Introduction
Antarctica is a model system for assessing climatic and oceanographic changes in the Southern Hemisphere as well as their impacts on the global climate (Pellizzari et al. Reference Pellizzari, Rosa, Yokoya, Gómez and Huovinen2020). Changes in ice cover and surface thermohaline circulation patterns are expected, especially in polar and temperate regions (Müller et al. Reference Müller, Laepple, Bartsch and Wiencke2009, Guillemin et al. Reference Guillemin, Dubrasquet, Reyes and Valero2018, López et al. Reference López, Macaya, Rivadeneira, Tala, Tellier and Thiel2018). It is necessary to understand how these changes will influence the biogeographical distributions of marine organisms, including macroalgae, which depend on oceanographic dispersal processes. Antarctic macroalgae are sentinels of climate change due to their reproductive cycles and physiology being highly adapted to the extreme conditions of polar environments (Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017), and their assemblages are bioindicators of abrupt environmental changes and processes in Antarctic ecosystems (Oliveira et al. Reference Oliveira, Pellizzari, Medeiros, Yokoya, Gómez and Huovinen2020). On the other hand, some opportunistic macroalgae can rapidly disperse and establish in new regions given suitable conditions (Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017, Reference Pellizzari, Rosa, Yokoya, Gómez and Huovinen2020). Therefore, obtaining a baseline for macroalgal biodiversity is an essential step in order to detect changes in the environment and to support conservation management.
The Weddell Sea (WS), bordered to its west by the eastern Antarctic Peninsula (EAP), hosts a cyclonic oceanographic system known as the Weddell Sea Gyre (Vernet et al. Reference Vernet, Geibert, Hoppema, Brown, Haas and Hellmer2019), which influences the global thermohaline circulation and, consequently, the climate of the Southern Hemisphere (Robertson et al. Reference Robertson, Visbeck, Gordon and Fahrbach2002). This area includes a transition between the Antarctic and sub-Antarctic polar zones and is one of the areas most sensitive to current climatic and oceanographic changes (Vernet et al. Reference Vernet, Geibert, Hoppema, Brown, Haas and Hellmer2019, Fraser et al. Reference Fraser, Morrison, Olmedo Rojas, Gómez and Huovinen2020). However, although the WS has been well studied in terms of glaciological and oceanographic features (Vernet et al. Reference Vernet, Geibert, Hoppema, Brown, Haas and Hellmer2019, Pehlke et al. Reference Pehlke, Brey, Konijnenberg and Teschke2022), its macroalgal assemblages, which are important providers of shelter and habitat for other organisms and, in combination with phytoplankton, contribute to primary productivity and carbon flux, remain poorly investigated. In this area, a very limited number of studies have been published to date. Hope Bay, Seymour Island and Paulet Island were studied between 1940 and 1975 (Skottsberg Reference Skottsberg1941, Neushul Reference Neushul1965, Moe & DeLaca Reference Moe and DeLaca1976). There have been no recent studies of this region's macroalgae, and no baseline of regional macroalgal biodiversity is available.
In comparison, the South Shetland Islands (SSI) are an archipelago to the north-west of the Antarctic Peninsula comprising 11 main islands and representing the area of the Antarctic with the greatest recorded macroalgal biodiversity. The SSI are facing some of the most rapid rates of climate change and other anthropogenic impacts anywhere in Antarctica (Turner et al. Reference Turner, Bindschadler, Convey, di Prisco, Fahrbach and Gutt2009, Dubrasquet et al. Reference Dubrasquet, Reyes, Sanchez, Valdivia and Guillemin2018). Seaweed inventories have established a significant biodiversity database for this region, in particular the compilations of Wiencke & Clayton (Reference Wiencke, Clayton and Wägele2002) and Wiencke et al. (Reference Wiencke, Amsler, Clayton, De Broyer, Koubbi, Griffiths, Raymond and Udekemd'Acoz2014), the comparative taxonomic lists provided for eight islands in the SSI by Pellizzari et al. (Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017) and the most recent checklist compiled for the entirety of Antarctica by Oliveira et al. (Reference Oliveira, Pellizzari, Medeiros, Yokoya, Gómez and Huovinen2020). Several macrophycological studies have also been performed in the western Antarctic Peninsula (WAP) region (Moe & DeLaca Reference Moe and DeLaca1976, Lamb & Zimmermann Reference Lamb and Zimmermann1977, Gallardo et al. Reference Gallardo, Pérez-Ruzafa, Flores-Moya and Conde1999, Amsler et al. Reference Amsler, Iken and McClintock2005, Reference Amsler, McClintock, Baker, GÓMEZ and HUOVINEN2020, Peters et al. Reference Peters, Amsler, Amsler, McClintock, Dunbar and Baker2005, Hommersand et al. Reference Hommersand, Moe, Amsler and Fredericq2009, Mystikou et al. Reference Mystikou, Peters, Asensi, Fletcher, Brickle and van West2014).
Polar macroalgae are adapted to extreme and stressful environmental conditions (Gómez & Houvinen Reference Gómez and Huovinen2015), such as wide seasonal variations in photoperiod (long periods of light and dark), irradiance and ultraviolet (UV) radiation exposure. Their distributions reflect adaptations to distinct environmental conditions, such as temperature, salinity, pH, ice dynamics and substrate availability for recruitment (Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017). Due to the isolation and the extreme abiotic stresses that the Antarctic macroalgal flora faces, it is characterized by a high degree of endemism (Wiencke & Clayton Reference Wiencke, Clayton and Wägele2002). However, studies have also supported the ability of rafts of floating macroalgae and their associated biota to overcome biogeographical barriers previously generally considered impenetrable to passive distribution, such as the Antarctic Circumpolar Current (ACC), carrying propagules, macroalgae currently not present in Antarctica and benthic invertebrates as epiphytes across the Southern Ocean (Barnes et al. Reference Barnes, Hodgson, Convey, Allen and Clarke2006, Fraser et al. Reference Fraser, Morrison, Hogg, Macaya, van Sebille and Ryan2018, Reference Wiencke, Amsler, Clayton and Van de Putte2020, Macaya et al. Reference Macaya, Tala, Hinojosa, Rothäusler, Gómez and Huovinen2020). Such floating beds of large brown seaweeds (e.g. Durvillaea antarctica) with the ability to drift for long periods can thereby become vectors for the expansion of species distributions and enable greater connectivity between different ecoregions, notably Patagonia, the sub-Antarctic and Antarctica.
In general, higher species richness, including both endemic and some widely distributed species, is expected at lower Antarctic latitudes (e.g. the SSI, 61–63°S) and around the sub-Antarctic islands (~50–55°S), and lower richness is expected at higher latitudes (EAP and WAP, > 65°S). The WS (bordering the EAP) also shows distinct features due to its complex ocean circulation, bathymetry and thermohaline patterns, in comparison with the SSI and WAP. Parts of the SSI are also subject to a history of intense human impact (e.g. Deception Island, King George Island; Hughes et al. Reference Hughes, Pertierra, Molina-Montenegro and Convey2015, López et al. Reference López, Macaya, Rivadeneira, Tala, Tellier and Thiel2018, McCarthy et al. Reference McCarthy, Peck, Hughes and Aldridge2019).
The present study compared macroalgal assemblages from distinct oceanographic regions, reporting new species records (NRs) of macroalgae from Vega Island (WS, north-east Antarctic Peninsula) and updating the available checklist for the SSI. The records and illustrations provided expand the utility of the Antarctic phycological database. Finally, we discuss the macroecological and biogeographical implications of the records presented in the context of possible changes in assemblage richness, composition and distribution under current climatic and oceanographic patterns and trends, providing a foundation for future integrative phylogeographical and molecular studies.
Materials and methods
Study sites
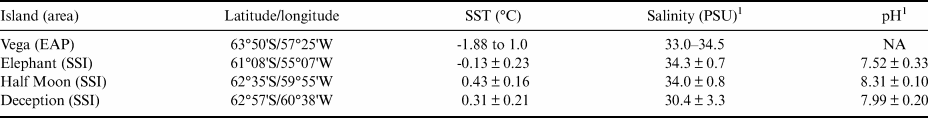
The WS exhibits hydrographical and topographical features linked to a cyclonic gyre, bounded to the south by the Antarctic continent, to the west by the Antarctic Peninsula and to the north by the ACC and mid-oceanic Scotia Ridge. The western sector of the WS is covered by extensive pack ice, even during the summer. The climate is influenced by continental atmospheric currents originating further south, by low-pressure systems from the north and by westerly winds crossing the Antarctic Peninsula. Cold air carried towards lower latitudes generates cold fronts in several regions of the Southern Hemisphere. These winds influence the temperatures experienced in the EAP and force ice to drift north-eastwards into the South Atlantic (Vernet et al. Reference Vernet, Geibert, Hoppema, Brown, Haas and Hellmer2019). The WS is stratified into three bathymetric zones, and its thermohaline patterns are described by Robertson et al. (Reference Robertson, Visbeck, Gordon and Fahrbach2002) and are shown in Table I.
Table I. Thermohaline surface water patterns in the Weddell Sea (Robertson et al. Reference Robertson, Visbeck, Gordon and Fahrbach2002) and from the South Shetland Islands (SSI) sample sites (retrieved from National Oceanic and Atmospheric Administration; NOAA). pH data from the sampled islands in the SSI are also presented.

1 Pellizzari et al. (Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017); SST data retrieved from NOAA.
EAP = eastern Antarctic Peninsula; NA = not available; PSU = practical salinity units; SST = sea surface temperature.
Sampling surveys in the present study were performed on Vega Island (WS), Elephant Island, Half Moon Island and Deception Island (SSI; Fig. 1). Vega Island (63°50'S; 57°25'W) is located east of the northern Antarctic Peninsula and is 8 km from James Ross Island. It has continuous permafrost, an annual mean atmospheric temperature of -4.5°C and a total area of 268 km2 (Robertson et al. Reference Robertson, Visbeck, Gordon and Fahrbach2002), ~70% of which is covered by ice. The mean sea surface temperature (SST) in the vicinity of the island between 2011 and 2021 was -1.41°C, with salinity between 33.0 and 34.5 practical salinity units (PSU; Table I; WOD/NOAA, 2021). The breakup and melt of coastal sea ice during the summer exposes limited areas of rocky substrate suitable for the growth of benthic organisms including macroalgae.

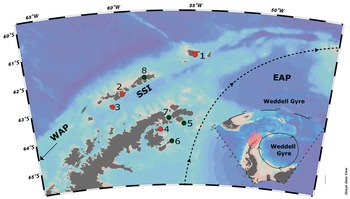
Fig. 1. Map of the Antarctic Peninsula and surrounding islands, showing sampled sites in the present study (red dots): 1 = Elephant Island; 2 = Half Moon Island; 3 = Deception Island (South Shetland Islands; SSI); 4 = Vega Island (eastern Antarctic Peninsula; EAP). Further islands in the Weddell Sea (EAP) and the SSI in the literature compilation for seaweed inventories (black dots): 5 = Paulet Island; 6 = Seymour Island; 7 = Hope Bay; 8 = King George (SSI). Map generated using Ocean Data View (https://odv.awi.de). WAP = western Antarctic Peninsula.
The SSI lie ~100 km north-west of the Antarctic Peninsula, separated from it by the Bransfield Strait. The northern-most of the SSI, Elephant Island, is located 885 km from Cape Horn at the south of the Drake Passage, in a transitional area between the northern WS and the eastern SSI, and also in a region of oceanographic connectivity between the EAP and sub-Antarctic South Georgia. Studies have reported high biological connectivity between Antarctica, South America, Oceania and New Zealand (Bussolini & Waters Reference Bussolini and Waters2015, Griffiths & Waller Reference Griffiths and Waller2016, Sanches et al. Reference Sanches, Pellizzari and Horta2016, González-Wevar et al. Reference González-Wevar, Segovia, Rosenfeld, Maturana, Jeldres and Pinochet2022). Elephant Island's rocky shores are unique in the Maritime Antarctic, showing rugged and irregular features, with the substrate in the mid- and upper subtidal zones consisting of pebbles and rocks.
Deception Island is a flooded volcanic caldera of ~12 km diameter. The island most recently erupted in the late 1960s and continues to be geothermally active, resulting in thermal anomalies in marine sediments and in the shallow seawater column of the flooded caldera (Port Foster) of up to 8°C (Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017). Active coastal fumaroles and hot springs are present in Fumarole Bay, Telefon Bay, Whalers Bay and Pendulum Cove. Beaches surrounding the caldera are composed of black sand, with some outcrops of volcanic rocks. Port Foster is a natural and sheltered harbour that was the site of a sizeable onshore whaling station during the early 20th century. It is one of the most popular visitor sites in Antarctica, with considerable tourist and national operator activity during the summer months (Hughes et al. Reference Hughes, Pertierra, Molina-Montenegro and Convey2015, López et al. Reference López, Macaya, Rivadeneira, Tala, Tellier and Thiel2018, McCarthy et al. Reference McCarthy, Peck, Hughes and Aldridge2019). Pellizzari et al. (Reference Pellizzari, Rosa, Yokoya, Gómez and Huovinen2020) considered the island to be one of the most sensitive and vulnerable areas to the impacts of any introduction of non-native marine species (see also Nuñez-Pons & Avila Reference Nuñez-Pons and Avila2014, Angulo-Preckler et al. Reference Angulo-Preckler, Figuerola, Núñez-Pons, Moles, Martín-Martín and Rull-Lluch2018). The island is already known to host the greatest number of non-native terrestrial species in the Antarctic continent (Hughes et al. Reference Hughes, Pertierra, Molina-Montenegro and Convey2015). Ongoing monitoring is key to identifying climate- or environmentally driven changes given the intensity of human activities around the island during the summer.
Half Moon Island is a small island close to Livingston Island and Deception Island, bordering an embayment of ~1.71 km2 formed in the Holocene at the junction of three smaller islands. Protected from strong winds and seas by Livingston Island, its rocky shoreline is characterized by mid- and upper intertidal zones formed of pebbles that are colonized by seaweed. Tourist activities on the island are increasing. Pellizzari et al. (Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017) partially reported the only recent phycofloristic survey of the island, with additional information from that survey being presented in the current study.
Sampling and laboratory analysis
The surveys took place during summer research cruises of the Brazilian Navy research vessel Almirante Maximiano (H41). Macroalgae were sampled in randomly distributed 100 m transects (n = 6), parallel to the coastline in the intertidal and shallow subtidal zones. Specimens were collected manually (n = 3 per transect), preferentially fertile, as follows: 1) Deception Island (|December 2014, 2016, 2017), 2) Elephant Island (December 2014), 3) Half Moon Island (|December 2013, 2016), 4) Vega Island (February 2017). Half Moon Island was also opportunistically sampled using a box corer (0.8 m2, 25 m depth, n = 3 launches). The subtidal zone assemblage is very different from that of the intertidal zone, but the species found in box core samples were included in the general checklist. Epiphytic diversity, including Cyanobacteria, was also identified to the lowest taxonomic level possible.
The collected material was initially sorted, washed and processed in the onboard laboratory, stored in polyethylene flasks and preserved using formalin (4% diluted in seawater). The material was labelled and transported to the laboratory in Brazil for further taxonomic analysis. Macroalgal specimens were analysed focusing on the morpho-anatomical features of their vegetative and reproductive regions. Histological sections and/or filaments were used to assess: cell size; number, position and shape of chloroplasts; shape and size of uni- and multi-cellular fertile structures; organization of branching; type, size and shape of the apical cells; and exclusive morphological structures. The material was analysed using stereoscopic and optical microscopy with phase contrast and image capture (Motic SMZ-161, OLYMPUS CX31, OLYMPUS CX41). Some specimens were stained (Rhodophyta) with Rose Bengal, and the images were treated and/or transformed into black and white to improve resolution and standardization. In some formalin-preserved specimens from polar waters it is common to lose the natural colour, and staining the material aims to provide improved contrast in the images to help with observation of certain important taxonomic features. Taxonomic identification was performed by comparing the specimens obtained with the available specialized bibliography of seaweeds from the sub-Antarctic, Antarctic Peninsula and SSI (primarily Lamb & Zimmermann Reference Lamb and Zimmermann1977, Ricker Reference Ricker1987, Wiencke & Clayton Reference Wiencke, Clayton and Wägele2002, Oliveira et al. Reference Oliveira, Pellizzari and Oliveira2009). Nomenclatural updates follow Guiry & Guiry (Reference Guiry and Guiry2023). Specimens representing NRs of macroalgae are in the process of deposition in the collection of the Museu Botânico Municipal (MBM) Herbarium (Curitiba Botanical Garden, Paraná, Brazil), which will allocate voucher codes.
Literature review, abiotic and biotic data: processing and analysis
The data obtained in the current study were combined with records from the literature and other data referring to macroalgae from other areas under the influence of the WS, gathered from the Google Scholar database and the Scientific Committee on Antarctic Research (SCAR) Biogeographic Atlas of the Southern Ocean - macroalgae - data platform (compiled by Wiencke et al. Reference Wiencke, Amsler, Clayton and Van de Putte2020) in order to carry out a comparative statistical analysis between the SSI, WAP and EAP sectors. Data were compiled in spreadsheets (Microsoft Office Excel ® 2010) and statistical analyses and graphical outputs were generated (R Core Team 2022). Moe & DeLaca (Reference Moe and DeLaca1976) reported macroalgal assemblages from Elephant Island. The macroalgal flora of King George Island is the most studied in the SSI (Oliveira et al. Reference Oliveira, Pellizzari and Oliveira2009, Quartino et al. Reference Quartino, Deregibus, Campana, Latorre and Momo2013, Valdivia et al. Reference Valdivia, Diaz, Holtheuer, Garrido, Huovinen and Gómez2014). Gallardo et al. (Reference Gallardo, Pérez-Ruzafa, Flores-Moya and Conde1999) reported collections of seaweeds from Livingston Island and Deception Island (SSI) and Trinity Island (Bransfield Strait). Pellizzari et al. (Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017) presented data comparing macroalgal assemblages from eight islands of the SSI, including the islands analysed here (Elephant, Deception, Half Moon). In addition to the present results from Vega Island, for the WS coastal region of the EAP the checklists of Skottsberg (Reference Skottsberg1941) for Paulet Island (63°36'S, 55°48'W) and Seymour Island (64°20'S 56°38'W) and of Neushul (Reference Neushul1965) and Moe & DeLaca (Reference Moe and DeLaca1976) for Hope Bay (63°23'S, 57°00'W) and Elephant Island (SSI) were consulted.
The macroalgal assemblage compositions were compared using presence/absence data applied in a permutational multivariate analysis of variance (PERMANOVA) based on Jaccard's dissimilarity matrix and tested for homoscedasticity using the betadisper function ('vegan' package; R Core Team 2020). One-way analysis of variance (ANOVA) was used to test for significant differences in richness among the analysed sectors (SSI, WAP and EAP). Data were tested for normality using the Shapiro-Wilk test and transformed (square root) when not normal, with the homoscedasticity of variances tested using the Bartlett function.
Venn diagrams were generated (R Studio software) to illustrate the richness shared between the islands or sectors (SSI, EAP and WAP). The frequencies of taxa were calculated using the Dajoz constancy index (CI). Using this index, those that occurred in > 50% of the samples were considered constant or conspicuous, those between 25% and 50% were considered accessory taxa and those occurring in < 25% of samples were considered accidental (rare).
SST was visualized using Ocean Data View software (https://odv.awi.de/), and principal component analysis (PCA) data were recovered from Surface Ocean CO2 Atlas (https://www.socat.info/), representing the sampling area of the present study using data obtained in 2016. The mean SST for each island, along with richness data for green, brown and red macroalgae, were used in a PCA.
Results
Diversity and richness
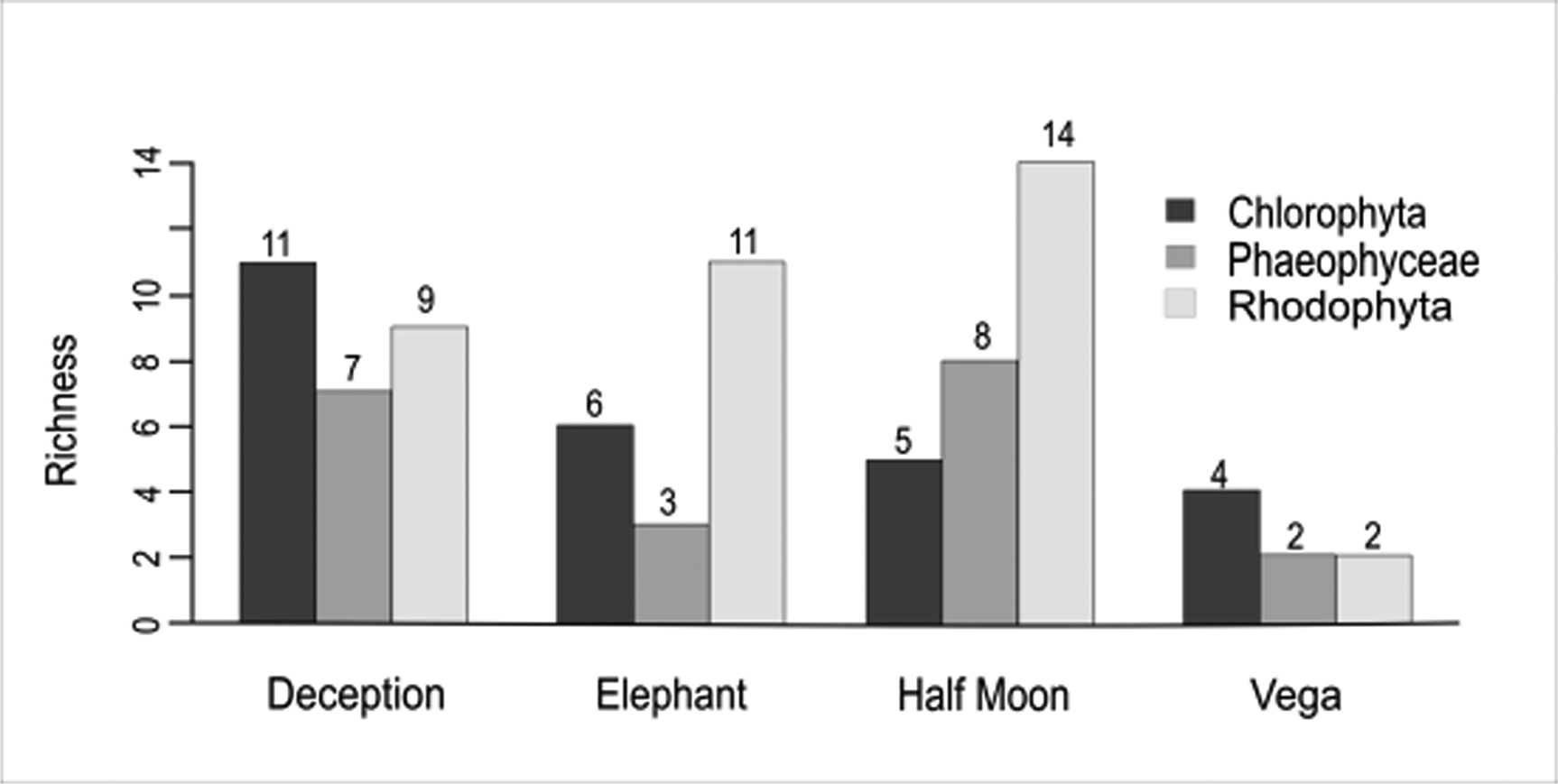
A total of 48 macroalgal taxa were recorded in the samples from the four islands analysed: 45 at the specific level, two at the generic level and one at the familial level (Table II). Among these, 13 species represented Chlorophyta (27%), 13 represented Phaeophyceae (27%) and 22 represented Rhodophyta (46%). Macroalgal species richness at Deception Island and Half Moon Island was greatest (27 species), followed by Elephant Island (20 species) and Vega Island (8 species; Fig. 2), the latter representing < 10% of the known richness of the SSI. The families contributing the most diversity were Ulotrichaceae and Delesseriaceae, followed by Desmarestiaceae.

Fig. 2. Number of species by macroalgal group on Deception, Elephant, Half Moon (South Shetland Islands) and Vega islands (eastern Antarctic Peninsula, Weddell Sea). Generated in RStudio with the Sciplot package.
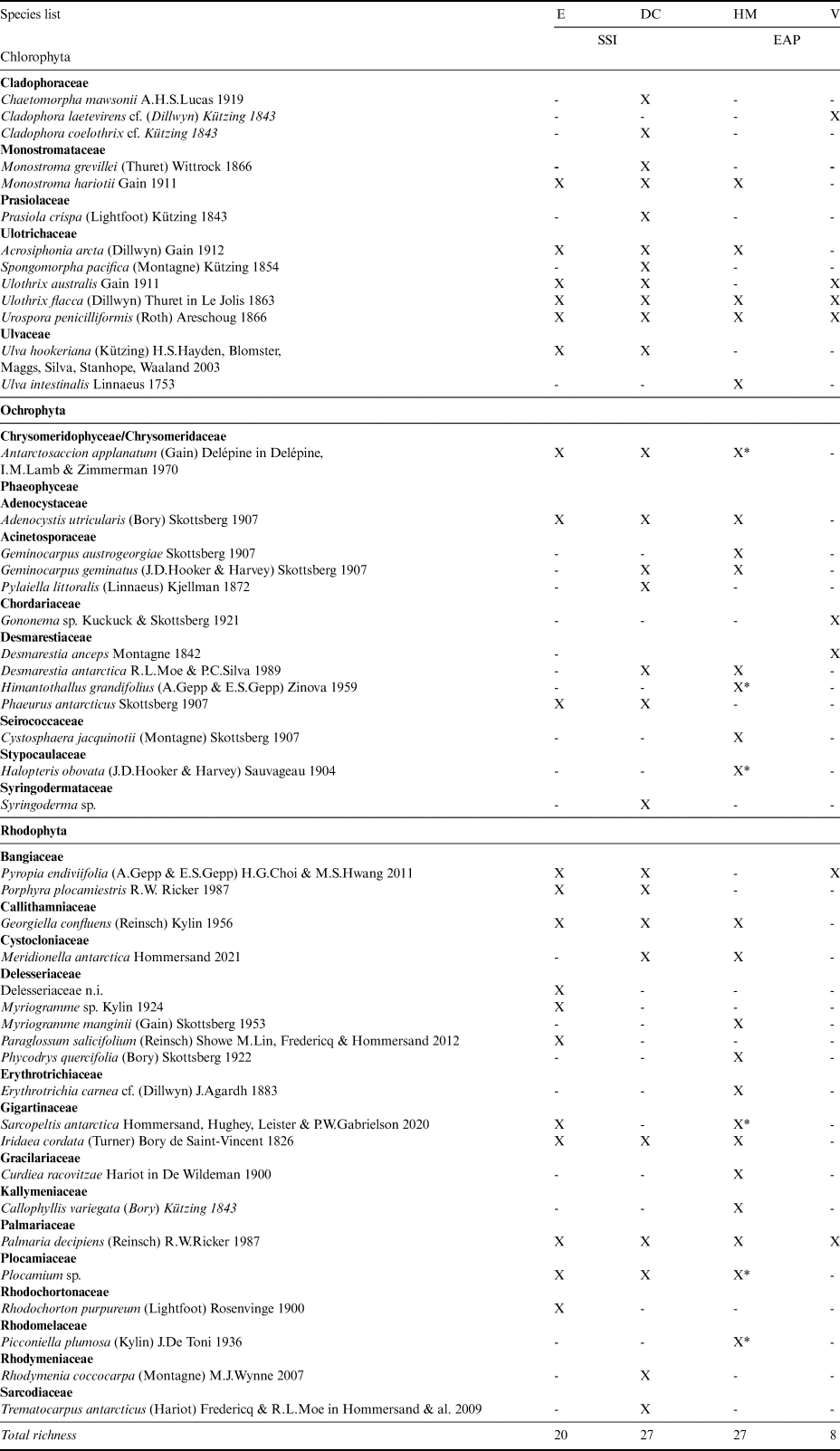
Table II. List of macroalgae - presence (X) or absence (-) by group and family in the Antarctic islands analysed.

DC = Deception Island; E = Elephant Island; EAP = eastern Antarctic Peninsula; HM = Half Moon Island; SSI = South Shetland Islands; V = Vega Island; X* = collected on Half Moon Island using a box corer.
As this is the first study of marine benthic macroalgae from Vega Island, all eight species records are new to the island. The following species are NRs either to the EAP sector or to the Maritime Antarctic coast. Amongst the Chlorophyta, Cladophora laetevirens is a cosmopolitan species distributed from the Arctic to the Falkland Islands, but this is the first record from Antarctica. Ulothrix australis is known from the SSI, but not from the EAP; Ulothrix flacca has previously only been reported from sub-Antarctic Kerguelen Island (49°20′S, 69°20′E). Urospora penicilliformis is also known from the SSI. Amongst the brown algae, Gononema cf. ramosum has previously only been reported from sub-Antarctic Macquarie Island and Desmarestia anceps is known from the SSI. Amongst the Rhodophyta, Pyropia endiviifolia is known from the SSI and various sub-Antarctic islands, and Palmaria decipiens is broadly distributed in Antarctica and the sub-Antarctic.
Several species were NRs for Elephant Island: Monostroma hariotii, Acrosiphonia arcta and U. australis (Chlorophyta) and Rhodochorton purpureum and Porphyra plocamiestris (Rhodophyta), although all have been recorded elsewhere in the SSI. The Half Moon Island and Deception Island records included five NRs for each island. For Half Moon Island these were Geminocarpus austrogeorgiae and Halopteris obovata (Phaeophyceae) and Callophyllis variegata, Erythrotrichia carnea and Phycodrys quercifolia (Rhodophyta). The following taxa were recorded in the opportunistic box corer samples from 25 m depth: Antarctosaccion applanatum, Himantothallus grandifolius, Halopteris obovata, Sarcopeltis antarctica, Plocamium sp. and Picconiella plumosa. For Deception Island, the NRs included Antarctosaccion applanatum, Geminocarpus geminatus and Phaeurus antarcticus (Phaeophyceae) and Meridionella antarctica and Porphyra plocamiestris (Rhodophyta). A high frequency of pennate colonial epiphyte diatoms was noted, in addition to filamentous cyanobacteria.
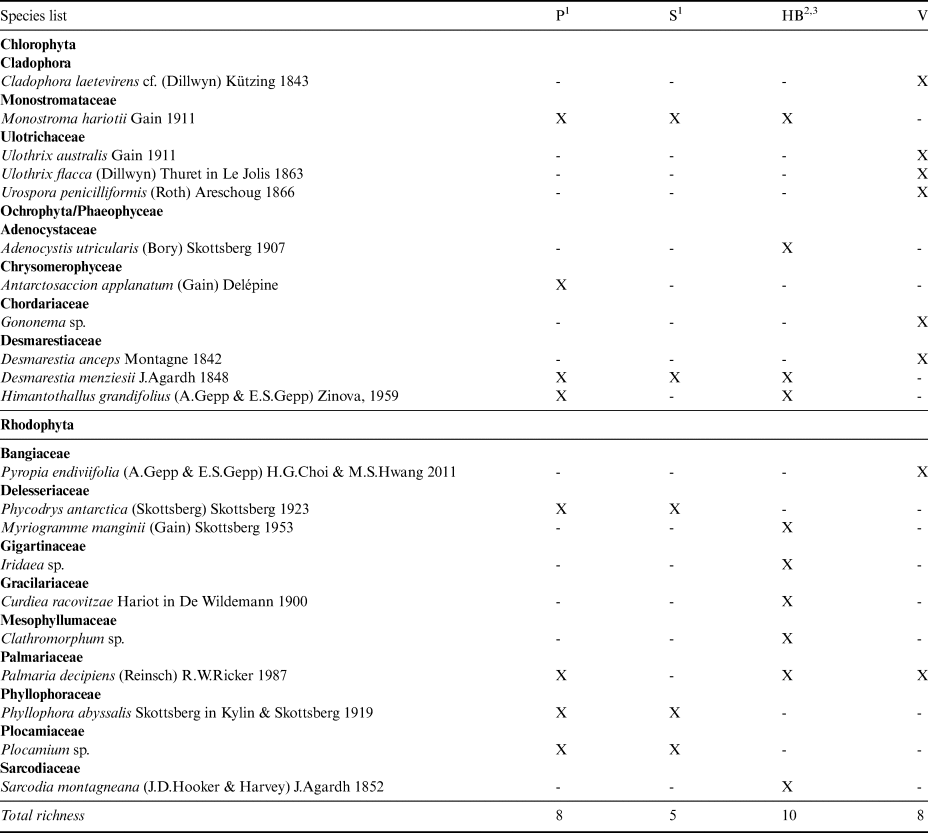
The three previous taxonomic studies reporting macroalgae in the EAP sector of the WS listed a total of 14 species from this region, including one species of Chlorophyta, four Phaeophyceae and nine Rhodophyta. Only Monostroma hariotii and Desmarestia menziesii were reported as conspicuous for the EAP locations included in the analysis using previous data (Paulet Island, Seymour Island and Hope Bay) and for Vega Island (current study). In total, our eight NRs for Vega Island (Table III) update the EAP list to 22 species.
Table III. Macroalgae list from the eastern Antarctic Peninsula in terms of presence (X) and absence (-).

1Skottsberg (Reference Skottsberg1941).
2Neushul (Reference Neushul1965).
3Moe & DeLaca (Reference Moe and DeLaca1976).
HB = Hope Bay; P = Paulet Island; S = Seymour Island; V = Vega Island, sampled in the present survey.
Figures S1–S5 show images of the sampled phycological material in order to provide a visual database and assist in further taxonomic identifications. Figures S1 & S2 illustrate the utility of staining specimens with Rose Bengal to elucidate morphological features. Figures S3–S5 show the filamentous macroalgae of the three different seaweed groups.
Literature data and statistical analyses
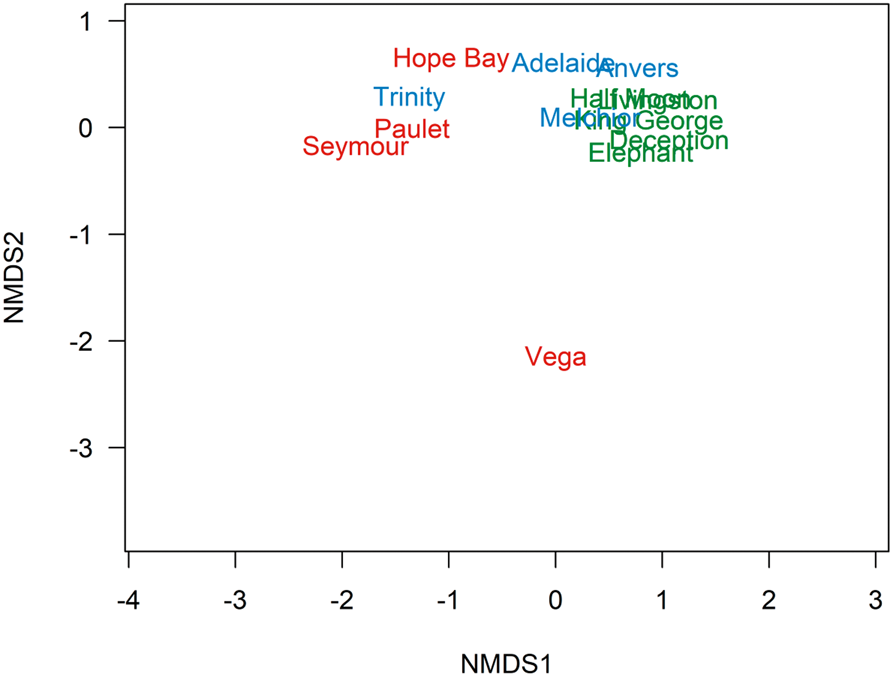
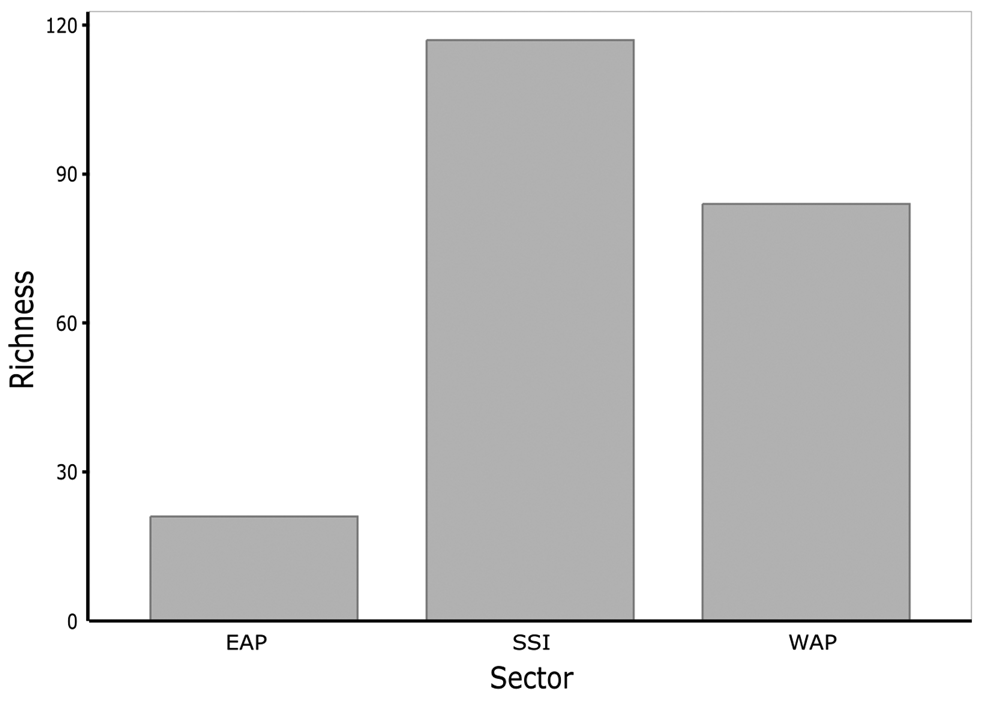
The PERMANOVA generated P values between sectors of: EAP vs SSI = 0.0240; EAP vs WAP = 0.7689; and SSI vs WAP = 0.0252. In the non-metric multidimensional scaling (nMDS), three distinct clusters were formed representing EAP, SSI and WAP when data from the current study were combined with literature data, suggesting distinct differences in richness between sectors, particularly for the EAP, where Vega Island presented the most distinct richness and was isolated in the nMDS (Fig. 3). The EAP showed the lowest richness, followed by the WAP, and then the SSI, with 21, 82 and 112 taxa, respectively (Fig. 4).

Fig. 3. Ordination plot by a non-multidimensional scaling (nMDS) based on Jaccard's index (presence-absence data) showing dissimilarity in the composition of macroalgal assemblages in the South Shetland Islands (blue), eastern Antarctic Peninsula (red) and western Antarctic Peninsula (green) sectors, based on the current study and literature data (see the ‘Materials and methods’ section for details of sources; stress = 0.074).

Fig. 4. Macroalgal richness of Antarctic islands in the three sectors analysed (South Shetland Islands (SSI), eastern Antarctic Peninsula (EAP) and western Antarctic Peninsula (WAP)) based on data obtained in the current study and from the literature (see the ‘Materials and methods’ section for details of sources).
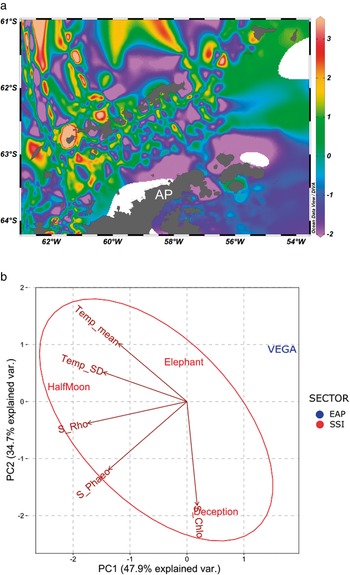
In the SST data, plotted using Ocean Data View (Fig. 5a), the pattern of higher temperatures surrounding the SSI when compared with the EAP is clear. The PCA also provided strong support for the isolation of Vega Island compared to the three islands analysed in the SSI (Fig. 5b). Considering the SSI, it also suggested that the shallow-water geothermal anomalies affecting Deception Island may have influenced the higher richness of Chlorophyta present (Fig. 2). Elephant Island and Half Moon Island, which are influenced by more typical temperature patterns for the SSI, showed ranked richness proportions of macroalgal groups as expected for this area (Rhodophyta < Phaeophyceae < Chlorophyta).

Fig 5. a. Sea surface temperature (SST) map generated using Ocean Data View software (https://odv.awi.de/) and data obtained from the Surface Ocean CO2 Atlas and database for the current study area during 2016 (https://www.socat.info/). AP = Antarctic Peninsula. b. Principal component analysis (PCA) generated using SST data and total richness of S_Chlo (Chlorophyta), S_Rho (Rhodophyta) and S_Phaeo (Phaeophyceae). Temp mean = sea surface temperature mean (°C); Temp_SD = sea surface temperature standard deviation.
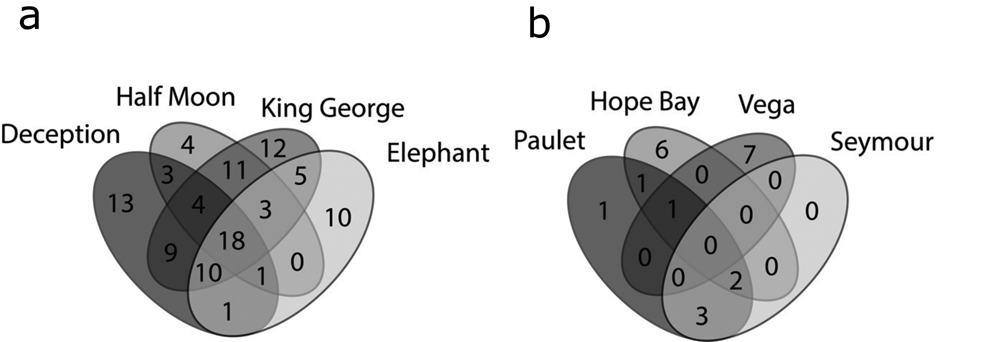
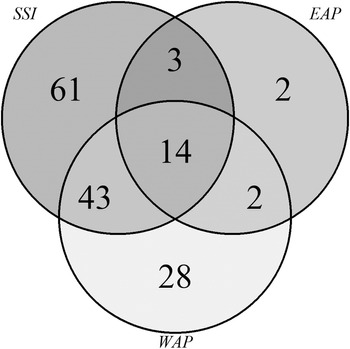
Venn diagrams (Fig. 6a) again showed that the SSI (Fig. 6a) had higher richness and shared numbers of species among the islands, while in the EAP (Fig. 6b) the richness values were lower and variable. Shared richness and number of exclusive (rare) taxa among the SSI, EAP and WAP are shown in Fig. 7. A total of 14 species were shared among the sectors, with 61, 2 and 28 taxa being exclusive for each sector (SSI, EAP and WAP, respectively). The two taxa exclusive to the EAP were C. laetevirens and the Ectocarpales Gononema sp. (cf. ramosum). Ulothrix was recorded only from the SSI and EAP.

Fig. 6. Venn diagrams based on the numbers of exclusive and shared (common) macroalgal species: a. South Shetland Islands and b. eastern Antarctic Peninsula, based on data obtained in the present study and from the literature (see the ‘Materials and methods’ section for details of sources).

Fig. 7. Venn diagram illustrating the number of exclusive and shared (common) macroalgal species compared among regions (South Shetland Islands (SSI), eastern Antarctic Peninsula (EAP) and western Antarctic Peninsula (WAP)) based on data obtained in the present study and from the literature (see the ‘Materials and methods’ section for details of sources).
The most common species in the data and literature collated here, including the WAP data, were A. applanatum, D. menziesii, D. anceps, Adenocystis utricularis, H. grandifolius, M. hariotii, U. penicilliformis, Clathromorphum sp., Curdiea racovitzae, Iridaea sp., Myriogramme manginii, Plocamium sp., P. endiviifolia and P. decipiens. Comparing the diversity of islands only from the EAP and SSI, the Dajoz CI indicated that, in the SSI, 36% of the species were conspicuous, contrasting with only 14% in the EAP. Considering both sectors in an integrated analysis, 9.3% of species were conspicuous, 53.3% were accessory and 37.4% were rare.
Discussion
The surveys of all of the studied islands generated new macroalgal records, increasing the known distributional ranges of the species concerned and suggesting that the regions surveyed still require further baseline and ongoing monitoring of their diversity. In the case of Vega Island, these results highlight the paucity of macroalgal studies in the north-east Antarctic Peninsula, suggesting the need for increased survey effort in this sector. The known macroalgal diversities of the SSI, EAP and WAP regions, including data retrieved from the literature, were significantly different (Figs 3 & 4), probably associated with distinct regional patterns of temperature (Fig. 5a,b), salinity, seasonal ice dynamics, sedimentology and substrate availability (Veit-Kohler et al. Reference Veit-Kohler, Durst, Schuckenbrock, Hauquier, Duran and Dorschel2018, Turner et al. Reference Turner, Guarino, Arnatt, Jena, Marshall and Phillips2020).
Macroalgal richness in the SSI was higher and its assemblage composition was statistically different from that of Vega Island and from neighbouring locations off the north-east Antarctic Peninsula (cf. Skottsberg Reference Skottsberg1941, Neushul Reference Neushul1965, Moe & DeLaca Reference Moe and DeLaca1976). Macroalgal assemblages in the SSI include a total of 104 recorded species (24 Chlorophyta, 28 Phaeophyceae and 52 Rhodophyta; Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017), ~90% of the macroalgal diversity recorded for Antarctica in total (Oliveira et al. Reference Oliveira, Pellizzari, Medeiros, Yokoya, Gómez and Huovinen2020), with Rhodophyta dominating richness, followed by Phaeophyceae and then Chlorophyta. In the present study, Deception Island and Vega Island presented exceptions to this generalization, showing higher richness of Chlorophyta. Pellizzari et al. (Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017) reported higher seaweed richness around Livingston Island, King George Island and Deception Island (86, 76 and 51 species, respectively). The data presented here further corroborate the lower richness around Elephant Island and Half Moon Island (32 and 36 species, respectively), despite including NRs.
The unusual volcanic environment of Deception Island was associated with lower richness (see also Nuñez-Pons & Avila Reference Nuñez-Pons and Avila2014, Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017, Angulo-Preckler et al. Reference Angulo-Preckler, Figuerola, Núñez-Pons, Moles, Martín-Martín and Rull-Lluch2018) compared to other islands in the SSI, including Elephant Island and Half Moon Island. Pellizzari et al. (Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017) reported a predominance of opportunistic Chlorophyta at Deception Island. The present study also reported NRs of green algae at Elephant Island and Vega Island. The SSI have experienced considerable climate warming since the mid-20th century (Turner et al. Reference Turner, Bindschadler, Convey, di Prisco, Fahrbach and Gutt2009), whilst the geothermal natural warming that characterizes Deception Island also contributes to making it one of the most vulnerable islands to colonization by non-native species in Antarctica. In this context, it has been recognized that the high level of human activity at Deception Island, in terms of both tourism and research activities, could influence future biological invasions (see Hughes et al. Reference Hughes, Pertierra, Molina-Montenegro and Convey2015, López et al. Reference López, Macaya, Rivadeneira, Tala, Tellier and Thiel2018, McCarthy et al. Reference McCarthy, Peck, Hughes and Aldridge2019).
Of the NRs obtained in the current study, only E. carnea, collected on Half Moon Island, had previously been recorded in the sub-Antarctic islands (Papenfuss Reference Papenfuss1964) and on the Ross Sea coast of Victoria Land (Wiencke & Clayton Reference Wiencke, Clayton and Wägele2002). R. purpureum, recorded from Elephant Island, has previously been recorded from the Antarctic Peninsula and sub-Antarctic islands (Papenfuss Reference Papenfuss1964, Hommersand et al. Reference Hommersand, Moe, Amsler and Fredericq2009). These NRs most probably illustrate the lack of baseline data and surveys, although them being the result of recent expansion of biogeographical distribution cannot be ruled out, as reported by Guillemin et al. (Reference Guillemin, Dubrasquet, Reyes and Valero2018) and Pellizzari et al. (Reference Pellizzari, Rosa, Yokoya, Gómez and Huovinen2020).
Wiencke et al. (Reference Wiencke, Amsler, Clayton, De Broyer, Koubbi, Griffiths, Raymond and Udekemd'Acoz2014) described West Antarctica as a macroalgal hotspot compared to East Antarctica. Our analyses, considering a more restricted area, suggest that the SSI in particular are a diversity hotspot compared to the WAP, and even more so compared to the EAP, consistent with reports by Sanches et al. (Reference Sanches, Pellizzari and Horta2016), Pellizzari et al. (Reference Pellizzari, Rosa, Yokoya, Gómez and Huovinen2020) and Oliveira et al. (Reference Oliveira, Pellizzari, Medeiros, Yokoya, Gómez and Huovinen2020). Of the eight species recorded here from Vega Island, only P. decipiens has been recorded in the limited number of studies available from other islands in the WS. These new regional records increase the known EAP diversity to 22 species from the previous total of 14 species reported in the studies of Neushul (Reference Neushul1965), Moe & DeLaca (Reference Moe and DeLaca1976) and Skottsberg (Reference Skottsberg1941). The scarcity of available data from the WS, including any application of modern molecular approaches, means that assessments of baseline diversity for the EAP and its connectivity with other regions remain unavailable.
Although numerous studies have tested for associations between oceanographic connectivity and the dispersal ability of marine biota (Bussolini & Waters Reference Bussolini and Waters2015), relatively few have considered oceanographic constraints on passively dispersing rafting assemblages and the influences that these may have on changing biogeographical distributions. Fraser et al. (Reference Fraser, Zuccarello, Spencer, Salvatore and Garcia2013) carried out a phylogeographical analysis using molecular markers of two widespread Southern Hemisphere macroalgal species, sampling in southern Chile, New Zealand and sub-Antarctic islands, following the path of the ACC. Both species A. utricularis and Bostrychia ntricate showed genetic signatures of recent dispersal across long distances, presumably by attachment to floating substrata such as wood or buoyant macroalgae. In terms of connectivity, Bussolini & Waters (Reference Bussolini and Waters2015) studied rafts of D. antarctica in New Zealand and observed three major regional genetic assemblages, suggesting that passive-drifting kelp rafts and associated organisms (mainly epiphytes and opportunistic species) may reflect broad oceanographic changes. The NR of the opportunistic species C. laetevirens at Vega Island is notable. The previous known southern distributional limit of this green algal species was the cold temperate Falkland Islands (Mystikou et al. Reference Mystikou, Asensi, DeClerck, Müller, Peters and Tsiamis2016), suggesting a possible distributional range extension. Notably, different species of Cladophora were recorded across the three sectors (Table S1), but, being cryptic, the genus requires detailed molecular investigation.
The compilation by Wiencke et al. (Reference Wiencke, Amsler, Clayton, De Broyer, Koubbi, Griffiths, Raymond and Udekemd'Acoz2014) of biogeographical information about Antarctic macroalgal richness indicated higher richness mainly in the SSI and along the lower latitudes of the WAP, with lower richness at higher latitudes in the Ross Sea and WS. Ecologically important Antarctic endemic species such as Himantothallus grandifolius, Cystosphaera jacquinotii, Ascoseira mirabilis (Phaeophyceae), Phycodrys antarctica and Trematocarpus antarcticus (Rhodophyta) are conspicuous in the SSI. This is consistent with the data we have compiled here where, among the 140 taxa identified (Table S1) across the three sectors, only 21 species were recorded from the EAP, in contrast to 112 from the SSI (61 exclusively) and 82 from the WAP. Seventeen species were common to the SSI and EAP, and only two were exclusive to the EAP. The lower richness observed at Vega Island is consistent with previous reports for the EAP (Skottsberg Reference Skottsberg1941, Neushul Reference Neushul1965, Moe & DeLaca Reference Moe and DeLaca1976, Wiencke & Clayton Reference Wiencke, Clayton and Wägele2002) compared with the WAP (Moe & DeLaca Reference Moe and DeLaca1976, Lamb & Zimmermann Reference Lamb and Zimmermann1977, Gallardo et al. Reference Gallardo, Pérez-Ruzafa, Flores-Moya and Conde1999, Amsler et al. Reference Amsler, Iken and McClintock2005, Reference Amsler, McClintock, Baker, GÓMEZ and HUOVINEN2020, Peters et al. Reference Peters, Amsler, Amsler, McClintock, Dunbar and Baker2005, Hommersand et al. Reference Hommersand, Moe, Amsler and Fredericq2009, Mystikou et al. Reference Mystikou, Peters, Asensi, Fletcher, Brickle and van West2014, Dubrasquet et al. Reference Dubrasquet, Reyes, Sanchez, Valdivia and Guillemin2018) and the SSI (Gallardo et al. Reference Gallardo, Pérez-Ruzafa, Flores-Moya and Conde1999, Wiencke & Clayton Reference Wiencke, Clayton and Wägele2002, Hommersand et al. Reference Hommersand, Moe, Amsler and Fredericq2009, Oliveira et al. Reference Oliveira, Pellizzari and Oliveira2009, Reference Oliveira, Pellizzari, Medeiros, Yokoya, Gómez and Huovinen2020, Nuñez-Pons & Avila Reference Nuñez-Pons and Avila2014, Sanchez et al. Reference Sanches, Pellizzari and Horta2016, Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017, Reference Pellizzari, Rosa, Yokoya, Gómez and Huovinen2020, Angulo-Preckler et al. Reference Angulo-Preckler, Figuerola, Núñez-Pons, Moles, Martín-Martín and Rull-Lluch2018, Dubrasquet et al. Reference Dubrasquet, Reyes, Sanchez, Valdivia and Guillemin2018).
Historically, some species of macroalgae, including Iridaea ‘cordata’, Gigartina skottsbergii and Plocamium ‘cartilagineum’, have been recorded from both sides of Drake Passage. According to Ocaranza-Barrera et al. (Reference Ocaranza-Barrera, González-Wevar, Guillemin, Rosenfeld and Mansilla2019), I. cordata populations from the Antarctic Peninsula and South America represent distinguishable evolutionary units (COI-5P) or sister species separated at the end of the Miocene. Similarly, Hughey et al. (Reference Hughey, Leister, Gabrielson and Hommersand2020) reported phylogenetic analyses of G. skottsbergii (Rhodophyta) obtained from Argentina, Chile and along the Antarctic Peninsula. They proposed, using both molecular analyses and morpho-anatomical studies, that two distinct species are currently represented within G. skottsbergii. They also suggested that these should be assigned to the new genus Sarcopeltis, containing the generitype Sarcopeltis skottsbergii from southern South America and the new species Sarcopeltis antarctica from the Antarctic Peninsula. Young et al. (Reference Young, von Salm, Amsler, Lopez-Bautista, Amsler, McClintock and Baker2013), using cox1 and rbcL gene sequencing, found that Plocamium specimens from the WAP were divided into two closely related phylogroups, although not at the species level; such divergence may in future contribute to the discovery of cryptic speciation in this genus in Antarctica. Shilling et al. (Reference Shilling, Heiser, Amsler, McClintock and Baker2021) used metabolomic profiling linked to patterns of genetic diversification in Plocamium sp. from the WAP. They reported that genetic differences probably underlie some of the chemical diversity found in this taxon, further supporting the suggestion of hidden diversity.
The longest record of glaciation available in our study region is from King George Island, where glaciers first developed in the Miocene (Davies et al. Reference Davies, Hambrey, Smellie, Carrivick and Glasser2012). Between 23 and 3 Ma, whilst there were considerable ice-sheet fluctuations, the Antarctic Peninsula ice sheet remained as a series of ‘island’ ice caps, probably with exposed habitats thought to have provided refuges for biota, a feature that is a requirement for the evolution of the high levels of Antarctic and regional endemism present today (Convey et al. Reference Convey, Stevens, Hodgson, Smellie, Hillenbrand and Barnes2009, Reference Convey, Biersma, Casanova-Katny, Maturana, Oliva and Ruiz-Fernández2020). The mountains of the Antarctic Peninsula probably acted as a nucleus for glaciation (Jordan et al. Reference Jordan, Riley and Siddoway2020), promoting a sequence of cycles of deglaciation and recession from the outer continental shelf and resulting in the regional biodiversity hotspot in the WAP (particularly the SSI and the lower-latitude WAP), which our data and analyses indicate is mirrored in the macroalgae (Table S1).
Patterns in the distribution of macroalgal diversity in and around the Antarctic Peninsula are clear (Figs 3, 4 & 7). However, the susceptibility of this algal flora to abiotic changes has received little attention and should be further addressed. Taxonomic studies of macroalgae are an important element of environmental monitoring. Recently, the movement of species between Antarctic, sub-Antarctic and Patagonian ecosystems, enabled by floating rafts of large brown seaweeds, has received attention (López et al. Reference López, Macaya, Rivadeneira, Tala, Tellier and Thiel2018, Macaya et al. Reference Macaya, Tala, Hinojosa, Rothäusler, Gómez and Huovinen2020). The relationships between macroalgal assemblages from biogeographically distant regions have been investigated using molecular and phylogenetic techniques (Hommersand et al. Reference Hommersand, Moe, Amsler and Fredericq2009, Fraser et al. Reference Fraser, Zuccarello, Spencer, Salvatore and Garcia2013, Ocaranza-Barrera et al. Reference Ocaranza-Barrera, González-Wevar, Guillemin, Rosenfeld and Mansilla2019, Oliveira et al. Reference Oliveira, Pellizzari, Medeiros, Yokoya, Gómez and Huovinen2020), reporting evidence of recent invasions, new colonization events and the occurrence of macroalgal species previously thought to be endemic to Antarctica around some sub-Antarctic islands, Patagonia and New Zealand (Fraser et al. Reference Fraser, Zuccarello, Spencer, Salvatore and Garcia2013, Bussolini & Waters Reference Bussolini and Waters2015, Sanches et al. Reference Sanches, Pellizzari and Horta2016, Pellizzari et al. Reference Pellizzari, Santos-Silva, Medeiros, Oliveira, Yokoya, Pupo and Rosa2017, Reference Pellizzari, Rosa, Yokoya, Gómez and Huovinen2020). Together, these studies suggest higher connectivity of algal populations than previously appreciated (Sanches et al. Reference Sanches, Pellizzari and Horta2016, Fraser et al. Reference Fraser, Morrison, Hogg, Macaya, van Sebille and Ryan2018, Gómez & Huovinen Reference Gómez and Huovinen2015). Furthermore, species fluxes may take place in both north-to-south and south-to-north directions (González-Wevar et al. Reference González-Wevar, Segovia, Rosenfeld, Maturana, Jeldres and Pinochet2022).
The morphological taxonomic and biogeographical data presented here advance our understanding of Antarctic macroalgal diversity and distribution, in particular including novel data for the EAP region of the WS and providing new comparative information of the regions considered. Further molecular and phylogenetic studies are required for the better description of species distributions and the identification of cryptic species to document patterns of species connectivity and flux and to provide a robust baseline of Antarctic macroalgal diversity, as these sentinel organisms face rapid regional climatic and other environmental changes and are subject to increasing anthropogenic influences.
Acknowledgements
The authors are grateful to the Brazilian Antarctic Program (PROANTAR SECIRM), Brazilian Navy and the Polar Vessel Almirante Maximiano (H41) for providing logistical support to the oceanographic expeditions and to the sampling effort. We also thank Drs M. Liliana Quartino, Juan Lírio and Silvia Coria (Instituto Antartico Argentino) for enabling sampling on Vega Island, Drs Ana Paula Ferreira and Inara Mendonça for discussions on statistical analysis and Dr Mariana Cabral de Oliveira for careful revision of the manuscript.
Financial support
This work was supported by funding from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Fundação Araucária. PC is supported by Natural Environment Research Council (NERC) core funding to the British Antarctic Survey (BAS) ‘Biodiversity, Evolution and Adaptation’ Team.
Author contributions
FP developed the approach and led the writing of the paper, with contributions from FPB, PC and LHR. JPdSdM and FP jointly performed the morphological analyses and photography; JPdSdM, MCS-S and VSO and performed the statistical analyses.
Supplemental material
Five supplemental figures and a supplemental table will be found at https://doi.org/10.1017/S095410202300010X.