had neurologic symptoms on or before the day of admission (encephalopathy [n = 7], focal weakness [n = 2], headache due to intracranial haemorrhage [n = 2]). Two children who had neurologic complications during the acute hospitalisation (one arterial ischaemic stroke and one transient ischaemic attack) had additional neurologic complications after discharge. One child who did not have a neurologic complication during the hospitalisation had a neurologic complication after discharge.
Table 3 summarises identified neurologic complications, including findings on neuroimaging, which was performed in only 38/68 (56%) of the cohort during the acute hospitalisation. More children with definite infective endocarditis underwent neuroimaging than those with possible infective endocarditis (72 versus 28%, p < 0.001), but neuroimaging was performed similarly with respect to sex (51% male versus 62% female, p = 0.45), age group (57 versus 56% of children ≥13 years old versus <13 years old at infective endocarditis symptom onset, p = 0.94), and those with and without CHD (51 versus 63%, p = 0.34). Figures 4, 5, and 6 demonstrate representative neuroimaging findings. Ischaemic injury and haemorrhage were the most common neurologic complications, occurring in 17 children each. Multi-focal punctate embolic infarcts (n = 9) and middle cerebral artery arterial ischaemic strokes (n = 5) were the most common ischaemic injuries, and multi-focal punctate haemorrhage (n = 7) and subarachnoid haemorrhage (n = 7) were the most common types of haemorrhages. Six children had multiple different types of haemorrhage. A similar proportion of children with haemorrhage, compared to those without haemorrhage, were being treated with anticoagulation or antiplatelet therapy prior to infective endocarditis symptom onset (63 versus 76%, p = 0.31). Eleven children had both haemorrhage and ischaemic stroke, four of whom had haemorrhagic transformation of an ischaemic injury. Intact intracranial aneurysms were visualised in five children, four of whom also had intracranial haemorrhage, two of which were subarachnoid haemorrhages. Other aneurysms may not have been detected if bleeding had already occurred at the time of vascular imaging with obliteration or compression of the aneurysm. One aneurysm was clipped, one was coiled twice, and three were managed medically. There were three children with seizures, all of whom also had intracranial haemorrhage and/or ischaemic injury, suggesting these were acute symptomatic. Eight children had encephalopathy, seven of whom also had intracranial pathology including ischaemic injury, intracranial haemorrhage, or meningitis, suggesting encephalopathy was symptomatic of underlying structural abnormalities or intracranial infection. The eighth child with encephalopathy had progressive deterioration of mental status to obtundation. This child had multi-system organ dysfunction and died before neuroimaging could be obtained.
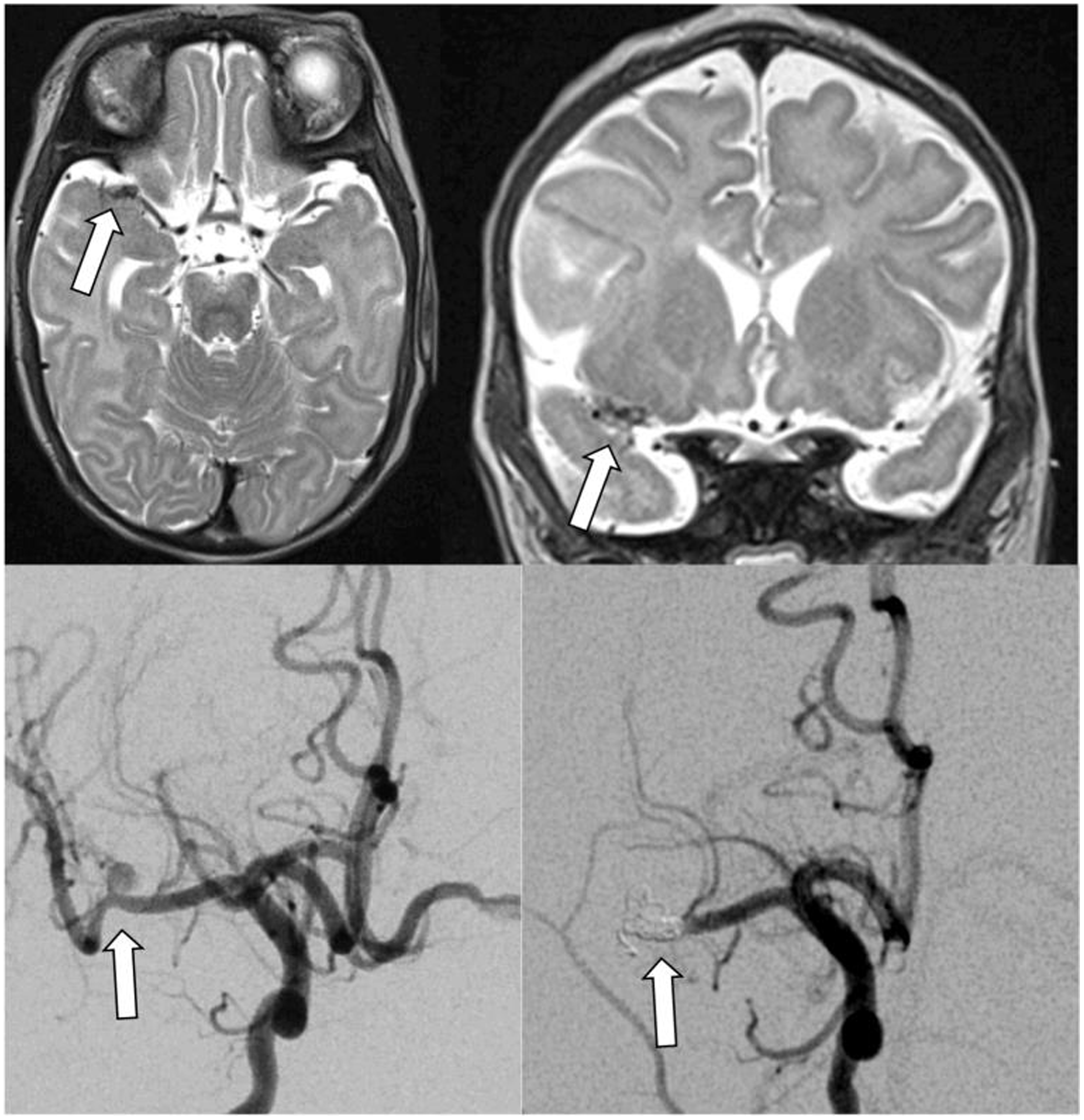
Figure 4. Infectious intracranial aneurysm in an infant with hypoplastic left heart syndrome and infective endocarditis with left middle cerebral artery stroke. Top panels show T2 axial (left) and coronal (right) MRIs with right middle cerebral artery infectious intracranial aneurysm. Bottom panels demonstrate the infectious intracranial aneurysm on conventional angiogram before (left) and after (right) coiling.
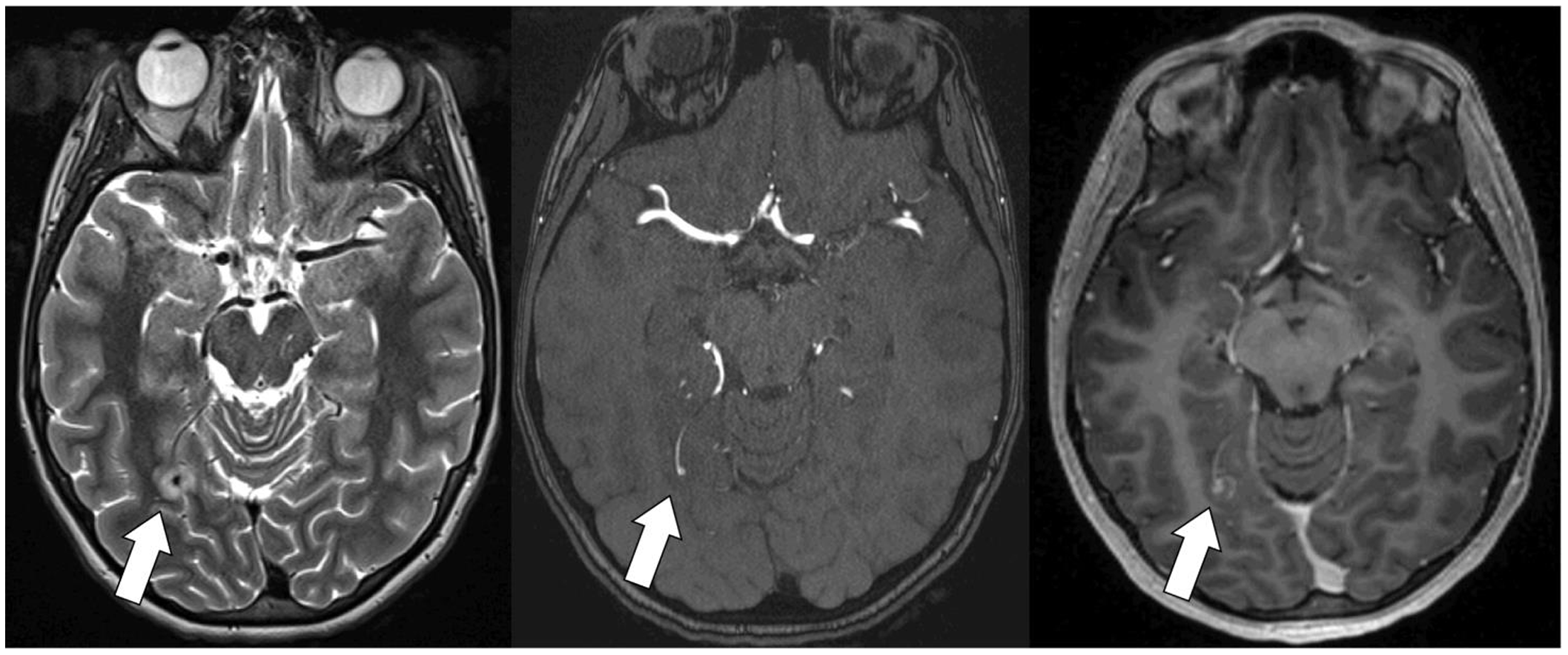
Figure 5. Previously healthy adolescent with a structurally normal heart with infective endocarditis and a small infectious intracranial aneurysm. T2 MRI (left) with oedema surrounding the infectious intracranial aneurysm. Time of flight magnetic resonance angiography (middle) demonstrates a small infectious intracranial aneurysm. T1 multi-planar reformation post-gadolinium image (right) demonstrates contrast enhancement.

Figure 6. Adolescent with DiGeorge syndrome and repaired truncus arteriosus with St. Jude’s valve in the mitral position and infective endocarditis. The head CT demonstrated multiple septic emboli with haemorrhagic transformation (small right frontal lesion demonstrated on this slice) (left panel). Head CT 48 hours later with massive haemorrhage before (middle panel) and after (right panel) craniectomy. CTA was not performed, but the large haemorrhage was presumed to be due to a ruptured infectious intracranial aneurysm.
Among the seven children who had subarachnoid haemorrhages, two had an intact aneurysm visualised, one had mild fusiform prominence of distal intracranial vessels proximal to the haemorrhage but without a clear aneurysm, and one did not have vascular imaging performed (though aneurysm was suspected). While one of these seven children was asymptomatic, among the remaining six, symptoms began a median of 18.5 days (IQR 2, 31) after the onset of endocarditis symptoms.
Fourteen of the 68 (21%) children had a headache. Among those with headache, 10 (71%) had a neurologic complication, including encephalopathy (n = 4), meningitis (n = 2), seizure (n = 1), ischaemic injury (n = 8), aneurysm (n = 4), abscess (n = 1), and intracranial haemorrhage (n = 9). Among the four children with headache who did not have an identified neurologic complication, only two underwent neuroimaging (head CT [n=1] and MRI brain [n=1]), and these studies were normal.
Five children had asymptomatic neurologic complications identified on screening neuroimaging only (abscess [n = 1]; aneurysm, leptomeningeal enhancement, and subarachnoid haemorrhage [n = 1]; subdural haemorrhage [n = 1]; punctate ischaemia and/or haemorrhage [n = 2]). The indications for screening neuroimaging included evaluation of fever or headache of unknown origin before the diagnosis of infective endocarditis, presence of systemic embolic infarcts, concern for systemic Candida infection, and screening imaging performed following bypass. One child with an asymptomatic intracranial abscess underwent neurosurgical drainage. None of the other children required intervention for their asymptomatic neurologic complications. Only one child with asymptomatic neurologic disease (subarachnoid hemorrhage and leptomeningeal enhancement) had an additional or progressive neurologic symptoms identified later (aneurysm identified).
On univariable analysis (Table 2), we found that children with an identified neurologic complication were older than those without (median age 13.2 [IQR 1.9, 16.2] years versus 5.0 [IQR 0.2, 12.8] years, p = 0.028), more often had a definite (rather than possible) diagnosis of infective endocarditis (96 versus 47%, p < 0.001), pathologic lesions (17 versus 2%, p = 0.041), large (>10mm) mobile vegetations (30 versus 11%, p = 0.048), vegetations with potential for systemic embolisation (65 versus 29%, p = 0.004), new valve regurgitation (52 versus 22%, p = 0.012), vascular phenomena by Duke criteria (74 versus 11%, p < 0.001), or a headache (63 versus 17%, p = 0.007). In contrast to published literature, neither Staphylococcus aureus nor use of anticoagulation was a risk factor for neurologic complications in this paediatric cohort.
Hospital course
Fifteen of the 23 children with an identified neurologic complication had additional or progressive neurologic complications during their acute hospitalisations including haemorrhage (n = 10), ischaemic injury (n = 9), aneurysm (n = 3), septic emboli (n = 2), transient ischaemic attack (n = 2), leptomeningeal enhancement (n = 2), and abscess (n = 1), which occurred at a median of 18 days (IQR 10, 47). Four children with symptomatic neurologic complications required neurosurgical intervention (three extraventricular drains, one washout, two haemorrhage evacuations, and two aneurysm interventions). Of the 23 children with neurologic complications, six (26%) died; there were no deaths in children without neurologic complications. Causes of death included progressive intracranial haemorrhage (n = 2), septic shock (n = 1), cardiac arrest in the context of multi-focal infarcts and subdural haemorrhage with mass effect (n = 1), cardiac arrest with diffuse systemic clotting (n = 1), and withdrawal of care in an infant due to severity of medical illness (leptomeningeal enhancement, multi-focal punctate haemorrhages) and pre-maturity (n = 1). Among survivors, there was no significant difference in total length of hospital stay between those with and without neurologic complications. Among the 17 surviving children who had a neurologic complication, 8 (47%) had residual neurological deficits. Deficits were mild in five, moderate in one, and severe in two.
Fourteen children underwent cardiac surgery during their acute hospitalisation, a median of 15.5 days (IQR 6, 33 days) after admission. Six additional children underwent cardiac surgery after discharge from their acute hospitalisation at a median of 57 days (IQR 36, 79 days) after admission. As expected, children with an identified neurologic complication had cardiac surgery more often than children without a neurologic complication (11/23 [48%] versus 9/45 [20%], p = 0.017), likely because embolic phenomena are a surgical indication. Neurological symptoms or neuroimaging abnormalities in those with asymptomatic neurologic diagnoses preceded cardiac surgery in 10 of 11 who underwent surgery (median time between neurologic symptom onset to cardiac operation 15 days, IQR 1, 34); one additional child had first neurologic symptom onset immediately post-operatively (arterial ischaemic stroke with haemorrhagic transformation). Two of the 11 children with a neurologic complication who had cardiac surgery had additional neurologic symptoms 17 and 32 days after surgery, respectively; these symptoms were not considered to be attributable to surgery. Finally, one additional child had multiple new small emboli on screening repeat MRI 5 days after surgery.
Discussion
To our knowledge, we present the largest paediatric cohort examining the frequency and characteristics of neurologic complications of infective endocarditis. We found that neurologic complications of infective endocarditis were common, occurring in at least 23 of 64 total children (34%, 95% CI 23–46%), or 22 of 43 children with definite infective endocarditis (51%, 95% CI 35–67%). Our findings are similar to estimates of the proportion of adults with IE with neurologic complications, Reference Heiro, Nikoskelainen, Engblom, Kotilainen, Marttila and Kotilainen11 but slightly higher than prior paediatric estimates. Reference AlBassri, Sheikho, Chaikhouni, Al Habshan and Kabbani12 At 1 year from endocarditis symptom onset, neurologic complication-free survival was only 58%. Mortality in this cohort occurred exclusively in children with neurologic complications. Risk factors for neurologic complications in this paediatric cohort included older age, clinical features associated with risk for embolic event (such as pathologic lesions, large and mobile vegetations, vegetations in a location with the potential for systemic embolisation), and a definite (versus possible) diagnosis of infective endocarditis.
While abnormal cardiac anatomy and prosthetic material related to CHD repair could lead to unique structural risk factors for intracranial embolisation of vegetations and subsequent neurologic complications in children with CHD and infective endocarditis, we did not observe a difference in neurologic complications between children with and without CHD, potential right-to-left intracardiac shunt, single-ventricle physiology, or systemic prosthetic valves. This may be due to a relatively small sample size of children with heterogeneous types of CHD. Future large, prospective, multi-site studies could allow construction of multi-variable regression models to examine independent risk factors for neurologic complications by underlying cardiac anatomy.
Nearly, a quarter of the neurologic complications documented in this paediatric cohort were asymptomatic, found incidentally on screening neuroimaging. Since our institution does not have a standard neuroimaging screening protocol for children with infective endocarditis, only 38 children (56%, 95% CI 43–68%) underwent neuroimaging during their acute hospitalisation. Of the 42 children without a documented neurologic complication, only 18 (43%) underwent screening neuroimaging. Up to 80% of adults with infective endocarditis have asymptomatic neurologic complications; Reference Duval, Iung, Klein, Brochet, Thabut and Arnoult18,Reference Hess, Klein, Iung, Lavallee, Ilic-Habensus and Dornic19 therefore, it is very possible that we underestimated the number of asymptomatic neurologic complications. Whether early identification and management of asymptomatic neurologic complications in children with infective endocarditis affect morbidity and mortality remains unknown but is probable in some cases. For example, early identification of a large infectious intracranial aneurysm may lead to stabilising interventions which could prevent later catastrophic haemorrhage. Similarly, early identification of neurologic embolic phenomenon may impact indications for cardiac surgery Reference Baddour, Wilson, Bayer, Fowler, Tleyjeh and Rybak6,Reference Bayer, Bolger, Taubert, Wilson, Steckelberg and Karchmer13,Reference Baltimore, Gewitz, Baddour, Beerman, Jackson and Lockhart15 if there is a treatable lesion. Further investigation is needed to determine whether early identification and treatment of neurologic complications leads to management changes and improved outcomes.
Our study has several limitations. First, the study was conducted at a single paediatric tertiary care referral centre which may introduce referral bias. Therefore, generalising these findings to different care contexts should be undertaken cautiously. Next, our data is from a retrospective cohort. Some details of the clinical courses may not have been available, there may have been more asymptomatic neurologic complications in this cohort that were not identified by their care team which could lead to misclassification bias, and we may have underestimated neurological deficits as certain deficits like cognitive problems may become apparent after discharge. Furthermore, we were unable to determine whether the timing of surgeries for infective endocarditis were affected by concerns about extension of existing haemorrhage or risks of anticoagulation or embolisation during the procedure. Also, our cohort size limits our ability to evaluate all potential risk factors for neurologic complications and to construct multi-variable models. Finally, we lack standardised long-term outcome data.
In conclusion, neurologic complications of infective endocarditis were common in this paediatric cohort with infective endocarditis (34%) and were associated with increased mortality. The true frequency of neurologic complications was likely higher than detected because asymptomatic neurologic complications may have been missed in the absence of routine screening neuroimaging. Older age, a diagnosis of definite infective endocarditis by Duke criteria, and clinical features associated with risk of embolic event were all associated with the presence of identified neurologic complications. Additional prospective studies are required to determine whether risk factors for infective endocarditis may vary based on underlying cardiac anatomy.
All children with infective endocarditis should have a multidisciplinary team approach to their care, by a specialised “endocarditis team.” In addition to early involvement of general cardiology, cardiac surgery, and infectious diseases, we suggest that all children with infective endocarditis have neurologic consultation, neurologic examination, and screening neuroimaging within the first 24–72 hours of their hospitalisation, if clinically stable to do so, and serially if IE treatment is extended over a long period of time. Additional prospective studies with screening neurologic examinations and neurovascular imaging are needed to determine whether early identification of neurologic abnormalities like infectious intracranial aneurysms and microemboli may direct management and ultimately reduce neurologic morbidity and overall mortality.
Supplementary material
For supplementary material accompanying this paper visit https://doi.org/10.1017/S1047951122001159
Data statement
A deidentified data set is available upon request.
Acknowledgements
None.
Financial support
Dr McGuire receives support from NIH K23 NS094069. Dr Licht receives support from NIH R01-NS072338, R01-NS60653, and the June and Steve Wolfson Family Foundation.
Conflicts of interest
None.












